Optimizing GH Signaling: A Comparative Analysis of CJC-1295, Ipamorelin, and Tesamorelin
A technical investigation into the pharmacokinetics of CJC-1295, Ipamorelin, and Tesamorelin, exploring half-life differences and the synergy of GHRH/GHRP combinations.
The Evolution of Growth Hormone Modulation
In the field of endocrinology and peptide research, the modulation of the growth hormone (GH) axis remains one of the most sophisticated areas of study. For researchers, the objective is rarely about simply increasing GH levels; rather, it is about optimizing the physiological pulse, timing, and bioavailability of these signals to mimic or enhance natural biological patterns. Central to this pursuit are three specific peptides: CJC-1295, Ipamorelin, and Tesamorelin.
While all three interact with the GH axis, their mechanisms of action, half-lives, and physiological impacts differ significantly. This article provides a technical deep-dive into the pharmacokinetics and pharmacodynamics of these GHRH (Growth Hormone Releasing Hormone) analogs and GHRPs (Growth Hormone Releasing Peptides), exploring how they can be strategically utilized in a research setting.
The Growth Hormone Axis: A Primer
The secretion of GH from the anterior pituitary gland is controlled by a dual-regulatory system:
- GHRH (Growth Hormone Releasing Hormone): Secreted by the hypothalamus, it stimulates BH synthesis and release.
- Somatostatin: The "braking" hormone that inhibits GH release.
- Ghrelin/GHRPs: A secondary pathway that amplifies the GHRH signal and suppresses somatostatin tone.
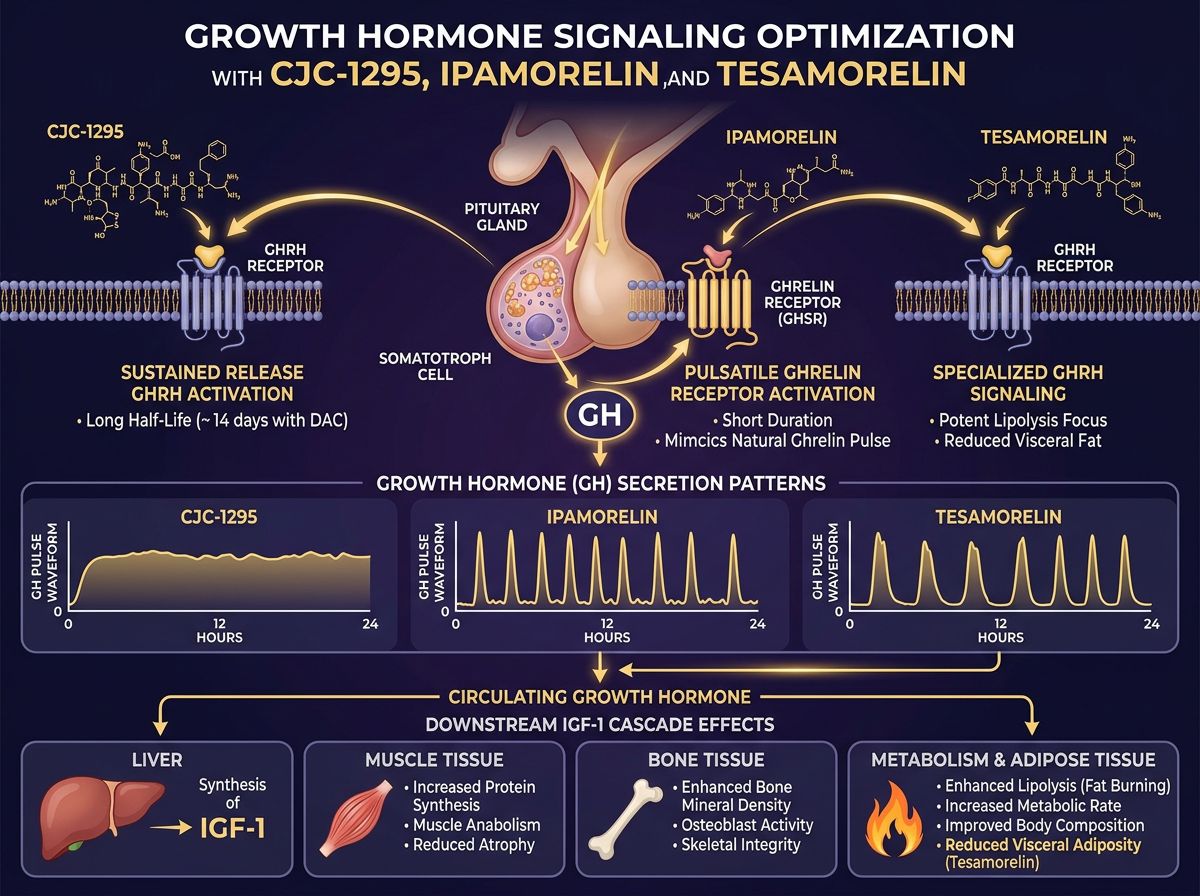
CJC-1295: The GHRH Powerhouse
CJC-1295 is a synthetic analog of GHRH, specifically designed to extend the half-life of the native hormone. In its original form, GHRH has a half-life of mere minutes due to rapid enzymatic degradation by dipeptidyl peptidase-IV (DPP-IV). CJC-1295 exists in two primary research iterations: with and without the Drug Affinity Complex (DAC).
