Peptide Storage, Handling, and Reconstitution: A Comprehensive Research Guide
A practical, evidence-based guide to proper peptide storage temperatures, reconstitution techniques, solvent selection, and stability optimization for research laboratories.
Introduction: Why Proper Handling Matters for Research Outcomes
The integrity of any peptide-based research program depends fundamentally on the quality and stability of the compounds being studied. Even the highest-purity research peptide can yield misleading or irreproducible results if improperly stored, reconstituted, or handled. Degradation products, aggregation, oxidation, and adsorption losses can all silently compromise experimental outcomes—turning what should be a well-controlled study into an exercise in troubleshooting artifacts.
This guide consolidates published research on peptide stability, pharmaceutical handling guidelines, and practical laboratory techniques to give researchers a comprehensive resource for maintaining peptide integrity from receipt through final use. Whether working with growth hormone secretagogues like Ipamorelin or Sermorelin, metabolic peptides such as Semaglutide and Tirzepatide, or tissue repair compounds like BPC-157 and TB-500, the principles of proper handling remain consistent across peptide classes.
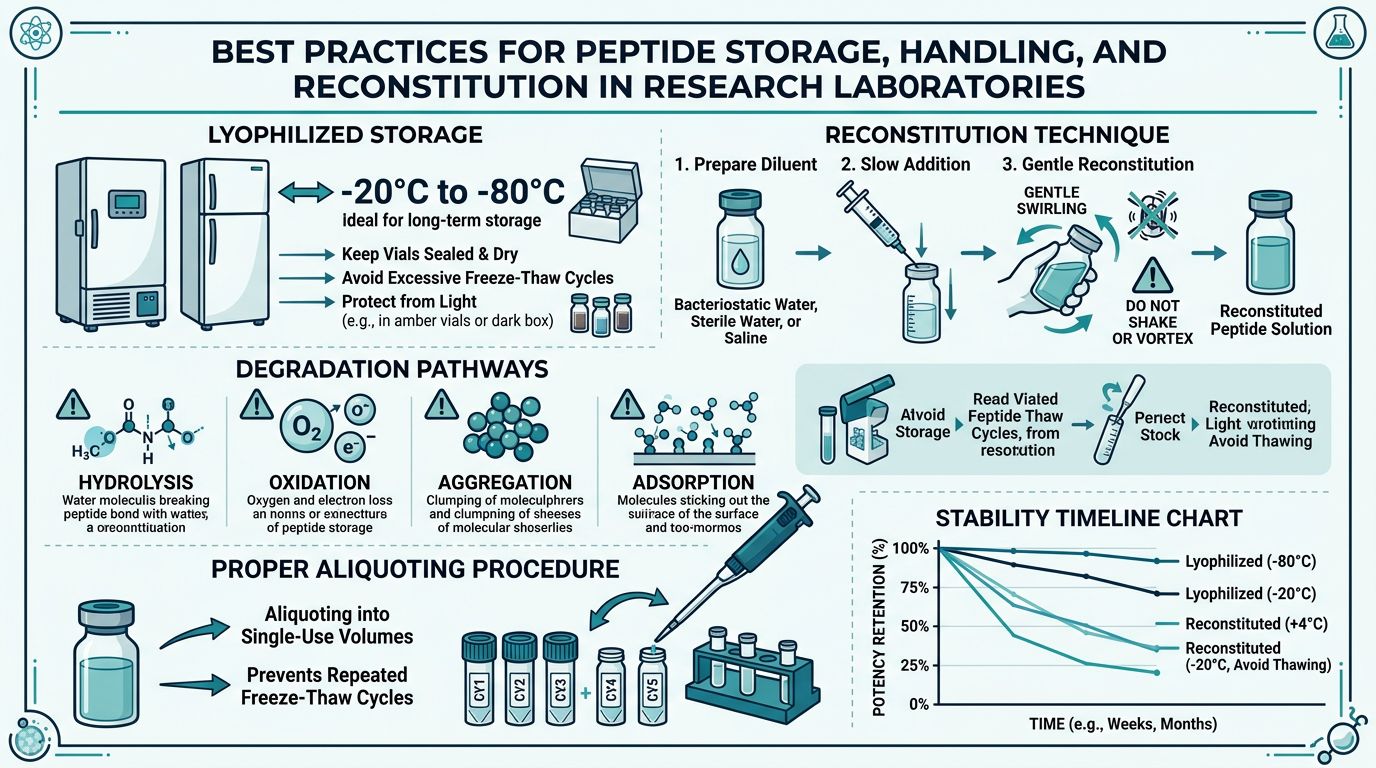
Understanding Peptide Degradation Pathways
Before discussing storage and handling protocols, it is essential to understand the chemical processes that degrade peptides. Research published in the Journal of Pharmaceutical Sciences has identified several primary degradation pathways that affect synthetic peptides in laboratory settings (Manning et al., 2010).
