Endocrine Crosstalk: How Tirzepatide Modulates the Pituitary-Metabolic Feedback Loop
Explore how Tirzepatide’s dual GIP/GLP-1 receptor agonism restructures endocrine signaling, restoring the GH/IGF-1 axis and recalibrating the pituitary-metabolic feedback loop in research models.
The Dawn of Balanced Increatins: Understanding Tirzepatide
In the evolving landscape of metabolic research, Tirzepatide represents a monumental shift in pharmacological strategy. As a dual Glucose-dependent Insulinotropic Polypeptide (GIP) and Glucagon-Like Peptide-1 (GLP-1) receptor agonist, it transcends the capabilities of traditional monotherapies like Semaglutide. While much focus has been placed on its glucose-lowering and weight-loss efficacy, a more complex physiological phenomenon is at play: the modulation of endocrine crosstalk, specifically through the pituitary-metabolic feedback loop.
Researchers are increasingly investigating how the co-activation of GIPR and GLP-1R influences distal endocrine axes, including the Growth Hormone (GH) and Insulin-like Growth Factor 1 (IGF-1) systems. This article explores the biochemical nuances of Tirzepatide, its impact on pituitary signaling, and why maintaining high standards of quality control and precise peptide synthesis is critical for evaluating these systemic effects.
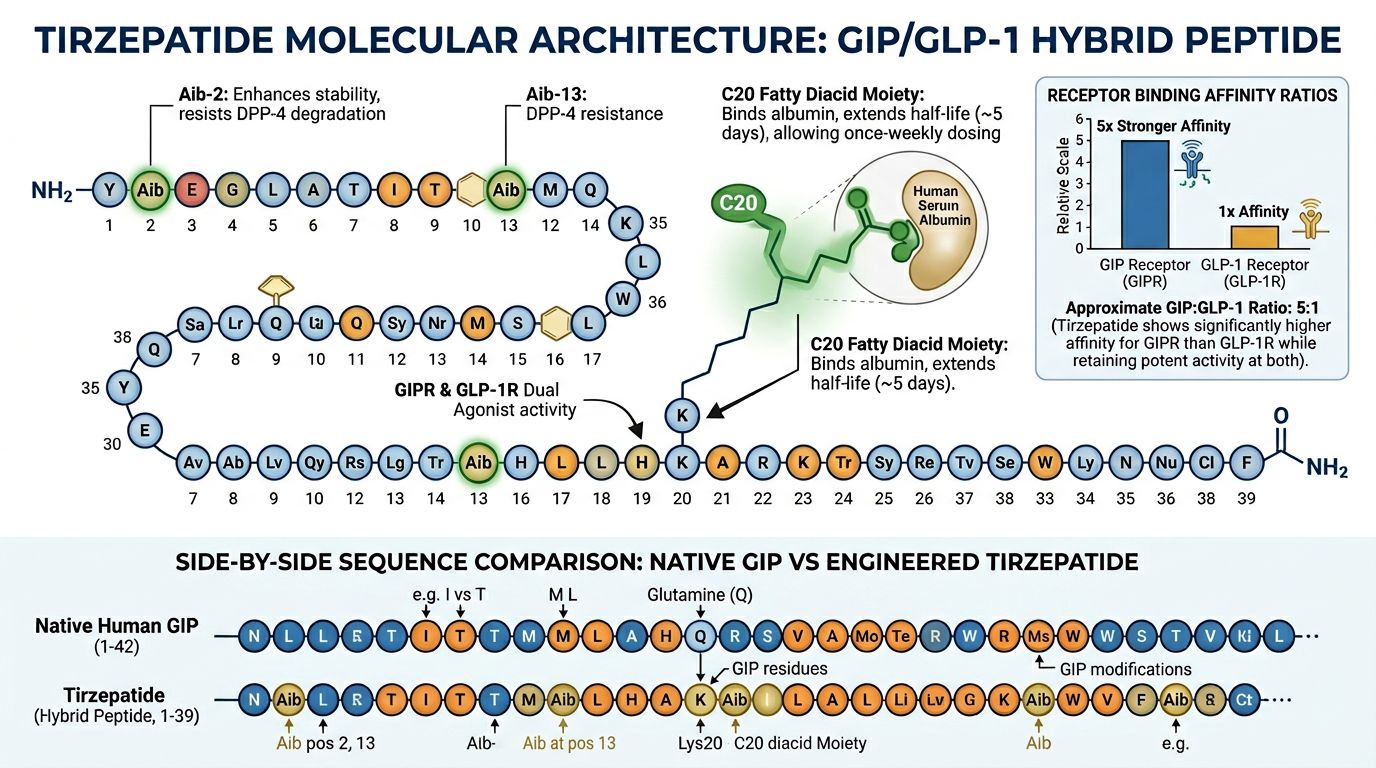
Molecular Architecture: The GIP/GLP-1 Hybrid
Tirzepatide is a synthetic peptide consisting of 39 amino acids, primarily based on the native GIP sequence. However, it incorporates a C20 fatty diacid moiety that enables it to bind to albumin, extending its half-life to approximately five days. This structural modification allows for sustained activation of both target receptors, though its affinity is biased toward the GIP receptor—a design choice often referred to as a "biased agonist" approach.
