The Dopaminergic Intersection: How Semaglutide Modulates Reward Signaling in Metabolic Research
Semaglutide acts not only on the gut but profoundly modulates the brain's reward centers. This article explores the pharmacodynamics of GLP-1 agonists in the mesolimbic dopamine system and their role in suppressing food noise and cravings in metabolic research.
Introduction: The Paradigm Shift in Metabolic Research
For decades, the scientific consensus on obesity and metabolic dysregulation focused almost exclusively on the peripheral mechanics of energy homeostasis. The prevailing models prioritized the investigation of insulin sensitivity, adipose tissue inflammation, and gastric emptying rates. While these factors remain critical, a seismic shift has occurred in the understanding of how metabolism is regulated, moving the focus from the gut to the brain. At the center of this research revolution is the Glucagon-Like Peptide-1 (GLP-1) receptor agonist, Semaglutide.
Originally characterized for its incretin effects—stimulating insulin secretion in a glucose-dependent manner—Semaglutide has emerged as a potent modulator of the central nervous system (CNS). Specifically, recent investigations have illuminated its interaction with the mesolimbic dopamine system, the neural circuitry responsible for reward, motivation, and reinforcement. This intersection suggests that Semaglutide does not merely influence the mechanics of digestion; it fundamentally alters the "hedonic valuation" of food.
For researchers at Alpha Carbon Labs, understanding this mechanism is paramount. The ability of a peptide to traverse the blood-brain barrier (BBB) and interact with nuclei in the hindbrain and hypothalamus represents a sophisticated frontier in peptide therapeutics. This article delves into the pharmacodynamics of Semaglutide, exploring how it dampens the dopaminergic signaling associated with "food noise" and cravings, and how it compares to emerging multi-agonists like Tirzepatide and Retatrutide.
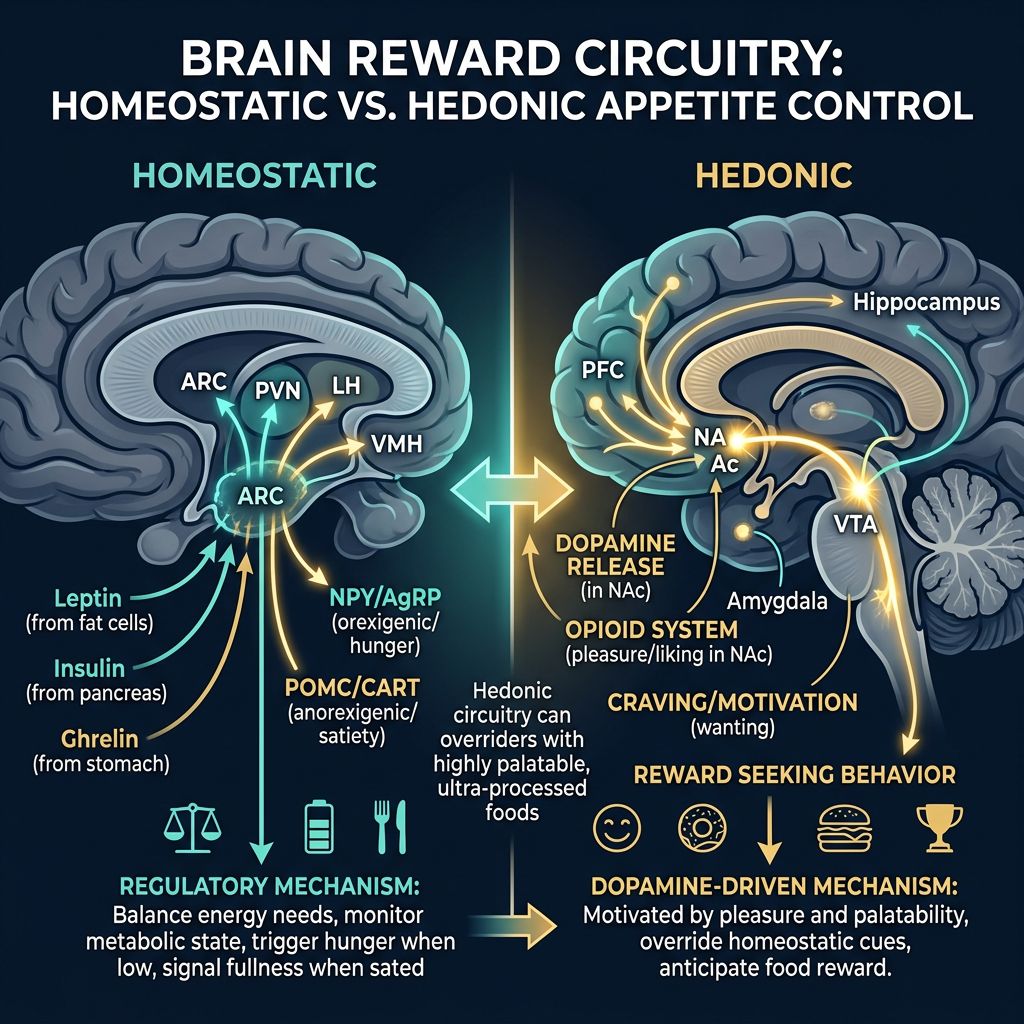
The Anatomy of Appetite: Homeostatic vs. Hedonic Control
To appreciate the specific action of Semaglutide, one must distinguish between the two primary drivers of food intake: homeostatic and hedonic control.