Hydrolysis
Hydrolysis—the cleavage of peptide bonds by water—is one of the most common degradation mechanisms. This process is accelerated by extremes of pH, elevated temperatures, and the presence of certain metal ions. Aspartate residues are particularly susceptible, undergoing spontaneous deamidation and isomerization at neutral pH. For peptides containing Asp-Pro or Asp-Gly sequences, hydrolysis can occur at measurable rates even at refrigerator temperatures over extended storage periods (Wakankar & Borchardt, 2006).
The practical implication is straightforward: peptides in aqueous solution degrade faster than lyophilized peptides. This is why most research peptides—including those from Alpha Carbon Labs—are supplied as lyophilized (freeze-dried) powders rather than pre-reconstituted solutions.
Oxidation
Methionine, cysteine, tryptophan, and histidine residues are vulnerable to oxidation by atmospheric oxygen, peroxides, and reactive metal ions. Oxidation can alter receptor binding affinity, reduce biological activity, and generate immunogenic degradation products. Research on therapeutic peptides has shown that even trace levels of dissolved oxygen can initiate oxidation cascades, particularly in metal-contaminated solutions (Li et al., 1995).
Peptides containing methionine residues—common in many growth hormone-related peptides—are especially susceptible. For example, oxidation of Met residues in growth hormone releasing peptides can significantly reduce their receptor binding affinity. This is one reason that inert gas overlays (nitrogen or argon) are recommended when preparing and storing peptide solutions.
Aggregation
Peptides can self-associate through hydrophobic interactions, hydrogen bonding, or disulfide exchange, forming oligomers, fibrils, or amorphous aggregates. Aggregation is promoted by high peptide concentrations, temperature fluctuations, agitation, and exposure to hydrophobic surfaces. Once initiated, aggregation can be autocatalytic—existing aggregates serve as nucleation sites for further aggregation (Wang et al., 2010).
This is particularly relevant for longer peptides and those with significant hydrophobic character. GLP-1 receptor agonists, for example, are known to form amyloid-like fibrils under certain conditions, which is why pharmaceutical formulations of compounds like Semaglutide include specific stabilizing excipients.
Adsorption
Peptides can adsorb onto container surfaces—glass, plastic, and metal—through electrostatic and hydrophobic interactions. At low concentrations (micromolar range), surface adsorption can result in significant loss of active peptide from solution. Studies have demonstrated that certain peptides can lose 10–50% of their concentration to container surfaces within hours of reconstitution if appropriate precautions are not taken (Senderoff et al., 1994).
Storage of Lyophilized Peptides
Lyophilized (freeze-dried) peptides are inherently more stable than their reconstituted counterparts. The removal of water dramatically slows hydrolysis, and the solid state limits molecular mobility, reducing aggregation and oxidation rates. However, improper storage can still compromise peptide integrity over time.
