Thermal Processing and Kinetic Stability: Understanding the Impact of Lyophilization on Peptide Integrity
Discover how lyophilization (freeze-drying) locks in peptide potency and why thermal processing quality determines the shelf life and effectiveness of products like BPC-157 and Semaglutide. Learn to spot high-quality manufacturing and store your research peptides for maximum stability.
When you start researching peptides to optimize your health—whether it’s for weight management, anti-aging, or enhanced recovery—you quickly run into a lot of scientific terms. You’ll hear about amino acid chains, reconstitution, and half-lives. But there is one concept that is arguably the most critical for ensuring the product you buy actually delivers the results you want: Lyophilization.
You might know it better as "freeze-drying."
If you have ever received a vial of peptides, you’ve likely noticed the white powder or solid "puck" at the bottom. That isn’t just dried liquid; it is the result of a highly sophisticated thermal process designed to pause time for that molecule. The quality of this process dictates whether your BPC-157 or Semaglutide remains potent for years or degrades into useless byproducts within weeks.
At Alpha Carbon Labs, we believe that understanding the "how" and "why" of manufacturing helps you make better decisions for your research. In this guide, we are looking under the hood of thermal processing and kinetic stability. We will strip away the heavy jargon and explain exactly how lyophilization protects peptide integrity, why the "cake" appearance matters, and how to spot quality processing in the research market.
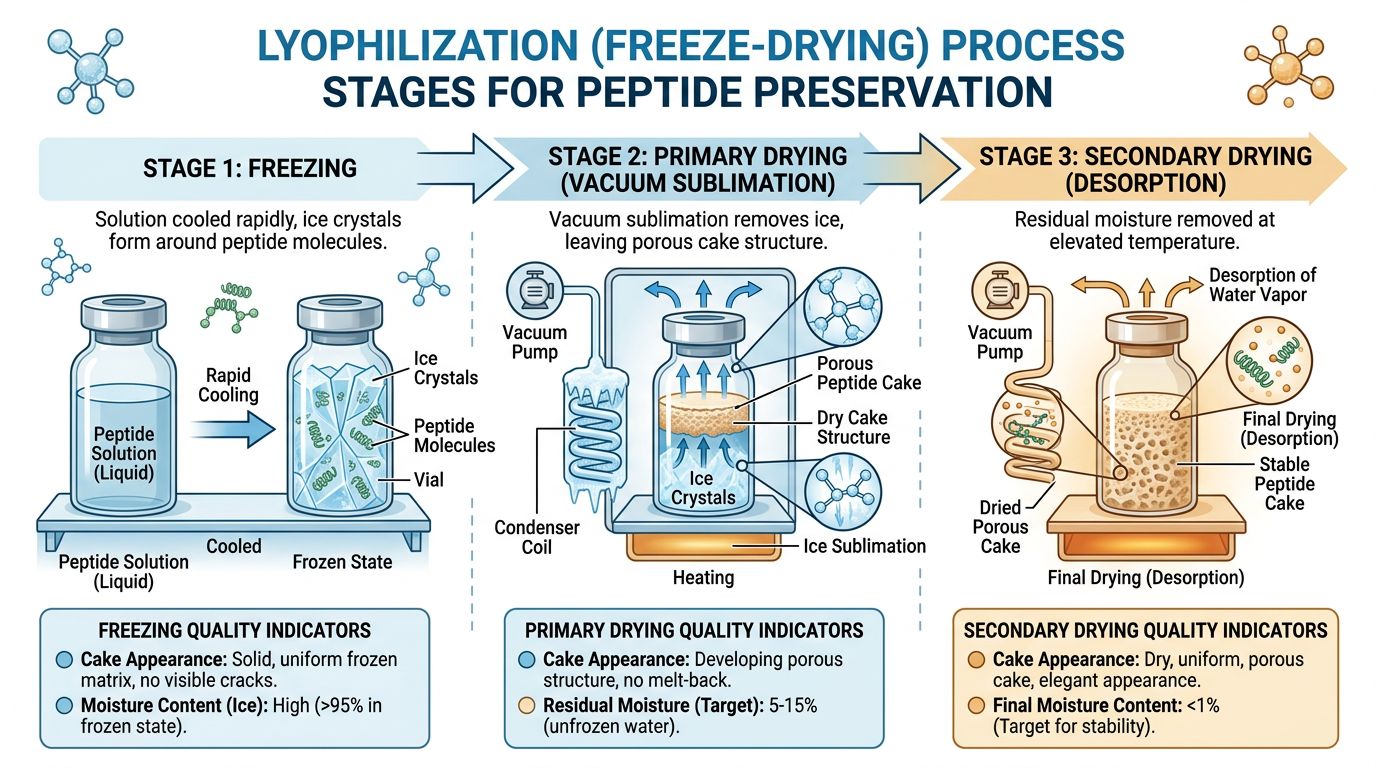
What is Lyophilization and Why Do We Do It?