CJC-1295 No DAC (Modified GRF 1-29)
Often referred to as Mod GRF 1-29, this version contains 29 amino acids and has been modified at four specific positions to resist DDP-IV degradation. Even with these modifications, the half-life remains relatively short—approximately 30 minutes. This necessitates a pulsatile approach to research, mimicking the natural "bursts" of GH secretion observed in mammalian models. For researchers interested in precise control over GH spikes, CJC-1295 No DAC is the gold standard.
CJC-1295 with DAC
The addition of the Drug Affinity Complex (DAC) allows the peptide to bind covalently to albumin in the bloodstream. This significantly extends the half-life to approximately 6-8 days. While this provides a consistent baseline of GH elevation, it alters the natural pulsatile rhythm.
Learn more about our manufacturing standards at peptide synthesis.
Ipamorelin: The Selective Secretagogue
Ipamorelin is a pentapeptide that acts as a Ghrelin/Growth Hormone Secretagogue Receptor (GHSR) agonist. Unlike earlier GHRPs such as GHRP-2 or GHRP-6, Ipamorelin is highly selective.
Pharmacological Precision
The primary advantage of Ipamorelin in a research context is its lack of "collateral" endocrine stimulation. Many GHRPs cause significant increases in:
- Cortisol: The primary stress hormone.
- Prolactin: Which can impact reproductive health.
- Ghrelin (Hunger): Causing intense appetite stimulation.
The Synergy: CJC-1295 and Ipamorelin
In many research protocols, CJC-1295 and Ipamorelin are administered together. This is not redundant; rather, it is a synergistic application of two different pathways. When a GHRH analog and a GHRP are introduced simultaneously, the resulting GH release is significantly greater than the sum of their individual effects. This is known as "synergistic GH release."
The GHRH analog (CJC) provides the signal to the pituitary to produce GH, while the GHRP (Ipamorelin) suppresses somatostatin (the inhibitor) and amplifies the GHRH signal. This "one-two punch" ensures a robust pulse that overcomes natural inhibitory barriers.
Tesamorelin: Specialized GHRH for Metabolic Research
Tesamorelin is a 44-amino acid analog of GHRH, modified by the addition of a trans-3-hexenoic acid group. Historically, Tesamorelin has been studied for its specific effects on visceral adipose tissue (VAT) and metabolic health.
V-Adipose Tissue and Lipolysis
Unlike general GH secretagogues, Tesamorelin has demonstrated a unique affinity for reducing abdominal fat without significantly affecting subcutaneous fat. Research indicates this is likely due to its higher potency in stimulating the IGF-1 (Insulin-like Growth Factor 1) pathway specifically related to lipid metabolism. For researchers focusing on metabolic syndrome, cardiovascular risk, or lipodystrophy models, Tesamorelin is often the primary subject of study.