Temperature Requirements
The International Conference on Harmonisation (ICH) guidelines and extensive pharmaceutical stability data support the following temperature recommendations for lyophilized peptides:
| Storage Duration | Recommended Temperature | Notes |
|---|---|---|
| Short-term (1–4 weeks) | 2–8°C (refrigerator) | Acceptable for most peptides; keep sealed and desiccated |
| Medium-term (1–6 months) | -20°C (standard freezer) | Preferred for routine research storage |
| Long-term (6+ months) | -20°C to -80°C | Ultra-low for maximum stability; essential for sensitive peptides |
A critical principle: avoid freeze-thaw cycles with lyophilized powder. Each time a vial is removed from the freezer, condensation can form on the powder before the vial equilibrates to room temperature, introducing moisture that accelerates degradation. Allow vials to reach room temperature (approximately 15–20 minutes) before opening to prevent condensation on the hygroscopic powder (Carpenter et al., 1997).
Light Protection
Many peptides are photosensitive, with tryptophan and tyrosine residues absorbing UV light and undergoing photodegradation. Peptides containing these residues—which includes most biologically active peptides—should be stored in amber vials or wrapped in aluminum foil. Even standard laboratory fluorescent lighting delivers enough UV to measurably degrade sensitive peptides over weeks of exposure (Kerwin & Remmele, 2007).
Moisture Control
Lyophilized peptides are hygroscopic—they readily absorb atmospheric moisture. Moisture uptake reintroduces hydrolysis and can catalyze other degradation pathways. Storage with desiccant packets (silica gel) inside sealed containers is recommended. For particularly sensitive peptides or humid laboratory environments, storage under inert gas (nitrogen or argon) provides additional protection.
Container Selection for Storage
Original sealed vials with crimped caps provide the best barrier against moisture and oxygen. Once the original seal is broken, transfer to clean, low-binding microcentrifuge tubes (polypropylene) is recommended over standard glass vials, as many peptides adsorb more strongly to glass. Silanized glass vials are an alternative for laboratories that prefer glass containers.
Reconstitution: Principles and Protocols
Reconstitution—dissolving lyophilized peptide in an appropriate solvent—is the step where most handling errors occur. Proper technique is essential to maintain peptide activity and obtain accurate concentrations for research use.
Solvent Selection
The choice of reconstitution solvent depends on the peptide's physicochemical properties, particularly its solubility profile. The following table summarizes recommended solvents for common peptide classes:
| Peptide Characteristics | Primary Solvent | Alternative / Co-solvent | Example Peptides |
|---|---|---|---|
| Hydrophilic, net positive charge | Sterile water | 0.1% acetic acid if poorly soluble | BPC-157, Ipamorelin, Sermorelin |
| Hydrophilic, net negative charge | Sterile water | Dilute NH₄OH (0.1%) if needed | Glutathione |
| Hydrophobic or mixed character | 0.1% acetic acid | Small volume DMSO + dilute with water | Melanotan II, PT-141 |
| Very hydrophobic | DMSO (small volume first) | Then dilute with sterile water or buffer | Longer lipophilic peptides |
| Acylated / fatty acid conjugated | Sterile water at slightly alkaline pH | Dilute NaOH to adjust pH 8–9 | Semaglutide, Tirzepatide |
Bacteriostatic water (sterile water containing 0.9% benzyl alcohol) is widely used in research settings where the reconstituted peptide will be stored and drawn from multiple times. The benzyl alcohol acts as a preservative, inhibiting microbial growth that could contaminate multi-use vials. For single-use applications, sterile water for injection (WFI) is preferred, as it eliminates any potential interaction between the preservative and the peptide.
Reconstitution Technique: Step by Step
The following protocol is adapted from pharmaceutical handling guidelines and peptide chemistry best practices (Bachem AG Technical Bulletin; ICH Q5C):
Step 1: Preparation
- Allow the lyophilized vial to equilibrate to room temperature (15–20 minutes from freezer).
