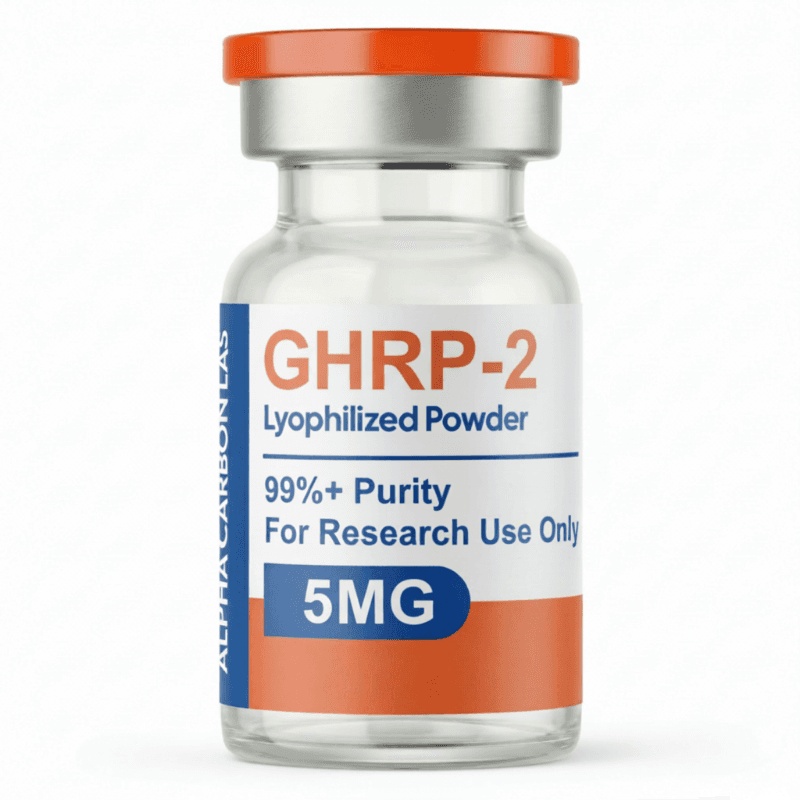
5MG
This product is for research purposes only. Not for human consumption.
Purity: >98% (HPLC verified)
Formulation: Lyophilized powder
Molecular Formula: C45H55N9O6
Molecular Weight: 817.97 g/mol
CAS Number: 158861-67-7
PubChem CID: N/A
Research Contents
GHRP-2
Overview
GHRP-2 (Growth Hormone Releasing Peptide-2), also known as Pralmorelin, is a synthetic hexapeptide with the sequence D-Ala-D-β-Nal-Ala-Trp-D-Phe-Lys-NH2 that acts as a potent growth hormone secretagogue by binding to and activating the ghrelin receptor (GHS-R1a). It was developed in the 1990s as an evolution of earlier GHRP compounds, offering enhanced potency and a more favorable effect profile.
Key Characteristics
GHRP-2 is considered one of the most potent members of the GHRP family, producing robust growth hormone release while having moderate effects on appetite stimulation - less pronounced hunger effects than GHRP-6 but more than Ipamorelin. This intermediate profile makes it a balanced choice for research applications where both GH stimulation and manageable appetite effects are desired.
Development History
The peptide was originally developed by Kaken Pharmaceutical and has been studied extensively in Japan and other countries for potential therapeutic applications including growth hormone deficiency, aging-related GH decline, and various catabolic conditions. It has been approved for diagnostic use in some countries under the name Pralmorelin.
Mechanism of Action
Like other GHRPs, GHRP-2 works through a mechanism distinct from and complementary to growth hormone releasing hormone (GHRH). While GHRH acts through its own receptor to stimulate GH synthesis and release, GHRP-2 acts through the ghrelin receptor to amplify GH pulsatility. When these two pathways are activated simultaneously, they produce synergistic effects with GH release exceeding the sum of individual responses.
Mechanism of Action
GHRP-2 exerts its growth hormone releasing effects primarily through binding to and activation of the growth hormone secretagogue receptor type 1a (GHS-R1a), the same receptor that responds to the endogenous hormone ghrelin.
Receptor Activation
Upon binding to GHS-R1a on somatotroph cells in the anterior pituitary, GHRP-2 triggers G-protein coupled receptor signaling cascades. The receptor couples primarily to Gq/11 proteins, leading to phospholipase C activation, generation of inositol trisphosphate (IP3) and diacylglycerol (DAG), and subsequent calcium mobilization from intracellular stores.
GH Release Cascade
The resulting increase in intracellular calcium triggers exocytosis of GH-containing secretory granules, producing acute GH release. Additionally, GHRP-2 stimulates GH gene transcription, supporting sustained GH synthesis. The peptide increases both the amplitude of GH pulses and, to some extent, pulse frequency, producing a robust stimulation of the somatotropic axis.