Peptides are generally unstable in a liquid state. When dissolved in water, the bonds that hold the amino acids together are vulnerable. They can be broken apart by heat, changes in pH, or simply the passage of time. This breakdown is called hydrolysis. If a supplier shipped you a pre-mixed liquid peptide, chances are high that a significant percentage of it would degrade before it ever reached your mailbox.
Lyophilization is the solution. It is a process that removes water from the peptide solution while keeping the product frozen. This transitions the water directly from a solid (ice) to a gas (vapor) without ever passing back through the liquid phase—a phenomenon known as sublimation.
The Three Stages of Freezing
To understand why some peptides work better than others, you have to look at the three critical stages of this process. If a lab rushes any of these steps to save money, the peptide structure can be damaged.
- Freezing: The product is cooled until it is completely solid. The speed of freezing matters. If frozen too slowly, large ice crystals form, which can act like microscopic knives, damaging the delicate peptide structure. Flash freezing is often preferred to create a glass-like solid structure.
- Primary Drying (Sublimation): This is the main phase. The pressure in the chamber is lowered (creating a vacuum), and a small amount of heat is applied. This causes the ice to vaporize instantly. About 95% of the water is removed here.
- Secondary Drying (Desorption): There are still water molecules "stuck" to the peptide surface. The temperature is raised slightly to break these final bonds, leaving a product that is often less than 1% moisture.