- Prepare your solvent, syringes, and a clean workspace. Wipe vial septum with an alcohol swab.
- Calculate the desired final concentration and required solvent volume. For example, to make a 5 mg/mL solution from a 10 mg vial, add 2 mL of solvent.
Step 2: Initial Dissolution
- Draw the calculated volume of solvent into a sterile syringe.
- Insert the needle through the vial septum and direct the solvent stream against the glass wall of the vial, not directly onto the peptide cake. This prevents foaming and mechanical disruption of the peptide.
- Add the solvent slowly. For larger volumes (>1 mL), add in portions.
Step 3: Dissolution
- Do NOT shake, vortex, or vigorously agitate the vial. Mechanical stress promotes aggregation and creates air-liquid interfaces that denature peptides.
- Gently swirl the vial in a circular motion, or roll it between your palms.
- Allow the peptide to dissolve passively. Most lyophilized peptides will dissolve within 1–5 minutes with gentle swirling. If the peptide does not dissolve within 10 minutes, the solvent choice may need to be reconsidered.
Step 4: Visual Inspection
- The resulting solution should be clear and free of visible particles. Opalescence or particulate matter may indicate aggregation or incomplete dissolution.
- If the solution is cloudy, do not add more solvent immediately. Instead, allow it to sit at room temperature for 15–30 minutes. Some peptides require time to fully hydrate and dissolve.
Step 5: Aliquoting (Recommended)
- For peptides that will be used over multiple days or weeks, dividing the reconstituted solution into single-use or few-use aliquots is strongly recommended.
- Use low-binding polypropylene microcentrifuge tubes.
- This minimizes freeze-thaw cycles and contamination risk from repeated needle punctures.
Concentration Calculations
Accurate concentration calculations are essential for reproducible research. The relationship is straightforward:
Concentration (mg/mL) = Peptide mass (mg) ÷ Solvent volume (mL)
For molar concentrations, you additionally need the molecular weight:
Concentration (mM) = [Peptide mass (mg) ÷ Molecular weight (g/mol)] ÷ Solvent volume (mL)
It is critical to account for the peptide content (purity and net peptide fraction) stated on the Certificate of Analysis. Research-grade peptides typically contain trifluoroacetate (TFA) counter-ions and residual moisture, meaning a vial labeled "10 mg" may contain only 7–8 mg of active peptide. The net peptide content, usually expressed as a percentage on the COA, should be factored into concentration calculations for quantitative studies. Alpha Carbon Labs provides detailed Certificates of Analysis with each product for this purpose.
Storage of Reconstituted Peptides
Once reconstituted, peptides are inherently less stable than their lyophilized form. The aqueous environment reactivates hydrolytic and oxidative degradation pathways. Proper post-reconstitution storage is critical for maintaining activity.
Temperature and Duration Guidelines
| Storage Condition | Expected Stability | Best Practices |
|---|---|---|
| Room temperature (20–25°C) | Hours to 1–2 days | Use immediately; avoid for storage |
| Refrigerated (2–8°C) | 1–4 weeks (peptide-dependent) | Standard for active-use solutions; use bacteriostatic water |
| Frozen (-20°C) | 1–3 months | Aliquot first; limit freeze-thaw to 2–3 cycles maximum |
| Ultra-frozen (-80°C) | 3–6+ months | Best for long-term storage of reconstituted aliquots |
Research by Cleland et al. (1993) demonstrated that repeated freeze-thaw cycling can cause significant peptide degradation through several mechanisms: ice crystal formation creates mechanical stress and concentrates solutes at ice-liquid interfaces, promoting aggregation; the pH of buffered solutions can shift dramatically during freezing as buffer components crystallize selectively; and the large surface area of ice provides nucleation sites for aggregation.
The practical recommendation is clear: aliquot reconstituted peptides into single-use volumes before freezing. This single practice eliminates the most common cause of reconstituted peptide degradation in research settings.