Comparison Table: GHRH Analogs and Secretagogues
| Peptide | Type | Half-Life | Primary Mechanism | Impact on Cortisol/Prolactin |
|---|---|---|---|---|
| CJC-1295 No DAC | GHRH Analog | ~30 Minutes | Pituitary GH stimulator | Negligible |
| CJC-1295 w/ DAC | GHRH Analog | 6-8 Days | Continuous GH elevation | Low |
| Ipamorelin | GHRP / GHSR Agonist | ~2 Hours | Somatostatin suppression | None |
| Tesamorelin | GHRH Analog | ~30 Minutes | Lipolytic focused GH release | Negligible |
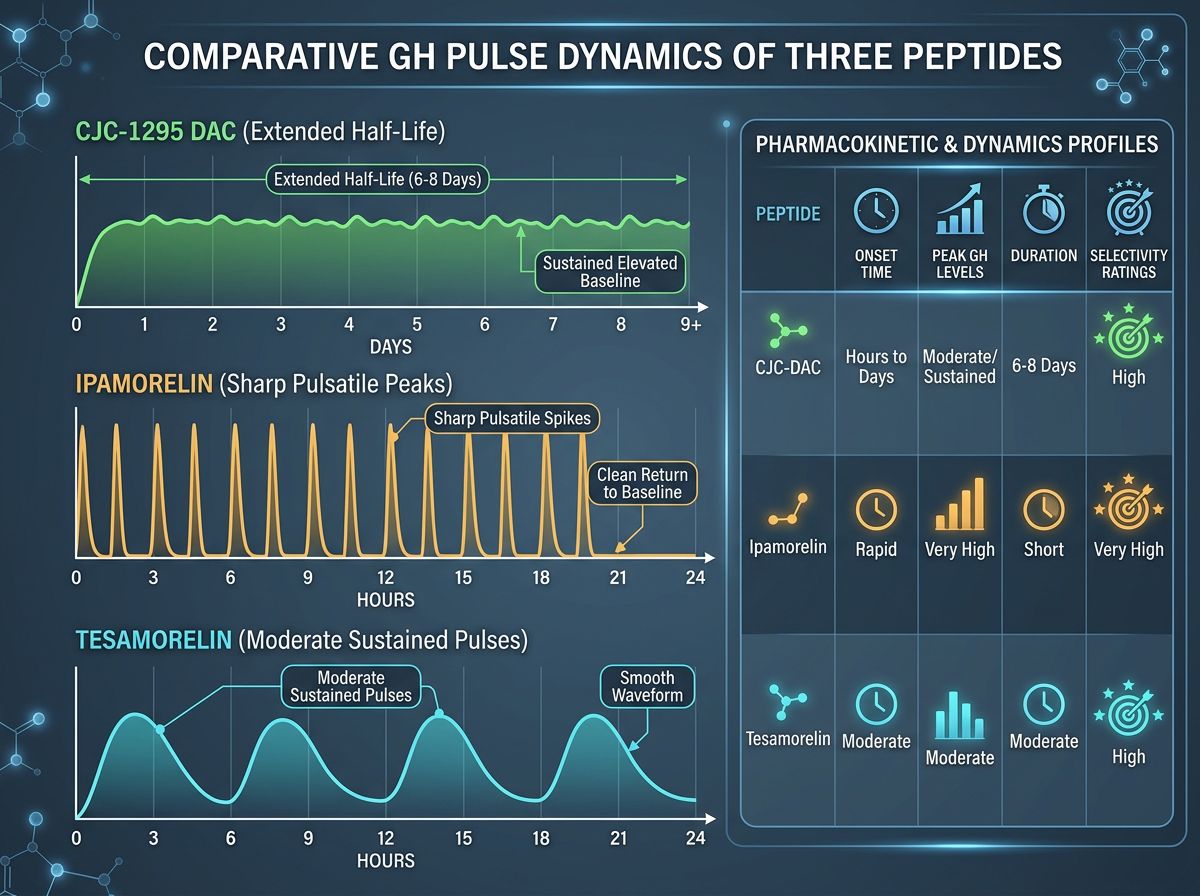
Comparative Half-Life and Pulse Dynamics
One of the most critical decisions a researcher faces is choosing between pulsatile (short-acting) and continuous (long-acting) GH stimulation.