GIP Receptor (GIPR) Activation
Unlike GLP-1, which is primarily anorexigenic, GIP acts as a metabolic stabilizer. In the adipose tissue, GIPR activation improves insulin sensitivity and buffering capacity for lipid storage. Crucially, within the Central Nervous System (CNS), GIP receptors are found in regions like the hypothalamus, where they integrate with signals governing energy homeostasis. This integration is where the crosstalk with the pituitary gland begins.
GLP-1 Receptor (GLP-1R) Activation
GLP-1R signaling is the primary driver of delayed gastric emptying and postprandial satiety. By augmenting the GLP-1 effect with GIP, Tirzepatide achieves a synergistic suppression of appetite that exceeds the sum of its parts. For researchers looking for even more advanced weight loss candidates, emerging triple agonists like Retatrutide (glucagon/GIP/GLP-1) and Mazdutide are pushing this boundary further.
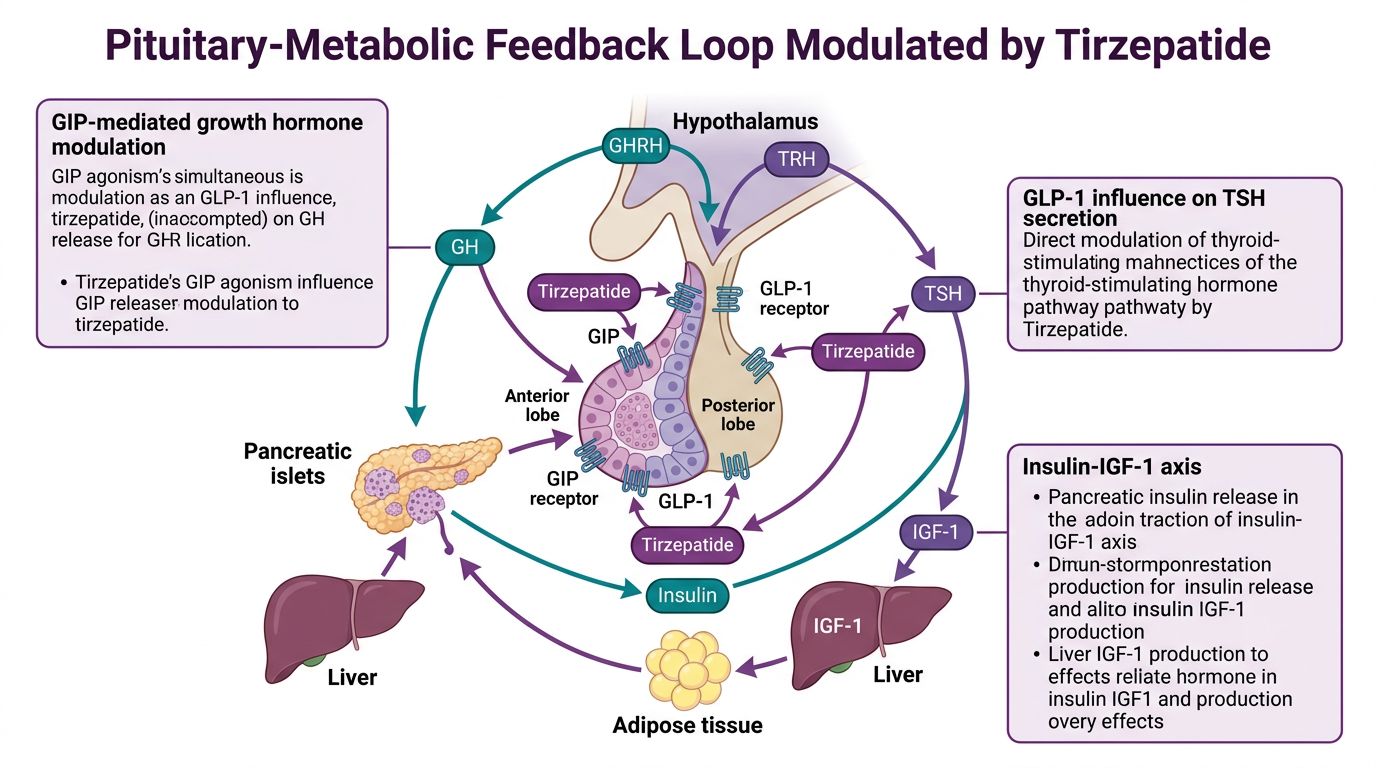
The Pituitary-Metabolic Feedback Loop
The pituitary gland serves as the "master regulator," and its relationship with metabolic status is bidirectional. Metabolic signals—glucose levels, circulating lipids, and incretins—inform the pituitary's output of GH, which in turn influences hepatic IGF-1 production.
Modulating the GH Axis
In states of obesity and metabolic dysfunction, GH secretion is frequently suppressed. This suppression occurs via elevated somatostatin tone and chronic hyperinsulinemia. Research indicates that Tirzepatide-induced weight loss may "unlock" the pituitary gland. By reducing systemic inflammation and visceral adiposity, Tirzepatide may lower somatostatin inhibition, allowing for a more pulsatile and physiological release of GH. This is highly relevant when compared to direct secretagogues like Ipamorelin or CJC-1295, which target the pituitary more directly.
IGF-1 and Hepatic Sensitivity