Peptide-Specific Stability Considerations
While general guidelines apply broadly, certain peptide classes have specific stability profiles worth noting:
Growth Hormone Secretagogues (Ipamorelin, CJC-1295, Sermorelin, Tesamorelin, GHRP-2, GHRP-6): These peptides are generally stable in aqueous solution at refrigerator temperature for 2–4 weeks. CJC-1295 with DAC (Drug Affinity Complex) benefits from the albumin-binding moiety that provides some additional stabilization in solution. MK-677 (Ibutamoren), being a non-peptide peptidomimetic, is inherently more stable than true peptides.
GLP-1 Receptor Agonists (Semaglutide, Tirzepatide, Retatrutide): The acylated fatty acid side chains on these peptides contribute to their extended pharmacokinetic profiles but can also promote aggregation at higher concentrations. Pharmaceutical formulations of semaglutide (Ozempic®) include a phosphate buffer at pH 7.4 with phenol and polysorbate 80 as stabilizers. For research-grade material, reconstitution in bacteriostatic water with minimal agitation is effective. These should be stored refrigerated and used within 28 days of reconstitution, consistent with FDA-approved product labeling for the pharmaceutical equivalents (Novo Nordisk, Ozempic® Prescribing Information, 2023).
Tissue Repair Peptides (BPC-157, TB-500): BPC-157 is notable for its exceptional stability. Unlike most peptides, BPC-157 is stable in gastric juice and resists enzymatic degradation—properties that derive from its gastric origin. In aqueous solution, it remains stable at refrigerator temperature for extended periods relative to other peptides. The BPC-157 + TB-500 blend and BPC-157 + TB-500 + GHK-Cu blend follow the same storage principles as their individual components.
Melanocortin Peptides (Melanotan II, PT-141): These peptides contain methionine residues susceptible to oxidation. Storage under inert gas and protection from light are particularly important. Reconstituted solutions should be used within 2–3 weeks when refrigerated.
Neuropeptides (Selank, Semax, Dihexa): Many neuropeptides are smaller and may adsorb more readily to container surfaces at low concentrations. Using low-binding tubes and slightly higher concentrations can mitigate losses.
Immune Peptides (Thymosin Alpha-1, LL-37): LL-37, as an antimicrobial peptide, has amphipathic properties that make it prone to aggregation at higher concentrations. Keep reconstituted concentrations moderate and store refrigerated.
Advanced Handling Techniques
Working with DMSO
Dimethyl sulfoxide (DMSO) is an effective solvent for hydrophobic peptides that resist dissolution in aqueous solutions. However, DMSO must be used judiciously:
- Use the minimum effective volume. Dissolve the peptide in a small volume of DMSO (typically 50–100 µL for a 5–10 mg vial), then dilute with aqueous solvent to the final volume. The final DMSO concentration should generally not exceed 1–5% to minimize solvent effects in biological assays.
- DMSO is not inert. At elevated temperatures, DMSO can oxidize methionine residues. Store DMSO stock solutions at -20°C or below.
- DMSO prevents true freezing at standard freezer temperatures. Solutions containing >10% DMSO will not freeze at -20°C, which can be advantageous (no freeze-thaw damage) or problematic (reduced stability benefit of freezing).
pH Optimization
Peptide solubility and stability are strongly pH-dependent. Most peptides have optimal stability near their isoelectric point (pI) for storage, but may require different pH conditions for solubility. As a general framework:
- Acidic peptides (net negative charge at neutral pH): Try dissolving at slightly basic pH (dilute ammonium hydroxide, pH 8–9).
- Basic peptides (net positive charge at neutral pH): Try dissolving at slightly acidic pH (0.1% acetic acid, pH 3–4).
- For research applications, PBS (phosphate-buffered saline, pH 7.4) is commonly used but may not be optimal for all peptides—phosphate can catalyze deamidation of asparagine residues (Wakankar & Borchardt, 2006).