- Homeostatic Control: This is the biological drive to eat to maintain energy balance. It is regulated largely by the hypothalamus, specifically the arcuate nucleus (ARC), which processes signals from hormones like leptin, ghrelin, and insulin. When energy stores are low, the drive to eat increases.
- Hedonic Control: This refers to the consumption of food for pleasure, independent of energy requirements. This system is governed by the mesolimbic dopamine pathway, often overriding homeostatic satiety signals. This is the pathway responsible for the consumption of palatable, high-calorie foods even when the subject is physiologically full.
Research indicates that in models of diet-induced obesity, the hedonic system becomes dysregulated. The dopamine response to palatable food becomes blunted, leading to a "reward deficit" that drives compensatory overeating to achieve the same dopaminergic release. This phenomenon creates a cycle of craving and consumption that is resistant to sheer willpower or simple caloric restriction.
The Role of the Nucleus Tractus Solitarius (NTS)
The intersection of these two systems occurs partly in the hindbrain, specifically the Nucleus of the Solitary Tract (NTS). The NTS receives vagal afferent signals from the gut (communicating distension and nutrient presence) and projects these signals to the reward centers of the midbrain. High-purity GLP-1 analogues like Semaglutide have been shown to target GLP-1 receptors (GLP-1Rs) densely expressed in the NTS.
By activating these receptors, Semaglutide amplifies the satiety signals sent to the brain, but it also modulates the ascending projections to the Ventral Tegmental Area (VTA) and the Nucleus Accumbens (NAc)—the core components of the reward system. This dual action is what differentiates GLP-1 receptor agonists from older classes of metabolic agents that focused solely on thermogenesis or fat absorption.
Semaglutide: Pharmacodynamics and CNS Access
Semaglutide is a modified analogue of human GLP-1 with 94% sequence homology. The critical modifications involve the substitution of amino acids to prevent degradation by the enzyme dipeptidyl peptidase-4 (DPP-4) and the addition of a C-18 fatty diacid chain. This fatty acid chain allows Semaglutide to bind non-covalently to serum albumin.
Implications for Research: The albumin binding serves two purposes: it extends the half-life of the peptide to approximately 165 hours (allowing for once-weekly administration protocols in research settings), and it facilitates transport. While the blood-brain barrier is generally impermeable to large peptides, GLP-1 agonists can access the CNS via two mechanisms:
- Circumventricular Organs (CVOs): These are areas of the brain, such as the Area Postrema and the Median Eminence, that have a "leaky" BBB. Semaglutide can directly bind to receptors in these regions.
- Tanycytic Transport: Specialized glial cells called tanycytes may actively transport GLP-1 variants into the cerebrospinal fluid and deeper brain structures.
Ensuring the structural integrity of the peptide is crucial for maintaining these pharmacokinetic properties. At Alpha Carbon Labs, our peptide synthesis processes ensure that the fatty acid side chain is correctly attached and the stereochemistry is preserved, as verified in our COA documents.