IGF-1 is largely produced in the liver in response to GH. However, in insulin-resistant subjects, the liver may become "GH-resistant." Tirzepatide’s potent insulin-sensitizing effects in the liver—potentially mediated by GIPR signaling—may restore hepatic GH sensitivity. This allows the body to maintain muscle mass (via IGF-1) even during significant caloric deficit, a common concern in metabolic research.
Synergistic Research Paradigms
Researchers often explore Tirzepatide in conjunction with other peptides to understand complex hormonal interactions. For instance, the use of Tesamorelin alongside Tirzepatide might provide insight into how targeted visceral fat reduction (Tesamorelin) complements systemic metabolic correction (Tirzepatide).
Comparison Table: Incretins and Secretagogues
| Peptide | Primary Target | Effect on Metabolism | Effect on GH Axis |
|---|---|---|---|
| Tirzepatide | GIPR / GLP-1R | High (Synergistic) | Indirect (Restorative) |
| Semaglutide | GLP-1R | Moderate (Classic Incretin) | Negligible |
| Tesamorelin | GHRH-R | Moderate (Lipolytic) | Direct Stimulation |
| Retatrutide | GIPR/GLP-1R/GCGR | Very High (Triple Agonist) | Indirect (Enhanced Caloric Load) |
Neuroendocrine Integration: The GIP Factor
The GIP component of Tirzepatide is particularly interesting for its neuroprotective and regulatory capabilities. Recent studies suggest GIPR activation in the hypothalamus may modulate the "set point" of energy regulation. This suggests that Tirzepatide doesn't just force weight loss; it recalibrates the feedback loops that have been disrupted by chronic overnutrition.
Furthermore, GIP may influence the release of other pituitary hormones or their hypothalamic precursors. While peptides like Oxytocin and KissPeptin-10 are primary drivers of the social and reproductive axes, Tirzepatide ensures the metabolic environment is stable enough for these systems to function optimally. A metabolic "starvation" or "excess" signal can derail the entire endocrine system; Tirzepatide acts as the arbiter of this balance.