Sterile Technique
Microbial contamination is a practical concern for reconstituted peptides, particularly multi-use vials stored for days to weeks. Best practices include:
- Using bacteriostatic water for multi-use reconstitution.
- Swabbing vial septa with 70% isopropanol before each needle puncture.
- Using a fresh sterile needle for each withdrawal.
- Working in a laminar flow hood when possible, especially for cell culture applications.
- Filtering through 0.22 µm syringe filters if sterility is critical for the application.
Common Mistakes and Troubleshooting
Mistake 1: Vortexing or Shaking Reconstituted Peptides
This is perhaps the most common handling error. Vigorous agitation creates air-liquid interfaces where peptide molecules unfold and aggregate. The high surface tension at these interfaces acts as a denaturant. Research has shown that even brief vortexing can cause measurable aggregation in susceptible peptides (Mahler et al., 2005). Always use gentle swirling or rolling motions.
Mistake 2: Using the Wrong Solvent
Adding sterile water to a hydrophobic peptide may result in visible precipitate or apparent dissolution followed by slow aggregation over hours. If a peptide does not dissolve in water within 10 minutes of gentle swirling, stop and consider alternative solvents rather than adding more water (which dilutes the final concentration without improving solubility).
Mistake 3: Storing Reconstituted Peptides at Room Temperature
The Arrhenius equation predicts that reaction rates roughly double for every 10°C increase in temperature. A peptide with a 4-week refrigerated stability may last only 3–4 days at room temperature. Always return reconstituted peptides to refrigeration or freezer storage immediately after withdrawing the needed volume.
Mistake 4: Repeated Freeze-Thaw Cycles
Each freeze-thaw cycle exposes the peptide to mechanical stress from ice crystal formation, pH shifts from selective buffer crystallization, and concentration effects at ice-liquid interfaces. Studies on pharmaceutical proteins have demonstrated measurable activity loss after as few as 3–5 freeze-thaw cycles (Bhatnagar et al., 2007). Aliquoting before the first freeze eliminates this problem entirely.
Mistake 5: Ignoring the Certificate of Analysis
The COA contains critical information: actual peptide content (which may differ from the labeled amount due to counter-ions and moisture), purity by HPLC, amino acid analysis results, and mass spectrometry confirmation. Researchers who skip the COA may miscalculate concentrations by 20–30%. Always review the COA before reconstitution and factor net peptide content into your calculations. All Alpha Carbon Labs products include detailed COA documentation.
Mistake 6: Contaminating Multi-Use Vials
Inserting a used needle, touching the septum with ungloved fingers, or drawing from a vial in a non-sterile environment introduces microorganisms that can both contaminate experiments and degrade the peptide through enzymatic activity. Strict aseptic technique is non-negotiable for multi-use vials.
Stability Testing: How to Verify Your Peptide Integrity
For critical research applications, verifying that stored peptides maintain their integrity is important. Several analytical methods are available:
Reversed-Phase HPLC (RP-HPLC): The gold standard for assessing peptide purity and detecting degradation products. A shift in retention time or the appearance of new peaks indicates degradation. Comparing a freshly reconstituted sample against a stored sample provides a straightforward stability assessment (Mant & Hodges, 2006).
Mass Spectrometry (MS): MALDI-TOF or ESI-MS can confirm molecular weight and detect oxidation (+16 Da for single oxidation), deamidation (+1 Da), and truncation products. This is the most definitive method for identifying specific degradation pathways.
Visual Inspection: While crude, visual inspection catches gross problems—precipitation, cloudiness (aggregation), color changes (oxidation of tryptophan produces yellow-brown discoloration). A clear, colorless solution that was originally clear and colorless is a necessary (but not sufficient) indicator of stability.
