This product is for research purposes only. Not for human consumption.
Purity: >98% (HPLC verified)
Formulation: Lyophilized powder
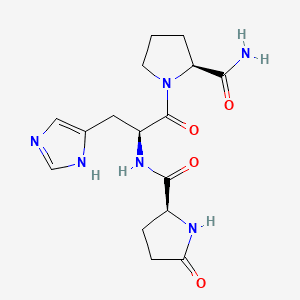
Molecular Formula: C55H75N17O13
Molecular Weight: 1182.3 g/mol
CAS Number: 33515-09-2
PubChem CID: 638678
Research Contents
Gonadorelin
Overview
Gonadorelin is a synthetic form of gonadotropin-releasing hormone (GnRH), also known as luteinizing hormone-releasing hormone (LHRH). It is a decapeptide (10 amino acids) with the sequence pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2 that is identical to the naturally occurring GnRH produced by the hypothalamus. GnRH is the master regulator of the reproductive system, controlling the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the pituitary gland.
Physiological Role
In the body, GnRH is released in a pulsatile manner from specialized hypothalamic neurons into the hypophyseal portal blood system, where it travels to the anterior pituitary to stimulate gonadotroph cells. This pulsatile secretion pattern is critical - the frequency and amplitude of GnRH pulses determine whether LH or FSH release predominates, allowing precise regulation of reproductive function throughout the menstrual cycle and across different physiological states.
Pharmaceutical Applications
As a pharmaceutical product, gonadorelin has been used diagnostically to assess pituitary function and therapeutically in specific reproductive disorders. Its pulsatile administration can stimulate gonadotropin secretion, while continuous administration leads to receptor downregulation and suppression of the reproductive axis - a property exploited therapeutically.
Clinical Significance
Gonadorelin represents one of the earliest peptide hormones to be successfully synthesized and developed for clinical use. Its discovery and characterization earned Andrew Schally and Roger Guillemin the 1977 Nobel Prize in Physiology or Medicine. The hormone remains an essential tool in reproductive endocrinology research and clinical diagnostics.
Mechanism of Action
Gonadorelin binds to GnRH receptors (GnRHR) on gonadotroph cells in the anterior pituitary gland. These receptors are G-protein coupled receptors that activate multiple intracellular signaling pathways to regulate gonadotropin synthesis and secretion.
Receptor Signaling Cascade
Upon GnRH binding, the receptor primarily couples to Gq/11 proteins, activating phospholipase C (PLC) which hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers calcium release from the endoplasmic reticulum, while DAG activates protein kinase C (PKC). The resulting calcium influx and PKC activation drive the exocytosis of LH and FSH granules and stimulate gene transcription for new gonadotropin synthesis.
Pattern-Dependent Response
The response to gonadorelin is highly dependent on the pattern of administration. Pulsatile delivery (mimicking natural GnRH secretion at intervals of 60-120 minutes) maintains and stimulates gonadotropin release by allowing receptor resensitization between pulses. In contrast, continuous exposure leads to GnRH receptor downregulation, internalization, and desensitization, paradoxically suppressing LH and FSH secretion - a property exploited therapeutically by long-acting GnRH agonists used in prostate cancer and endometriosis treatment.
Downstream Effects
The released gonadotropins then act on the gonads (testes and ovaries) to stimulate sex hormone production and gametogenesis. LH stimulates testosterone production in testicular Leydig cells and progesterone production in ovarian theca cells, while FSH supports spermatogenesis and follicular development respectively. This hypothalamic-pituitary-gonadal axis represents a classic neuroendocrine feedback system.
Research Findings
Research on gonadorelin and the GnRH system has been fundamental to understanding reproductive endocrinology and has led to numerous clinical applications in fertility medicine and hormone-dependent disease management.
Diagnostic Applications
Diagnostic studies have used gonadorelin stimulation tests to assess pituitary gonadotroph function and differentiate between hypothalamic and pituitary causes of hypogonadism. In the GnRH stimulation test, blood samples are taken before and after gonadorelin administration (typically 100 mcg IV or SC) to measure LH and FSH responses. A robust gonadotropin rise indicates intact pituitary function, while a blunted response suggests pituitary dysfunction. This test has been valuable in evaluating delayed puberty, amenorrhea, and other reproductive disorders.
Therapeutic Research in Hypogonadism
Clinical research has shown that pulsatile gonadorelin administration can induce ovulation in women with hypothalamic amenorrhea and stimulate spermatogenesis in men with hypogonadotropic hypogonadism. Studies using programmable portable pumps that deliver gonadorelin at physiological pulse intervals have demonstrated successful induction of puberty, fertility restoration, and maintenance of reproductive function in patients with GnRH deficiency.
Assisted Reproduction Research
Studies in assisted reproductive technology have explored gonadorelin for ovulation induction and controlled ovarian stimulation. While pulsatile GnRH therapy can achieve more physiological follicular recruitment than exogenous gonadotropins, it has largely been supplanted by recombinant gonadotropins for practical reasons including the need for pump devices and frequent dose adjustments.
Pulse Frequency Studies
Research into the pulsatile GnRH system has elucidated critical aspects of reproductive physiology, including the importance of pulse frequency in determining LH versus FSH dominance. Fast GnRH pulse frequencies (one pulse per hour) favor LH release, while slower frequencies favor FSH - explaining hormonal patterns across the menstrual cycle and enabling therapeutic manipulation of the LH:FSH ratio.
Research Applications
- Pituitary function testing and diagnostics
- Hypogonadotropic hypogonadism research
- Ovulation induction studies
- GnRH receptor pharmacology research
- Reproductive axis regulation studies
- Pulse generator and hypothalamic function research
- Delayed puberty evaluation
- Fertility restoration research
- Neuroendocrine feedback system studies
Safety Profile
Gonadorelin has a well-established safety profile from decades of clinical and diagnostic use. When administered appropriately, it is generally well-tolerated with predictable, manageable side effects.
Common Side Effects
Side effects are typically mild and transient, including headache, nausea, abdominal discomfort, flushing, and lightheadedness. With subcutaneous or intravenous administration, local injection site reactions such as pain, redness, or swelling may occur but are usually minor.
Allergic Reactions
Allergic reactions to gonadorelin are rare but have been reported. These may include skin rash, urticaria, bronchospasm, and in very rare cases, anaphylaxis. Appropriate precautions should be taken during administration, particularly in patients with history of peptide allergies.
Hormonal Effects
The primary consideration with gonadorelin use is the subsequent hormonal changes it induces. Acute gonadotropin release leads to temporary increases in sex hormone levels, which may cause mild symptoms related to estrogen or testosterone elevation. In diagnostic testing, these effects are transient and clinically insignificant.
Therapeutic Use Considerations
Therapeutic use requires pulsatile delivery systems (portable pumps) which are complex, expensive, and require patient training. Continuous rather than pulsatile administration leads to the opposite effect - suppression of gonadotropins - so proper administration technique is essential. Monitoring of reproductive hormone responses during therapy is important to ensure appropriate dosing.
Regulatory Status
Gonadorelin is approved for diagnostic use in many countries and has a long history of safe clinical application. Its use in research settings continues to provide valuable insights into reproductive endocrinology with an acceptable safety profile.
Scientific References
Customer Reviews
No reviews yet. Be the first to review this product!
Please sign in to write a review.
Research Use Only
This product is intended for research purposes only and is not for human consumption, therapeutic use, or diagnostic applications. Please ensure compliance with all applicable regulations and institutional guidelines.