Secondary Metabolic Cascades: Regulatory Considerations in Long-Term Retatrutide Research
Explore the complex secondary metabolic cascades of Retatrutide, including its long-term effects on the HPA axis and gallbladder motility in triple-agonist research designs.
Introduction: The Evolution of Metabolic Poly-Agonism
The landscape of metabolic research has shifted dramatically from single-pathway interventions to the complex orchestration of multiple signaling cascades. While Semaglutide established the efficacy of GLP-1 receptor agonism, and Tirzepatide demonstrated the synergistic power of dual GLP-1/GIP activation, the emergence of Retatrutide represents a paradigm shift. Retatrutide is a "triple agonist" targeting the glucagon receptor (GCGR), the glucose-dependent insulinotropic polypeptide receptor (GIPR), and the glucagon-like peptide-1 receptor (GLP-1R).
This triple agonism achieves weight loss and metabolic correction through a multi-pronged approach: slowing gastric emptying, enhancing insulin secretion, and crucially, increasing energy expenditure via glucagon signaling. However, as research moves into long-term longitudinal studies, it is imperative to examine the secondary metabolic cascades—downstream effects that extend beyond simple weight loss and glycemia into the Hypothalamic-Pituitary-Adrenal (HPA) axis and gallbladder physiology. For researchers utilizing high-purity peptides, understanding these nuanced interactions is paramount to experimental integrity.
Molecular Architecture and Triple Agonist Synergy
Retatrutide is a 39-amino-acid peptide based on the GIP sequence, modified with a C-18 fatty diacid moiety to extend its half-life and enable weekly administration. Its affinity for the three receptors is carefully balanced to maximize metabolic output while minimizing side effects. Unlike its predecessors, the inclusion of glucagon receptor agonism introduces a potent thermogenic component.
Receptor Profiles and Potency
The efficacy of Retatrutide is predicated on its specific EC50 values for the three target receptors. In comparative studies, it shows approximately:
- Strong GIP receptor agonism (8-fold potency of native GIP)
- Balanced GLP-1 receptor agonism (0.3-fold potency of native GLP-1)
- Significant Glucagon receptor agonism (0.4-fold potency of native glucagon)
This balance prevents the hyperglycemia typically associated with glucagon while capturing its benefits for hepatic lipid oxidation and metabolic rate. For researchers seeking to compare these effects to dual-agonist profiles, exploring the Tirzepatide data provides a necessary baseline for GIP/GLP-1 synergy without the GCGR component.
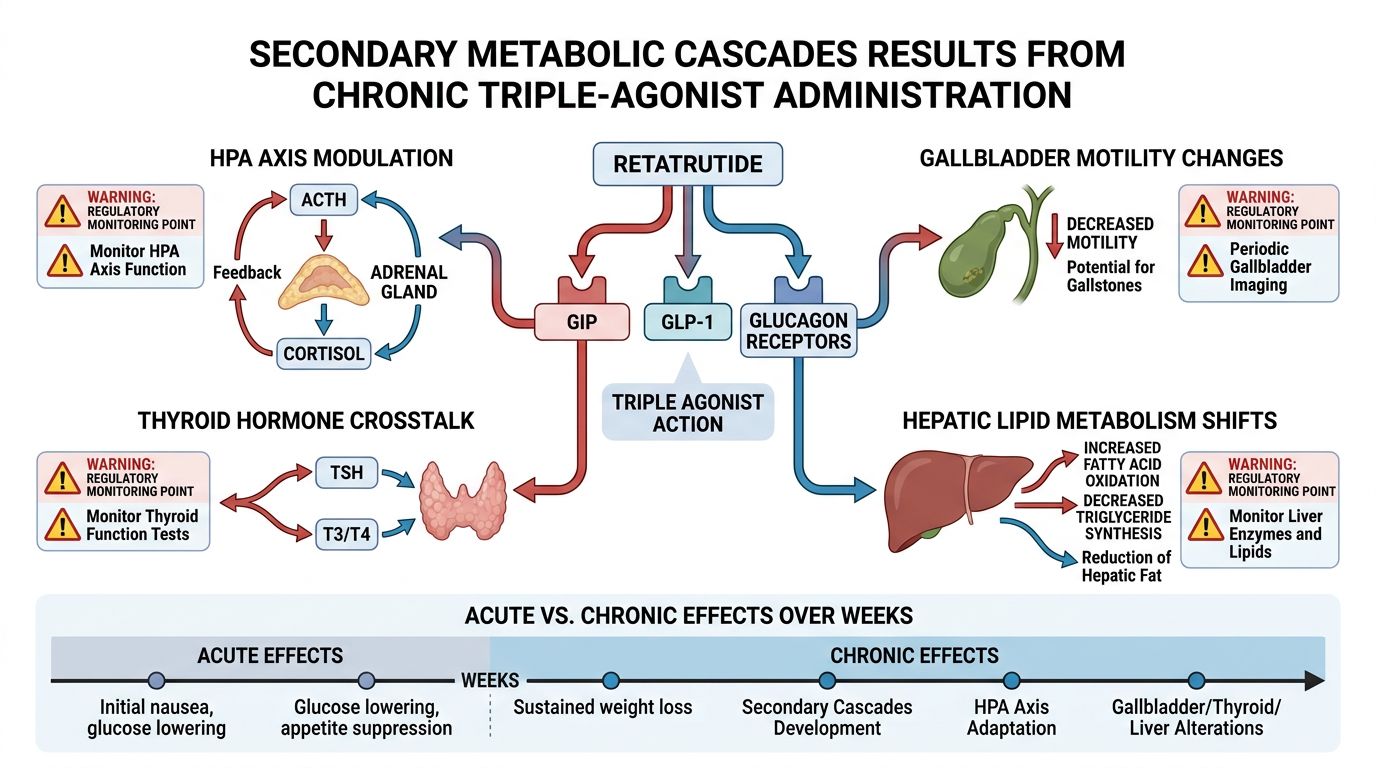
Secondary Cascades: The HPA Axis and Chronic Administration