Bioactivity Assays: Ultimately, the most relevant stability test is whether the peptide retains biological activity. For cell-based assays, comparing dose-response curves from fresh versus stored peptide preparations reveals functional degradation that chemical analysis might miss.
Special Considerations by Application
In Vitro Cell Culture Studies
Peptides used in cell culture require sterile filtration (0.22 µm) after reconstitution. DMSO concentrations above 0.1–0.5% may affect cell viability and should be accounted for in experimental controls. Serum-containing media can stabilize peptides through protein binding but may also interfere with binding assays. For receptor binding studies, serum-free conditions are typically preferred despite reduced peptide stability.
In Vivo Animal Studies
For injection studies, peptides are typically reconstituted in bacteriostatic water or normal saline (0.9% NaCl). Solutions should be isotonic and near physiological pH (7.2–7.4) to minimize injection site irritation. Prepare fresh solutions daily when possible; if multi-day preparation is necessary, aliquot and freeze individual doses.
Analytical / Structural Studies
For circular dichroism (CD), NMR, or X-ray crystallography, peptide purity requirements are higher (>95–98%). Use the highest available grade of water (Type 1 / Milli-Q) and freshly prepared buffers. Some structural techniques require specific deuterated solvents (D₂O for NMR) or specific buffer systems that are compatible with the analytical method.
Equipment and Supply Recommendations
Proper peptide handling requires appropriate laboratory supplies. The following equipment list covers the essentials:
| Item | Purpose | Key Specifications |
|---|---|---|
| Low-binding microcentrifuge tubes | Aliquoting and storage | Polypropylene, 0.5 mL or 1.5 mL |
| Insulin syringes | Accurate small-volume measurement | U-100, 0.5 mL or 1.0 mL, 29–31 gauge |
| Sterile syringe filters | Sterilization of reconstituted peptides | 0.22 µm PVDF or PES membrane |
| Bacteriostatic water | Multi-use reconstitution | 0.9% benzyl alcohol, USP grade |
| Alcohol prep pads | Septum disinfection | 70% isopropanol |
| Desiccant packets | Moisture protection during storage | Silica gel, indicating type |
| Amber vials or aluminum foil | Light protection | UV-blocking |
| Calibrated pipettes | Accurate volume measurement for concentrations | P20, P200, P1000 with certified tips |
Shipping and Receiving Best Practices
Peptide stability during shipping is an often-overlooked consideration. Lyophilized peptides are generally stable during standard shipping (ambient temperature for 3–5 days), as the absence of water prevents the primary degradation pathways. However, extreme heat during summer months can accelerate solid-state degradation for sensitive peptides.
Upon receipt of peptides:
- Inspect packaging for damage or evidence of temperature excursion (if temperature indicators are included).
- Transfer lyophilized vials to appropriate storage temperature immediately—do not leave at room temperature longer than necessary.
- Review the Certificate of Analysis and verify the product matches your order (peptide identity, purity, quantity).
- Record the lot number and storage date for your laboratory records.
- Alpha Carbon Labs ships all peptides with appropriate packaging and provides detailed shipping information to ensure product integrity during transit.
Quick Reference: Reconstitution Decision Tree
For researchers who need a rapid decision framework, the following process covers the most common scenarios:
1. Is the peptide soluble in water?
- Most small, hydrophilic peptides (BPC-157, Ipamorelin, Sermorelin, CJC-1295, GHRP-2, GHRP-6, TB-500, AOD9604, Epithalon, Selank, Semax) → Use bacteriostatic water or sterile water.
2. Does the peptide have hydrophobic character or poor aqueous solubility?
- Melanocortin peptides (Melanotan II, PT-141), some longer peptides → Try 0.1% acetic acid first. If still insoluble, use small volume DMSO followed by aqueous dilution.
3. Is the peptide acylated with a fatty acid chain?
- Semaglutide, Tirzepatide, Retatrutide → Use bacteriostatic water; gentle dissolution required; allow extra time. If difficult to dissolve, slight pH adjustment to 8.0 with dilute NaOH may help.