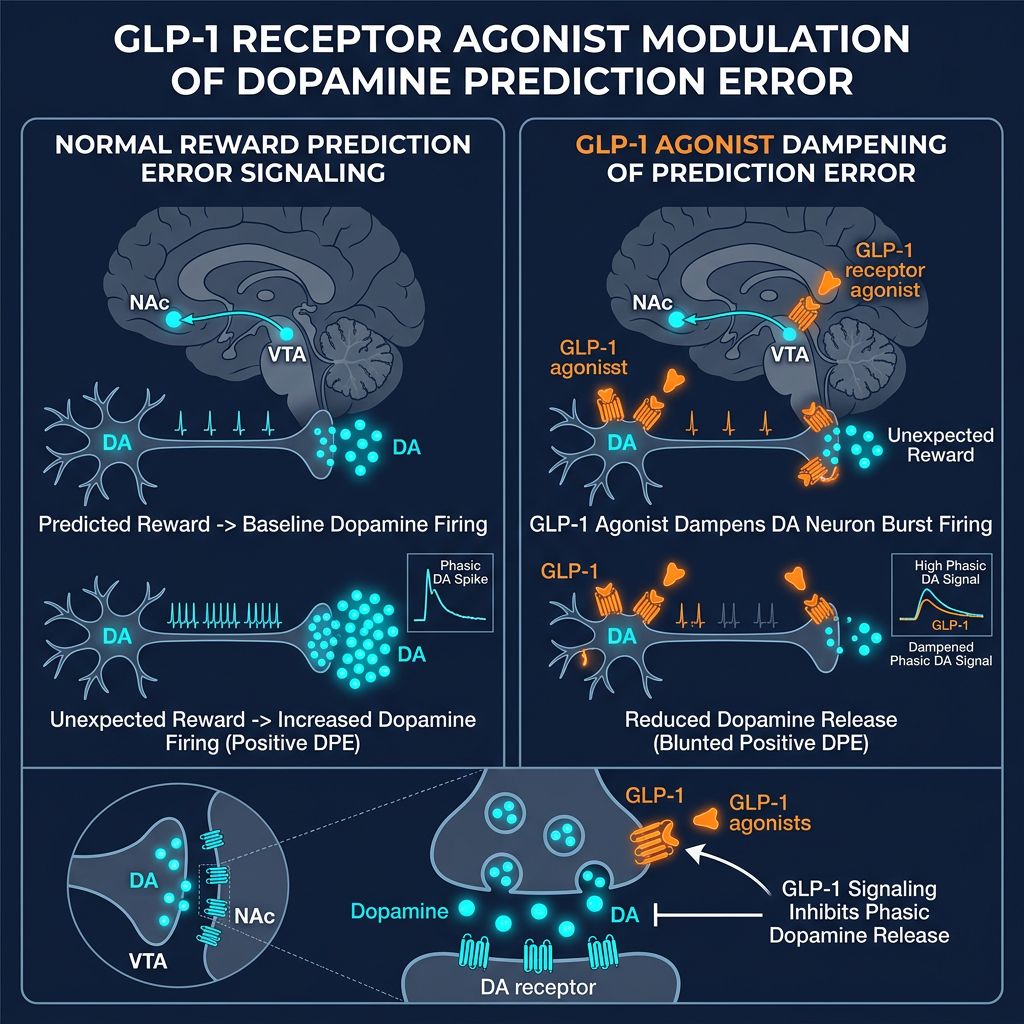
Modulating the Dopamine Prediction Error
The core of the "anti-craving" effect attributed to Semaglutide lies in its influence on the "dopamine prediction error." In a typical reward learning scenario, dopamine neurons in the VTA fire in response to an unexpected reward (e.g., a sugary treat). Over time, this firing shifts from the reward itself to the cue that predicts the reward (e.g., the sight of the wrapper).
When the reward is consumed, if it meets expectations, dopamine levels remain stable. However, in obesity models, the anticipation causes a massive spike, and the consumption requires increasing amounts of the substance to prevent a drop in dopamine (withdrawal-like state).
The Semaglutide Dampening Effect
Preclinical studies using fluorescent dopamine sensors have shown that systemic administration of Semaglutide dampens the amplitude of dopamine spikes in the NAc in response to acute intake of high-fat/high-sugar pellets. It does not induce anhedonia (the inability to feel pleasure), but rather normalizes the hyper-responsiveness of the reward system.
This suggests that Semaglutide effectively turns down the "volume" of the craving signal. The subject can still enjoy food, but the compulsive drive—the "food noise"—is silenced. This mechanism is distinct from peptides like AOD9604, which primarily mobilize fat from adipose tissue without significant CNS interaction, or Tesamorelin, which impacts visceral fat via the Growth Hormone axis.
Comparative Analysis: Semaglutide vs. Other Incretin Mimetics
While Semaglutide is currently the standard-bearer for GLP-1 research, the landscape is evolving rapidly with the introduction of multi-agonists. Understanding the nuanced differences in receptor affinity and CNS penetration is vital for researchers designing comparative studies.