The result? A peptide that is "kinetically stable." This means the degradation process has been slowed down so drastically that the product can remain stable at room temperature for significant periods, and for years when refrigerated.
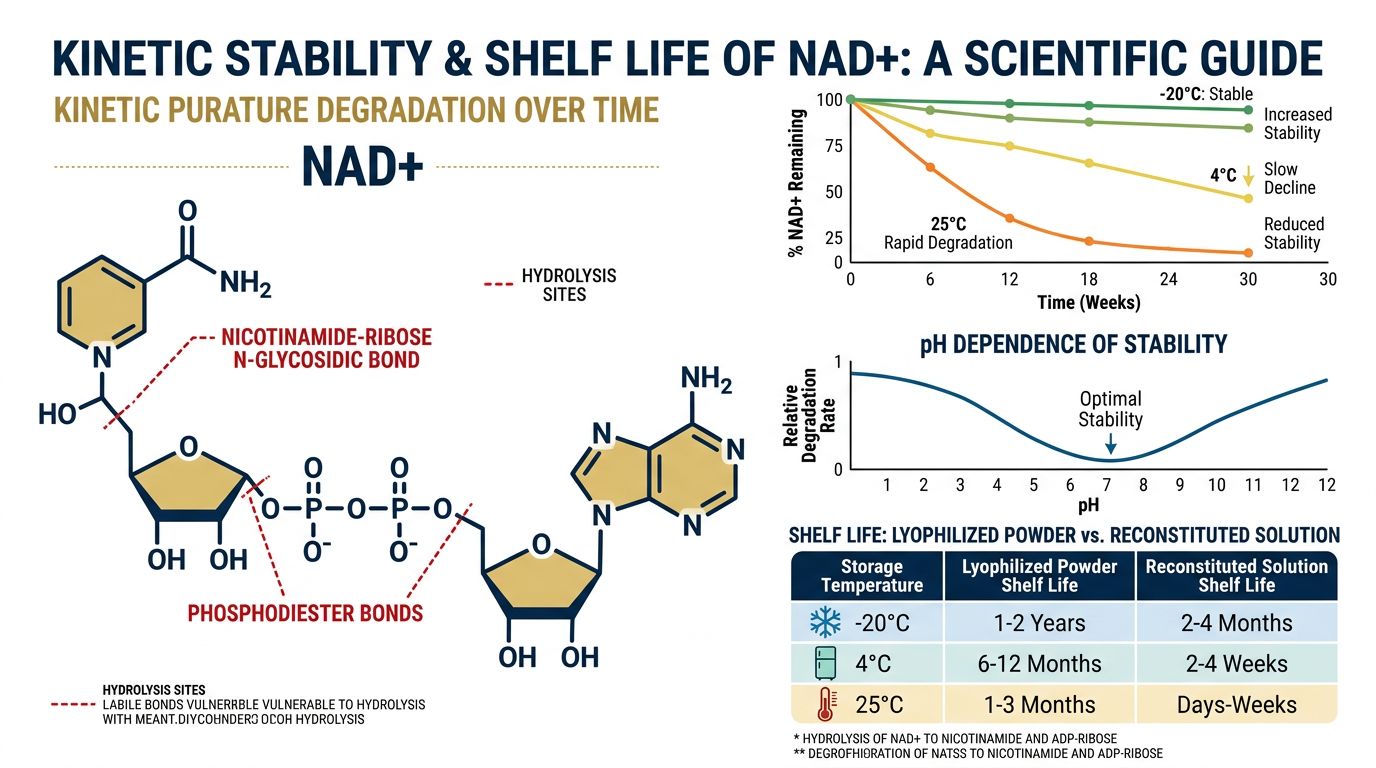
Kinetic Stability: The Science of Shelf Life
You may see terms like "High Purity" or "99% Pure" on a label, but purity refers to what is in the vial immediately after manufacturing. Stability refers to what is in the vial 3, 6, or 12 months later.
Kinetic stability is essentially the resistance of the peptide to change over time. In a solid, lyophilized state, the movement of molecules is restricted. They literally cannot move fast enough to bump into each other and react chemically. This is vital for complex peptides like Tirzepatide, which has a long and heavy chain of amino acids that can easily twist or break if given the chance.
Why Temperature Control Matters
Even in a dried state, heat is the enemy. Thermal energy makes molecules vibrate. If a lyophilized vial is exposed to extreme heat (like sitting in a mailbox in Arizona in July), those vibrations can increase enough to cause degradation, even without water present. However, a properly freeze-dried product is significantly more resilient than a liquid one.
For the end-user, this means that while we always recommend storing your lyophilized vials in the fridge or freezer, a short excursion at room temperature during shipping is rarely an issue for high-quality, properly dried research compounds.