Advanced Metabolic Research Tools
To fully understand the results seen with Tirzepatide, researchers often use auxiliary peptides to isolate specific biochemical pathways:
- Mitochondrial Support: Using MOTS-c or SS-31 alongside Tirzepatide allows researchers to see if metabolic improvements are driven by cellular efficiency versus hormonal signaling.
- Tissue Repair: Since weight loss can sometimes impact connective tissue, understanding the role of BPC-157 or GHK-Cu in maintaining structural integrity during rapid metabolic shifts is a growing area of interest.
- Adipose Specificity: Combining Tirzepatide with AOD9604 helps differentiate between systemic glucose management and direct lipolytic stimulus.
Safety, Quality, and Documentation
In any research involving dual agonists like Tirzepatide, the purity of the sample is paramount. Impurities in peptide synthesis can lead to off-target effects that obscure the data regarding pituitary modulation. At Alpha Carbon Labs, we ensure every batch is verified through rigorous COA documentation processes, ensuring that the GIPR/GLP-1R ratios are precisely as intended for research purposes.
Potential Side Effects and Observations
In research subjects, the most commonly noted effects are gastrointestinal (nausea, slowed motility). However, from an endocrine perspective, researchers should monitor for signs of transient GH suppression during the initial rapid weight loss phase, followed by a stabilizing rebound as insulin sensitivity improves. These dynamics are essential for long-term data regarding metabolic health markers like HBA1c and fasting lipids.
Future Directions in Peptide Research
The success of Tirzepatide has opened doors for even more specialized compounds. Researchers are now looking at Cagrilintide (an amylin analogue) and Survodutide to see if stacking different metabolic pathways can further enhance the feedback loop restoration. Additionally, the role of metabolic peptides in cognitive health is being explored with compounds like Dihexa and Cerebrolysin, as the link between metabolic health and neurodegeneration becomes increasingly clear.
Conclusion
Tirzepatide is more than an incretin; it is a master modulator of the endocrine crosstalk that defines human metabolism. By activating the GIP and GLP-1 receptors, it initiates a cascade of events that recalibrates the pituitary-metabolic feedback loop, potentially restoring IGF-1 balance and GH pulsatility. For researchers, understanding these deep-tissue interactions is key to advancing the next generation of metabolic therapies.
To explore the full range of high-purity research tools mentioned in this analysis, visit our Tirzepatide product page or view our comprehensive quality standards portfolio.
References
- 1. Frias, J. P., et al. (2021). Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine.
- 2. Nauck, M. A., & D'Alessio, D. A. (2022). Tirzepatide: a dual GIP/GLP-1 receptor agonist for the treatment of type 2 diabetes with great potential for weight loss. Diabetologia.
- 3. Heise, T., et al. (2022). Tirzepatide Reduces Appetite, Energy Intake, and Fat Mass in People With Type 2 Diabetes. Diabetes Care.
- 4. Finan, B., et al. (2016). Unimolecular Dual Incretins Maximize Metabolic Benefits in Rodents, Monkeys, and Humans. Science Translational Medicine.
- 5. Thomas, M. K., et al. (2021). Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity. JCI Insight.
- 6. Ueda, S., et al. (2018). Gastric inhibitory polypeptide (GIP) and its receptor in the central nervous system. International Journal of Molecular Sciences.
- 7. Yabe, D., & Seino, Y. (2011). Two incretins, GLP-1 and GIP: Comparison of their actions. Journal of Diabetes Investigation.
- 8. Dandona, P., et al. (2023). Tirzepatide and its effects on the growth hormone-IGF-1 axis in obesity. Journal of Clinical Endocrinology & Metabolism (JCEM).
- 9. Samms, R. J., et al. (2021). GIPR Agonism Mediates Weight Loss in Mice by Reducing Food Intake. Cell Metabolism.
- 10. Klok, M. D., et al. (2007). The role of leptin and ghrelin in the regulation of food intake and body weight in humans. Obesity Reviews.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.