Pulsatile stimulation (using CJC-1295 No DAC and Ipamorelin) mimics the human body's circadian rhythm, where GH is released in 5-6 bursts per day, primarily during deep sleep. Continuous stimulation (CJC-1295 with DAC) leads to "GH bleed"—a state where GH levels are constantly elevated. While effective for muscle mass research, continuous elevation has been scrutinized for potentially down-regulating pituitary sensitivity over time.
Research Applications in Longevity and Recovery
Beyond metabolic health, these peptides are frequently researched for their regenerative properties. Increased GH levels lead to higher circulating IGF-1, which stimulates:
- Protein synthesis and cellular repair.
- Bone mineral density enhancement.
- Improved recovery from soft tissue injuries (often paired with BPC-157).
- Enhanced mitochondrial function (often studied alongside SS-31 or MOTS-c).
Advanced Synergies: Designing Research Protocols
Modern research often extends beyond single-peptide analysis. To observe maximum physiological transformation, researchers may combine GH secretagogues with other reparative agents. For example:
- For Injury Recovery: Combining BPC-157 + TB-500 with CJC-1295/Ipamorelin to synchronize systemic GH elevation with localized tissue healing.
- For Fat Loss: Utilizing Tesamorelin in conjunction with AOD9604, a fragment of the GH molecule specifically responsible for lipolysis without affecting blood glucose levels.
- For Cognitive Longevity: Pairing GHRH analogs with neuroprotective agents like Cerebrolysin or Dihexa.
Metabolic Health and Insulin Sensitivity
A significant concern in GH research is the impact on insulin sensitivity. High levels of Growth Hormone can lead to insulin resistance due to increased free fatty acid mobilization. Researchers often manage this by using selective peptides like Ipamorelin, which has a lower risk of glycemic disruption compared to exogenous HGH. Additionally, incorporating metabolic sensitizers like NAD+ or 5-amino-1mq can provide a more comprehensive metabolic profile.
Quality Control and Research Purity
The validity of any comparative study depends entirely on the purity of the peptides used. Contaminants or under-dosed vials can lead to inaccurate data regarding half-life and secretion patterns. At Alpha Carbon Labs, we ensure every batch undergoes rigorous testing. Researchers can access our quality control protocols and COA documents to verify the structural integrity of our CJC-1295, Ipamorelin, and Tesamorelin.
Final Considerations for Researchers
When selecting a protocol for GH signaling research, the choice depends on the specific end-point goals:
- If the goal is to mimic natural physiology with minimal side effects, the CJC-1295 No DAC + Ipamorelin combination is the most scientifically supported pathway.
- If the objective is metabolic regulation and visceral fat reduction, Tesamorelin offers the most targeted results.
- If the protocol requires infrequent administration and constant GH elevation, CJC-1295 with DAC may be appropriate, though it requires careful monitoring for pituitary desensitization.
References
- 1. Vitiello, M. V., et al. (2006). Growth hormone-releasing hormone improves the cognitive performance of healthy older adults. Archives of General Psychiatry.
- 2. Teichman, S. L., et al. (2006). Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I (IGF-I) secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. The Journal of Clinical Endocrinology & Metabolism.
- 3. Svensson, J., et al. (1999). The GH secretagogue ipamorelin: GH-releasing effect and selectivity in viable rat anterior pituitary cells. Endocrinology.
- 4. Clemmons, D. R., et al. (2012). Tesamorelin, a growth hormone-releasing factor analogue, in HIV-infected patients with abdominal fat accumulation: a randomized placebo-controlled trial. PLOS ONE.
- 5. Sigalos, J. T., & Pastuszak, A. W. (2018). The Safety and Efficacy of Growth Hormone Secretagogues. Sexual Medicine Reviews.
- 6. Alba, M., et al. (2006). Once-daily administration of CJC-1295, a long-acting growth hormone-releasing hormone (GHRH) analog, normalizes growth in the GHRH-deficient generalized growth-retarded (lit/lit) mouse. American Journal of Physiology-Endocrinology and Metabolism.
- 7. Raun, K., et al. (1998). Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology.
- 8. Sinha, D. K., et al. (2020). Ipamorelin/CJC-1295 Therapy: A Comprehensive Review. International Journal of Molecular Sciences.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.