4. What concentration should I target?
- General recommendation: 1–5 mg/mL for storage stocks. Dilute to working concentrations immediately before use. Higher concentrations improve stability (less surface adsorption as a percentage) but may promote aggregation for certain peptides.
Conclusion: Investment in Handling Pays Research Dividends
The difference between a successful peptide research program and one plagued by irreproducible results often comes down to handling practices. The principles outlined in this guide—proper storage temperatures, appropriate solvent selection, gentle reconstitution technique, aliquoting to avoid freeze-thaw cycles, and aseptic handling—represent a modest investment of time and attention that pays significant dividends in data quality and reproducibility.
Every peptide that degrades due to improper handling represents wasted research funds and, more importantly, potentially misleading data that can misdirect future experiments. By implementing the evidence-based practices described here, researchers can maximize the value of their peptide reagents and the reliability of their experimental outcomes.
For researchers seeking high-purity peptides with comprehensive documentation, Alpha Carbon Labs provides detailed Certificates of Analysis, proper lyophilization, and rigorous quality control to ensure that your research compounds arrive in optimal condition—giving you the best possible starting point for the careful handling practices described in this guide.
All peptides referenced in this article are available from Alpha Carbon Labs for research purposes only. These products are not intended for human consumption and are sold exclusively for legitimate scientific research.
References
- 1. Manning, M. C., et al. (2010). Stability of Protein Pharmaceuticals: An Update. Pharmaceutical Research, 27(4), 544-575.
- 2. Wakankar, A. A., & Borchardt, R. T. (2006). Formulation considerations for proteins susceptible to asparagine deamidation and aspartate isomerization. Journal of Pharmaceutical Sciences, 95(11), 2321-2336.
- 3. Li, S., et al. (1995). Chemical Instability of Protein Pharmaceuticals: Mechanisms of Oxidation and Strategies for Stabilization. Biotechnology and Bioengineering, 48(5), 490-500.
- 4. Wang, W., et al. (2010). Immunogenicity of protein aggregates—concerns and realities. International Journal of Pharmaceutics, 431(1-2), 1-11.
- 5. Senderoff, R. I., et al. (1994). Aqueous stability of human epidermal growth factor 1-48. Pharmaceutical Research, 11(12), 1712-1720.
- 6. Carpenter, J. F., et al. (1997). Rational design of stable lyophilized protein formulations: some practical advice. Pharmaceutical Research, 14(8), 969-975.
- 7. Kerwin, B. A., & Remmele, R. L. (2007). Protect from light: Photodegradation and protein biologics. Journal of Pharmaceutical Sciences, 96(6), 1468-1479.
- 8. Cleland, J. L., et al. (1993). The development of stable protein formulations: a close look at protein aggregation, deamidation, and oxidation. Critical Reviews in Therapeutic Drug Carrier Systems, 10(4), 307-377.
- 9. Mahler, H. C., et al. (2005). Induction and analysis of aggregates in a liquid IgG1-antibody formulation. European Journal of Pharmaceutics and Biopharmaceutics, 59(3), 407-417.
- 10. Bhatnagar, B. S., et al. (2007). Protein stability during freezing: separation of stresses and mechanisms of protein stabilization. Pharmaceutical Development and Technology, 12(5), 505-523.
- 11. Mant, C. T., & Hodges, R. S. (2006). Mixed-mode hydrophilic interaction/cation-exchange chromatography (HILIC/CEX) of peptides and proteins. Journal of Separation Science, 31(9), 1573-1584.
- 12. Novo Nordisk. (2023). Ozempic® (semaglutide) Prescribing Information. U.S. Food and Drug Administration.
- 13. ICH Harmonised Tripartite Guideline. (1995). Quality of Biotechnological Products: Stability Testing of Biotechnological/Biological Products Q5C.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.