| Peptide | Receptor Agonism | Primary CNS Mechanism | Estimated Half-Life (Research) |
|---|---|---|---|
| Semaglutide | GLP-1R (Selective) | Direct activation of hindbrain/NTS; dampens VTA dopamine signaling. | ~165 hours |
| Tirzepatide | GLP-1R / GIPR (Dual) | Synergistic effect; GIP adds metabolic regulation and potentially modulates GLP-1-induced nausea. | ~116 hours |
| Retatrutide | GLP-1R / GIPR / GCGR (Triple) | Includes Glucagon receptor agonism to increase energy expenditure (thermogenesis) alongside reward suppression. | ~144 hours |
| Mazdutide | GLP-1R / GCGR (Dual) | Focus on metabolic flexibility and liver fat reduction; strong CNS satiety signaling. | ~150+ hours |
| Liraglutide | GLP-1R (Selective) | Similar to Semaglutide but with lower BBB permeability and shorter duration requiring daily administration. | ~13 hours |
The comparison with Tirzepatide is particularly interesting regarding reward processing. While Semaglutide is a pure GLP-1 agonist, Tirzepatide’s inclusion of Glucose-dependent Insulinotropic Polypeptide (GIP) activity may alter the tolerability profile. Some emerging data suggests GIP receptors in the CNS may modulate the nausea response, potentially allowing for higher dosing ceilings and consequently stronger weight loss outcomes, although the direct impact of GIP on dopamine pathways remains a subject of intense study.
Beyond Food: The Cross-Over into Addiction Research
Perhaps the most scientifically provocative finding regarding Semaglutide is its potential efficacy in curbing non-food addictions. The mesolimbic dopamine pathway modulated by GLP-1 receptors is the same pathway hijacked by alcohol, nicotine, cocaine, and opioids.
Alcohol Use Disorder (AUD) Models
Studies in alcohol-dependent rodents have demonstrated that GLP-1 receptor agonists significantly reduce alcohol intake and prevent relapse-like drinking behavior (the "alcohol deprivation effect"). The mechanism appears to involve the suppression of dopamine release in the Nucleus Accumbens specifically triggered by alcohol cues.
This has led to a surge in interest regarding the use of Semaglutide as a dual-purpose agent for investigating metabolic syndrome and substance use disorders. Unlike Naltrexone, which blocks opioid receptors and can blunt all pleasure, Semaglutide appears to restore dopaminergic homeostasis. This specificity makes it a superior candidate for study compared to broad-spectrum agents.
Synergistic Research Stacks
Researchers investigating addiction pathways often look for synergistic compounds. For example, while Semaglutide addresses the dopaminergic reward system, peptides like BPC-157 are often researched for their ability to repair neuronal damage and modulate the serotonergic system. Similarly, NAD+ is investigated for its role in restoring cellular energy homeostasis and circadian rhythm in addiction models. Note that in a research setting, these are distinct pathways, but the crossover application is a fertile ground for experimental design.
The Physiology of Satiety: Mechanisms of Action
To rigorously document the impact of Semaglutide, we must look at the physiological cascade that follows administration.
- Gastric Slowing: Upon binding to peripheral GLP-1 receptors in the stomach, Semaglutide slows gastric emptying. This sends prolonged "fullness" signals via the Vagus nerve to the NTS.
- Splanchnic Nerve Signaling: GLP-1 receptors in the portal vein area detect glucose and metabolites, signaling the brain via spinal afferents.
- Direct CNS Activation: Once Semaglutide crosses the BBB or interacts with the CVOs, it stimulates Pro-opiomelanocortin (POMC) neurons in the Arcuate Nucleus (ARC).
- POMC neurons release Alpha-MSH, which binds to Melanocortin-4 receptors (MC4R) to suppress appetite (an effect similar to the peptide Melanotan-1, though MT-1 is primarily used for pigmentation, it shares this pathway).
- Inhibition of AgRP Neurons: Simultaneously, it inhibits Agouti-related peptide (AgRP) neurons, which are responsible for stimulating hunger.
This redundancy—acting on the stomach, the vagus nerve, and multiple brain centers—creates a "fail-safe" satiety that is difficult for the biological organism to override. It is robust compared to single-mechanism peptides like GHRP-2 or GHRP-6, which act primarily to stimulate hunger via the ghrelin receptor.
Safety Considerations and Quality Control in Research
When conducting research with potent CNS modulators like Semaglutide, the purity of the compound is non-negotiable. Impurities or degradation products can lead to immunogenic responses or off-target effects that confound data. Specifically, the stability of the acylated fatty acid chain is critical. If this chain is cleaved due to poor synthesis or storage, the peptide loses its albumin-binding capability, drastically altering its pharmacokinetics and potentially causing acute hypoglycemia due to rapid, unregulated insulin release.