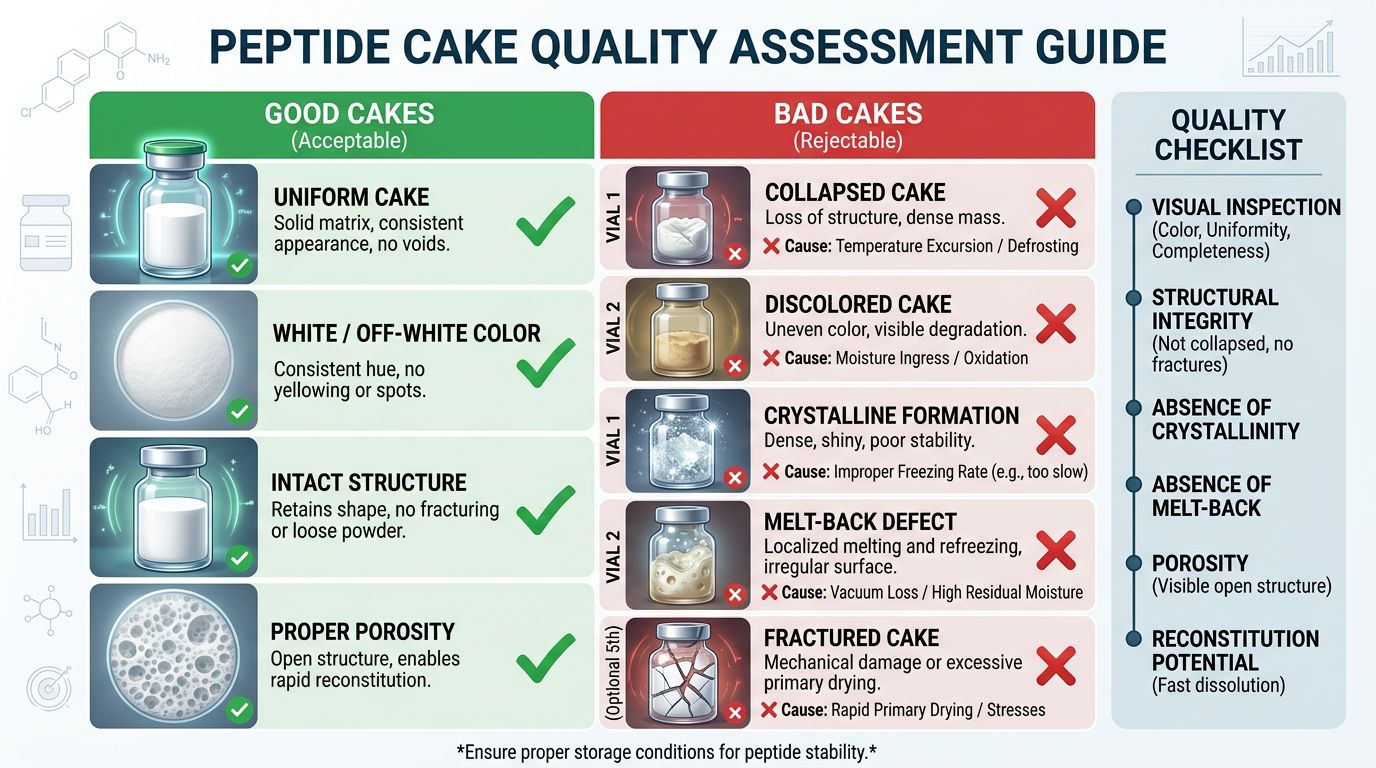
The "Cake": Cryoprotectants and Visual Quality
One of the most common questions we receive is: "Why does my peptide look like a solid puck, but the last one I bought was loose powder?"
This appearance refers to the "cake." The structure of the cake is determined by the cryoprotectants (bulking agents) used during the freeze-drying process. Pure peptides are often microscopic in quantity. If you lyophilized 5mg of TB-500 without a filler, you would barely see a few grains of dust in the vial.
To optimize stability and give the product a visible structure, labs use excipients. These act as "molecular bodyguards."
Common Cryoprotectants
- Mannitol: A sugar alcohol that is excellent at creating a rigid, elegant crystalline structure (the "puck"). It prevents the vial from collapsing and ensures the peptide dissolves quickly when you add water.
- Sucrose/Trehalose: These sugars form a "glassy" matrix. They surround the peptide molecules and protect them from the stress of freezing. They are particularly good for preventing protein unfolding.
Is a puck better than powder? Not necessarily. A solid puck usually indicates a "crystalline" freeze-drying cycle (often using Mannitol), while a looser powder might indicate an "amorphous" cycle. However, a collapsed or shrunken puck can be a sign of poor thermal processing—specifically, that the temperature got too high during the primary drying phase.
| Visual Aspect | What It Might Mean | Quality Verdict |
|---|---|---|
| Solid, Uniform Puck | Proper freeze-drying cycle using Mannitol. Good structural integrity. | Excellent |
| Loose White Powder | The cake may have broken during shipping (common) or a different bulking agent was used. | Generally Good (if purity verifies) |
| Shrunken or Melted Look | "Meltback" occurred. The product got too warm during drying, retaining moisture. | Poor Stability Risk |
| Yellow Discoloration | Oxidation or chemical reaction during processing. | Do Not Use |
Specific Peptides and Their Fragility
Not all peptides are created equal. Some are tanks, and some are like fine crystal glassware. Understanding the fragility of the specific research compound you are working with can help you store and handle it better.