Synergy with GHRH
GHRP-2's effects are additive to and synergistic with GHRH, which works through a separate receptor (GHRH-R) coupled to Gs proteins and the cAMP pathway. Combined administration of GHRP-2 and GHRH analogs produces GH release substantially greater than either agent alone, a property that has been exploited in both research and diagnostic applications.
Appetite Effects
Beyond the pituitary, GHRP-2 activates ghrelin receptors in the hypothalamus, producing moderate orexigenic (appetite-stimulating) effects. These effects are less pronounced than with GHRP-6 but more significant than with Ipamorelin, positioning GHRP-2 in an intermediate category among GHRPs regarding hunger stimulation.
Secondary Hormonal Effects
GHRP-2 produces mild, transient increases in cortisol and prolactin through ghrelin receptor activation in corticotrophs and lactotrophs. These effects are generally not clinically significant at typical doses and normalize between administrations.
Research Findings
GHRP-2 has been extensively studied in both preclinical and clinical research, with a substantial body of published literature characterizing its pharmacology, efficacy, and safety profile.
Clinical Pharmacology
Clinical studies in healthy subjects have demonstrated dose-dependent GH release following GHRP-2 administration. Intravenous or subcutaneous doses of 1-2 mcg/kg produce significant elevations in serum GH, with peak levels typically occurring 15-30 minutes post-administration. The GH response to GHRP-2 is reproducible and maintained with repeated dosing, without significant tachyphylaxis.
Diagnostic Applications
GHRP-2 (as Pralmorelin) has been approved in Japan and studied elsewhere as a diagnostic agent for assessing pituitary GH secretory capacity. The GHRP-2 stimulation test can differentiate between GH deficiency of hypothalamic versus pituitary origin and has been used to evaluate patients with suspected growth hormone deficiency.
Combination Studies
Research combining GHRP-2 with GHRH analogs has documented the synergistic GH-releasing effects of this combination. Studies show that GHRP-2 plus GHRH produces GH responses 2-3 fold greater than either agent alone, providing a powerful tool for GH axis stimulation and diagnostic testing.
Age-Related Research
Studies in elderly subjects have shown that GHRP-2 can partially restore youthful patterns of GH secretion in aging individuals who exhibit age-related decline in GH output. This has led to research interest in GHRP-2 for addressing somatopause and age-related changes in body composition and function.
Body Composition Studies
Research examining body composition effects has demonstrated that chronic GHRP-2 administration can increase lean body mass, reduce fat mass, and improve markers of metabolic health. Studies in various populations including elderly subjects and those with catabolic conditions have shown favorable effects on body composition parameters.
Sleep Studies
Research has also explored GHRP-2's effects on sleep architecture, given the relationship between GH secretion and sleep. Studies suggest GHRP-2 administration may enhance slow-wave sleep and improve sleep quality, consistent with GH's role in sleep regulation.
Research Applications
- Growth hormone secretion research
- Age-related GH decline studies
- Body composition research
- Sleep quality and architecture studies
- Pituitary function diagnostics
- GH deficiency assessment research
- Sarcopenia and muscle wasting studies
- Metabolic function research
- GHRH synergy and combination studies
- Ghrelin receptor pharmacology research
Safety Profile
GHRP-2 has demonstrated generally acceptable safety and tolerability across numerous clinical studies, though it has not been approved for therapeutic use in most jurisdictions outside of its diagnostic application.
Common Effects
The most frequently reported effects include increased appetite and hunger (moderate compared to GHRP-6), transient flushing or warmth sensation following injection, mild water retention in some subjects, and occasional dizziness or lightheadedness. These effects are generally dose-related and manageable.
Hormonal Effects
GHRP-2 produces mild, transient elevations in cortisol and prolactin that are generally not clinically significant and normalize between doses. These effects are less pronounced than with some other GHRPs and are not typically problematic at standard research doses.
Injection Site Reactions
With subcutaneous administration, local injection site reactions including redness, pain, or itching may occur but are typically mild and transient. Proper injection technique and site rotation help minimize these effects.
Long-Term Considerations
Theoretical concerns with chronic GHRP-2 use parallel those of other GH secretagogues, including potential effects of sustained GH/IGF-1 elevation on glucose metabolism, tissue growth, and cardiovascular function. Long-term safety data in humans remains limited, as most clinical studies have been of relatively short duration.
Quality Assurance
As with all research peptides, quality and purity are important considerations. Ensuring peptides are obtained from reputable sources with appropriate testing and documentation helps minimize risks associated with impurities or degradation products.
Scientific References
Customer Reviews
No reviews yet. Be the first to review this product!
Please sign in to write a review.
Research Use Only
This product is intended for research purposes only and is not for human consumption, therapeutic use, or diagnostic applications. Please ensure compliance with all applicable regulations and institutional guidelines.