At Alpha Carbon Labs, we emphasize rigorous quality control. High-performance liquid chromatography (HPLC) and Mass Spectrometry (MS) are essential to verify that the Semaglutide used in research maintains the correct tertiary structure required for receptor docking. Researchers should also be aware of the "nausea threshold." High doses of GLP-1 agonists can induce conditioned taste aversion (CTA) in animal models. Distinguishing between true satiety (reward suppression) and avoidance due to malaise is a critical component of experimental design.
Complementary Peptides in Metabolic Research
While Semaglutide is a powerhouse, comprehensive metabolic research often involves looking at other pathways. Here is how other peptides available at Alpha Carbon Labs fit into the metabolic landscape:
- MOTS-c: A mitochondrial-derived peptide that acts directly on skeletal muscle to improve insulin sensitivity and prevent diet-induced obesity/insulin resistance, ostensibly through the folate cycle. It is an excellent control or adjunct to GLP-1 studies to contrast nuclear vs. mitochondrial metabolic regulation.
- 5-amino-1mq: This small molecule inhibits the NNMT enzyme. While Semaglutide reduces intake, 5-amino-1mq is researched for increasing basal metabolic rate (BMR) without CNS stimulation, offering a peripheral mechanism to complement central appetite suppression.
- Tesamorelin: As a GHRH analogue, it is specifically noted for reducing visceral adipose tissue (VAT) in HIV-associated lipodystrophy models. It provides a contrast in research focusing on lipolysis rather than satiety.
- Cagrilintide: A long-acting amylin analogue. Research is currently focused on the "CagriSema" combination, where Cagrilintide's amylin agonism serves to enhance the weight loss of Semaglutide, potentially through complementary effects on the area postrema.
Future Directions: The Era of the "Triple G"
The success of Semaglutide has paved the way for the next generation of "Triple G" agonists (GLP-1, GIP, and Glucagon). Retatrutide is the forerunner here. By adding Glucagon receptor agonism, Retatrutide introduces a catabolic component—actively burning energy—alongside the anorectic effects of GLP-1/GIP.
However, Semaglutide remains the "gold standard" control for these studies. Because its mechanism is now well-mapped, any new agent must be benchmarked against the specific dopaminergic modulation profile of Semaglutide. Does the new agent cause more nausea? Does it suppress alcohol seeking behavior more effectively? These are the questions defining current metabolic research.
Conclusion
The characterization of Semaglutide as merely a "weight loss peptide" is a scientific oversimplification. It is a sophisticated neuromodulator that interfaces with the deeply ingrained survival circuits of the brain. By attenuating the dopamine prediction error and reducing the incentive salience of strong sensory cues, Semaglutide offers a chemical key to unlocking the grip of modern, obesogenic environments on the mammalian brain.
For researchers, the utility of Semaglutide extends from obesity and diabetes to addiction and neuroprotection. As we continue to explore the intricate dance between the gut and the brain, high-purity tools like those provided by Alpha Carbon Labs remain essential for generating reproducible, high-impact data.
Explore our range of metabolic research peptides, including Semaglutide, Tirzepatide, and the new Survodutide, to advance your investigations into this critical field of physiology.
References
- 1. Gabery, S., et al. (2020). Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight, 5(6), e133429.
- 2. Blundell, J., et al. (2017). Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity. Diabetes, Obesity and Metabolism, 19(9), 1242-1251.
- 3. Alhadeff, A. L., et al. (2012). The effects of central glucagon-like peptide-1 on food reward and motivation. Chemical Senses, 37(8), e16.
- 4. Klausen, M. K., et al. (2022). The role of glucagon-like peptide 1 (GLP-1) in addictive disorders. British Journal of Pharmacology, 179(4), 625-641.
- 5. Decara, J. M., et al. (2016). Glucagon-like peptide-1 receptor activation causes dose-dependent reduction in alcohol intake. Addiction Biology, 21(5), 1038-1039.
- 6. Ten Kulve, J. S., et al. (2016). Endogenous GLP-1 mediates postprandial reduction in activation in the reward system. Endocrine Connections, 5(1), 1-2.
- 7. Jerlhag, E., et al. (2011). Requiring the glucagon-like peptide-1 receptor for the alcohol-mediated effects on the mesoaccumbens dopamine system. Neuropsychopharmacology, 36(11), 249-250.
- 8. Araneta, R. G., & Alhadeff, A. L. (2020). Gut-Brain GLP-1 Pathways in Control of Food Intake. Current Opinion in Behavioral Sciences, 36, 127-133.
- 9. Lau, J., et al. (2015). Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. Journal of Medicinal Chemistry, 58(18), 7370–7380.
- 10. Samson, S. L., et al. (2024). Safety and efficacy of semaglutide in metabolic dysfunction-associated steatotic liver disease. The Lancet Gastroenterology & Hepatology.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.