The "Fragile" Class: Growth Hormone Secretagogues
Peptides related to growth hormone secretion, such as CJC-1295 and Frag 176-191, are notoriously sensitive. They have specific folding patterns that must be maintained to be effective. For these compounds, the lyophilization cycle is critical. If the lab rushes the freezing step, the bioactivity can drop significantly before it ever reaches the user.
Furthermore, once you reconstitute these (add water), their clock ticks faster than others. While BPC-157 is somewhat robust, Frag 176-191 should be handled gently—no vigorous shaking, just a gentle swirl.
The "Heavyweights": GLP-1 Agonists
Modern weight management titans like Semaglutide and Retatrutide are larger molecules. Their challenge isn't just fragility; it's solubility. A proper lyophilization process ensures that when you introduce bacteriostatic water, the peptide enters the solution clearly and doesn't clump. If you see persistent cloudiness in a GLP-1 peptide that doesn't clear up after a few minutes, it could indicate that the peptide degraded or aggregated during a poor manufacturing cycle.
The "Metal" Peptides: GHK-Cu
GHK-Cu (Copper peptide) is unique because it is bound to a copper ion. This gives it a distinct blue color. The challenge here is ensuring the copper ion stays attached during the phase change from liquid to gas. Superior thermal processing preserves that deep blue hue, which is a visual indicator of the copper bond integrity.
The Consumer Guide to Proper Reconstitution
We’ve discussed what happens in the lab. Now, let’s discuss what happens in your home. You are the final part of the stability equation. Once you introduce water to the lyophilized powder, you have broken the "stasis" created by the freeze-drying process. Kinetic stability drops, and the countdown begins.
Choosing the Right Solvent
For 99% of research peptides, Bacteriostatic Water is the gold standard. It contains 0.9% benzyl alcohol, which does two things:
- It keeps the solution sterile by preventing bacterial growth.
- It slightly stabilizes the peptide structure compared to plain sterile water.
Note on Plain Sterile Water: Use this only if you plan to use the entire vial immediately. It has no preservatives, so once opened, bacteria can grow rapidly.
The "Gentle Swirl" Technique
When you inject the water into the vial, aim for the glass wall, not directly onto the powder. The force of the stream can damage delicate peptides like Ipamorelin. Once the water is in, do not shake the vial like a protein shake. This creates bubbles and shear stress that can snap peptide bonds.
Instead, roll the vial gently between your palms or swirl it slowly until the powder is fully dissolved. If it takes a few minutes, be patient. Good things take time.
Advanced Storage: Maximizing Your Investment
You’ve invested in premium research compounds; don't let bad storage habits waste your money. Here is the hierarchy of storage stability based on the kinetic principles we’ve discussed.
1. Long-Term Storage (Unmixed Powder)
Best: Deep Freezer (-20°C). Kept here, lyophilized peptides can remain stable for years.
Good: Refrigerator (4°C). Stable for 1-2 years.
Acceptable: Cool, dark cabinet (Room Temp). Good for 1-3 months, depending on the specific peptide.
Pro Tip: Keep vials away from light. UV radiation destroys peptide bonds just as fast as heat does.
2. Active Use (Reconstituted Liquid)
Mandatory: Refrigerator (4°C).
Once mixed, most peptides like PT-141 or Melanotan II are viable for roughly 30 days. After this window, potency doesn't drop to zero instantly, but it begins a steady decline, and the risk of bacterial contamination increases.
How to Spot Quality Processing
As a consumer, you can't see the lyophilizer settings or the vacuum pressure charts. However, there are tell-tale signs of a high-quality product vs. a "bathtub chemistry" batch.