The HPA axis is the primary neuroendocrine system responsible for the "fight or flight" response and chronic stress adaptation. Long-term administration of potent metabolic agonists can exert subtle but significant pressure on this axis via cross-talk in the paraventricular nucleus (PVN) of the hypothalamus.
Cortisol Dynamics and Metabolic Flux
GLP-1 and glucagon receptors are expressed in the hypothalamus. Acute administration of GLP-1R agonists has been shown to transiently increase ACTH and cortisol levels in rodent models. In the context of Retatrutide, the addition of the glucagon component may amplify this effect. Glucagon plays a role in glucose mobilization during stress; therefore, triple agonism induces a "pseudo-fasting" state that the HPA axis may interpret as a nutritional stressor.
Glucocorticoid Resistance and Insulin Sensitivity
Chronic elevation of metabolic throughput can sometimes lead to compensatory shifts in glucocorticoid sensitivity. Researchers must monitor whether long-term Retatrutide exposure modulates 11β-HSD1 activity, the enzyme responsible for regenerating active cortisol. This is a critical regulatory consideration because high local cortisol levels in adipose tissue can counteract the weight-loss benefits of the peptide, even in the presence of AOD9604 or other lipolytic agents.
Gallbladder Motility and Biliary Kinetics
One of the most discussed regulatory concerns in long-term incretin-mimetics research is gallbladder health. All GLP-1R agonists are associated with a dose-dependent decrease in gallbladder motility, primarily due to the inhibition of cholecystokinin (CCK) secretion and direct neurohormonal effects on the gallbladder wall.
The Triple Agonist Impact on Bile Homeostasis
The inclusion of glucagon agonism in Retatrutide complicates the biliary profile. While GLP-1 inhibits gallbladder emptying, glucagon is known to relax the Sphincter of Oddi and increase bile flow (choleretic effect). In the short term, this may partially counteract the stasis caused by GLP-1. However, the rapid weight loss induced by triple agonism increases the concentration of cholesterol in bile, leading to a "lithogenic" (stone-forming) environment.
| Peptide Component | Effect on Gallbladder | Biomarkers to Monitor |
|---|---|---|
| GLP-1 Agonism | Reduced contractility / Stasis | CCK levels, Gallbladder Volume |
| GIP Agonism | Neutral to slightly inhibitory | Post-prandial GIP levels |
| Glucagon Agonism | Sphincter relaxation / Bile flow | Bile acid composition |
Researchers investigating long-term effects should consider co-administration protocols or monitoring biliary sludge through ultrasonography in animal models. The use of hepatoprotective peptides or antioxidants such as Glutathione may be warranted in specific experimental designs looking to isolate metabolic effects from secondary biliary stress.