1. Transparency in Documentation
Does the supplier provide Certificates of Analysis (CoA)? A CoA doesn't just show purity; it verifies that the substance in the vial matches the label claim. At Alpha Carbon Labs, we believe in radical transparency. You can view our CoA Documents for every batch, ensuring that the 99% purity we claim is verified by third-party HPLC testing.
2. Consistent Fill Levels
While the look of the "cake" can vary (puck vs. powder), the amount of substance in the vials should look relatively consistent across a single batch. Large variations might indicate poor dosing machinery or inconsistent drying.
3. Solubility
A properly lyophilized peptide should dissolve readily. If you are struggling with clumps that won't dissolve after 10 minutes of gentle swirling, it may indicate a problem with the pH formulation or the extensive presence of impurities.
The Alpha Carbon Labs Commitment
We understand that our customers aren't just buying chemicals; they are investing in their physical optimization and well-being. That is why we don't cut corners on thermal processing.
Our manufacturing partners utilize advanced lyo-cycles tailored to the specific molecular weight and hydrophobicity of each peptide. We don't use a "one size fits all" drying cycle. Sensitive compounds like SS-31 get a gentler, longer cycle to preserve mitochondrial-protective properties, while more robust peptides get optimized cycles for perfect solubility.
When you choose Alpha Carbon Labs, you are choosing a product where the kinetic stability has been engineered from the molecular level up.
Summary: The Key Takeaways
- Lyophilization is a freeze-drying process that removes water to stop degradation, allowing peptides to be stored for long periods.
- Kinetic Stability means the peptide is "locked" in place. Heat and water unlock it, leading to breakdown.
- The "Cake" (puck vs. powder) is determined by cryoprotectants like Mannitol. Both can be high quality, but "melted" cakes are a red flag.
- Reconstitution requires care. Use Bacteriostatic water, inject gently, and never shake the vial.
- Quality Matters. Proper thermal processing ensures you get the full potency of the peptide, whether it's for weight loss, healing, or anti-aging.
Ready to experience the difference that professional-grade processing makes? Explore our full catalog of stabilized, high-purity research peptides and take the next step in your optimization journey.
References
- 1. Tang, X. C., & Pikal, M. J. (2004). Design of freeze-drying processes for pharmaceuticals: practical advice. Pharmaceutical research, 21(2), 191–200.
- 2. Manning, M. C., et al. (2010). Stability of Protein Pharmaceuticals: An Update. Pharmaceutical Research, 27, 544–575.
- 3. Carpenter, J. F., et al. (1997). Rational Design of Stable Protein Formulations: Theory and Practice. Pharmaceutical Biotechnology, 13, 1–25.
- 4. Kasper, J. C., & Friess, W. (2011). The freezing step in lyophilization: Physico-chemical fundamentals, freezing methods and consequences on process performance and quality attributes of biopharmaceuticals. European Journal of Pharmaceutics and Biopharmaceutics, 78(2), 248–263.
- 5. Chang, B. S., & Patro, S. Y. (1995). Freeze-Drying Process Development for Protein Pharmaceuticals. Molecular Biology and Biotechnology.
- 6. Wang, W. (2000). Lyophilization and development of solid protein pharmaceuticals. International Journal of Pharmaceutics, 203(1-2), 1–60.
- 7. B Bhatnagar, B. S., et al. (2007). Impact of thermal treatment on the physical stability of frozen formulations. Journal of Pharmaceutical Sciences.
- 8. Patel, S. M., et al. (2010). Lyophilized Drug Product Cake Appearance: What Is Acceptable? Journal of Pharmaceutical Sciences, 99(10).
- 9. Franks, F. (1998). Freeze-drying of bioproducts: putting principles into practice. European Journal of Pharmaceutics and Biopharmaceutics, 45, 221-229.
- 10. Lai, M. C., & Topp, E. M. (1999). Solid-state chemical stability of proteins and peptides. Journal of Pharmaceutical Sciences, 88(5), 489–500.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.