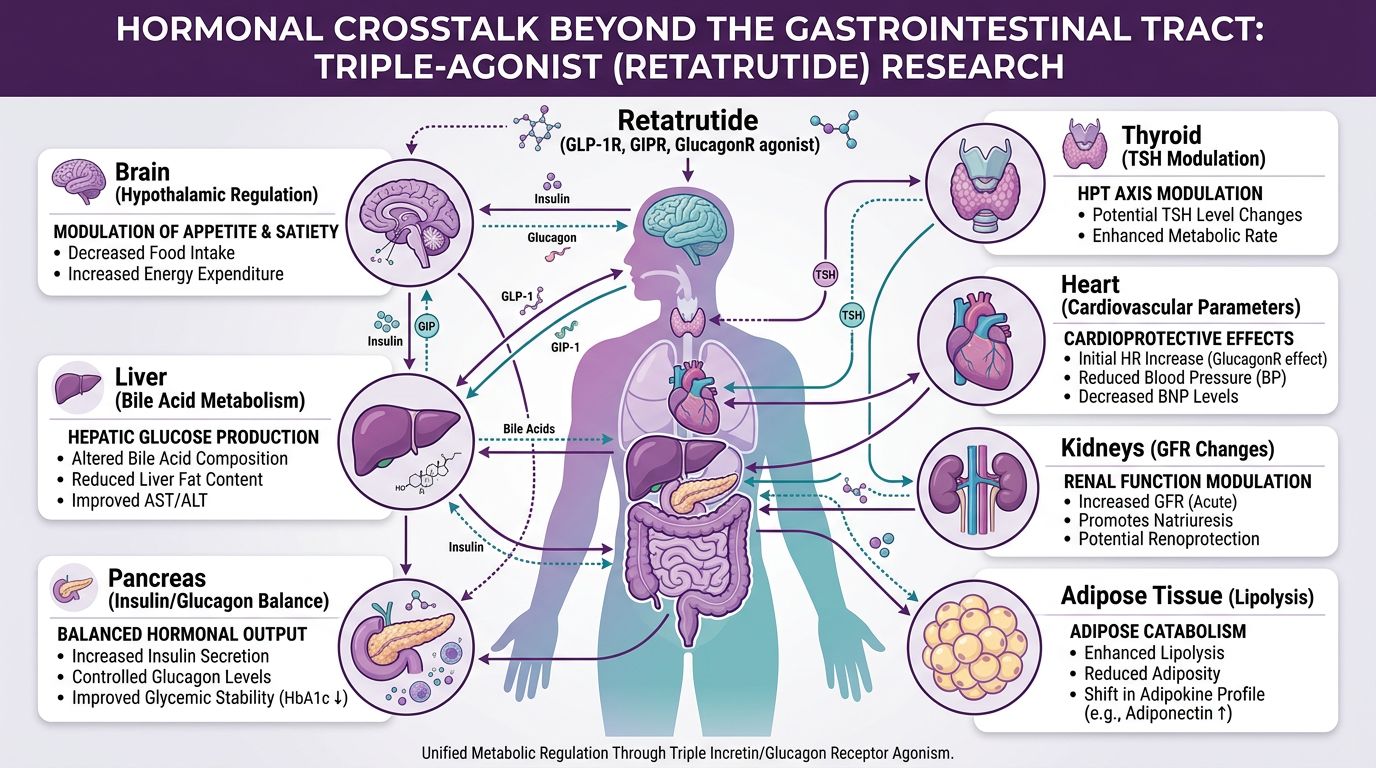
Hormonal Crosstalk: Beyond the Gastrointestinal Tract
The metabolic cascades of Retatrutide extend into the thyroidal and gonadal axes. Rapid shifts in adiposity and the introduction of glucagon-mediated thermogenesis can influence the basal metabolic rate (BMR) and the secretion of thyroid-stimulating hormone (TSH).
Thyroid Axis Modulation
In animal models, glucagon agonism has been linked to increased conversion of T4 to T3 in peripheral tissues, which accounts for part of its thermogenic effect. This mimics the action of certain growth hormone secretagogues like Sermorelin or Ipamorelin, but via a distinct, non-GHRH mediated pathway. Monitoring the T3/rT3 ratio is essential for understanding the efficiency of energy expenditure during Retatrutide research.
The Gonadal Axis and Leptin Feedback
As adipose tissue is rapidly depleted, leptin levels fall. This drop in leptin signals to the hypothalamus that energy stores are low, which can lead to a compensatory downregulation of the Gonadotropin-Releasing Hormone (GnRH) pulse generator. For studies focusing on reproductive health or hormonal restoration, researchers often pair metabolic agents with KissPeptin-10 or Gonadorelin to maintain axis integrity while pursuing metabolic correction.
Practical Application: Optimization of Research Protocols
To navigate these secondary cascades, research protocols must prioritize quality and rigorous monitoring. At Alpha Carbon Labs, we emphasize the importance of using peptides with verified purity to ensure that observed metabolic cascades are a result of the peptide itself and not contaminants. Researchers can verify our standards through our Quality Control processes and COA Documents.
Synergistic Combinations in Research
In some research contexts, Retatrutide is being studied alongside other peptides to mitigate secondary effects or enhance specific outcomes:
- For Muscle Preservation: Rapid weight loss can lead to sarcopenia. Pairing with IGF-1LR3 or Tesamorelin can help maintain lean mass.
- For Mitochondrial Health: To support the increased metabolic demand of triple agonism, MOTS-c or SS-31 may be utilized to optimize mitochondrial respiration.
- For Anti-Inflammatory Support: The inclusion of KPV (Lysine-Proline-Valine) can assist in managing the systemic low-grade inflammation often associated with metabolic flux.
The Future of Triple Agonism Research
The focus on Retatrutide isn't just about weight loss; it's about the total metabolic "reboot." Recent data suggests that the triple agonist approach may be particularly effective in treating Non-Alcoholic Fatty Liver Disease (NAFLD) and its progression to NASH. The glucagon component is crucial here, as it directly stimulates hepatic fatty acid oxidation, a feature lacking in Semaglutide monotherapy.
However, the regulatory considerations—meaning the body's internal feedback mechanisms—must be the center of future study designs. Understanding how the HPA axis adapts to long-term glucagon signaling will determine whether triple agonists can be used as sustained interventions or if they require "cycling" protocols similar to those used with HGH Fragment 176-191.
Summary of Regulatory Considerations
- Dosing Escalation: Slow titration is essential to allow the HPA axis and gallbladder to adapt to decreased gastric motility and increased metabolic rate.
- Intermittent Monitoring: Periodic assessment of cortisol, TSH, and biliary markers provides a comprehensive view of the secondary cascades.
- Peptide Integrity: Using peptides synthesized via high-standard peptide synthesis ensures that researchers are seeing the true pharmacological profile of the triple agonist.
Conclusion
Retatrutide represents the pinnacle of current incretin-based research, offering unprecedented metabolic control through its triple-agonist mechanism. Yet, the complexity of its interactions with the HPA axis and the biliary system requires a sophisticated approach to research. By acknowledging these secondary metabolic cascades, researchers can design more effective studies that not only track weight loss and glycemia but also understand the systemic regulatory adaptations to chronic administration.
Selection of high-quality research materials like Retatrutide and complementary peptides from a trusted supplier like Alpha Carbon Labs ensures that your findings are reproducible and scientifically sound. As the field progresses, the integration of triple agonism with mitochondrial peptides like NAD+ and tissue-repair agents like BPC-157 will likely define the next generation of metabolic health research.
References
- 1. Coskun, T., et al. (2022). LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: From discovery to clinical proof of concept. Cell Metabolism, 34(9), 1234-1247.
- 2. Jastreboff, A. M., et al. (2023). Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine, 389(6), 514-526.
- 3. Müller, T. D., et al. (2022). The development of next-generation poly-agonists for the treatment of metabolic disease. Nature Reviews Drug Discovery, 21, 201–223.
- 4. Heppner, K. M., et al. (2015). Glucagon regulation of energy expenditure and body weight. Molecular Metabolism, 4(2), 64-67.
- 5. Finan, B., et al. (2016). Multireceptor Agonists for the Treatment of Metabolic Disease. Endocrine Reviews, 37(6), 637-651.
- 6. Evers, A., et al. (2023). From dual to triple-agonists: The next generation of metabolic drugs. Journal of Peptide Science, 29(2), e3451.
- 7. Alhushki, W., et al. (2024). Triple agonists and gallbladder health: Implications for long-term administration. Gastroenterology Research and Practice.
- 8. Nauck, M. A., & Quast, D. R. (2023). Triple Agonism: The Future of Incretin-Based Therapy. Diabetologia, 66, 1789–1803.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.