The Dual Agonism Advantage: Investigating Tirzepatide’s Impact on Metabolic Homeostasis
Explore the science behind Tirzepatide, a breakthrough dual GIP and GLP-1 receptor agonist, and discover how its unique molecular structure delivers superior metabolic outcomes.
Introduction: The Evolution of Incretin-Based Research
In the landscape of metabolic research, the discovery of the incretin effect—the phenomenon where oral glucose elicites a significantly larger insulin response than intravenous glucose—revolutionized our understanding of endocrine signaling. For decades, the primary focus of pharmacological intervention was directed toward the Glucagon-Like Peptide-1 (GLP-1) receptor. However, recent breakthroughs have highlighted the synergistic potential of combining GLP-1 signaling with Gastric Inhibitory Polypeptide (GIP, also known as Glucose-dependent Insulinotropic Polypeptide). This shift has led to the development of Tirzepatide, a synthetic 39-amino-acid peptide that represents a new class of dual-agonist therapeutics.
Tirzepatide is not merely a combination of two molecules; it is a single peptide engineered to activate both GIP and GLP-1 receptors. This "twincretin" approach addresses the limitations observed in mono-agonist therapies like Semaglutide, offering a more comprehensive modulation of metabolic homeostasis. Research indicates that the dual agonism of Tirzepatide produces superior outcomes in glucose regulation, lipid metabolism, and adiposity reduction compared to isolated GLP-1 activation.
Molecular Structure and Biochemical Engineering
The structural integrity of Tirzepatide is fundamental to its pharmacological profile. It is a linear peptide sequence based on the native GIP sequence, but modified to achieve a highly specific "imbalanced" agonism. To ensure the highest research standards, the purity of such sequences must be verified via COA documentation and rigorous quality control protocols.
Sequence Modifications and Lipidation
Tirzepatide incorporates several key structural modifications that distinguish it from native incretins:
- Aib Substitution: Inclusion of two non-proteogenic α-aminoisobutyric acid (Aib) residues at positions 2 and 13. These residues confer stability against Dipeptidyl Peptidase-4 (DPP-4) degradation, which typically inactivates native GIP and GLP-1 within minutes.
- C20 Fatty Diacid Side Chain: A C20 fatty diacid moiety is attached via a linker to the Lysine residue at position 20. This allows for high-affinity binding to albumin, extending the half-life of the peptide to approximately 5 days.
- Selective Residues: Critical residues from both GIP and GLP-1 are interspersed to ensure the molecule exhibits high affinity for both GIPR and GLP-1R, though it is optimized with a higher potency toward the GIP receptor.
This structural complexity requires sophisticated peptide synthesis techniques to maintain the precise orientation of the lipophilic side chain and the amino acid sequence integrity.
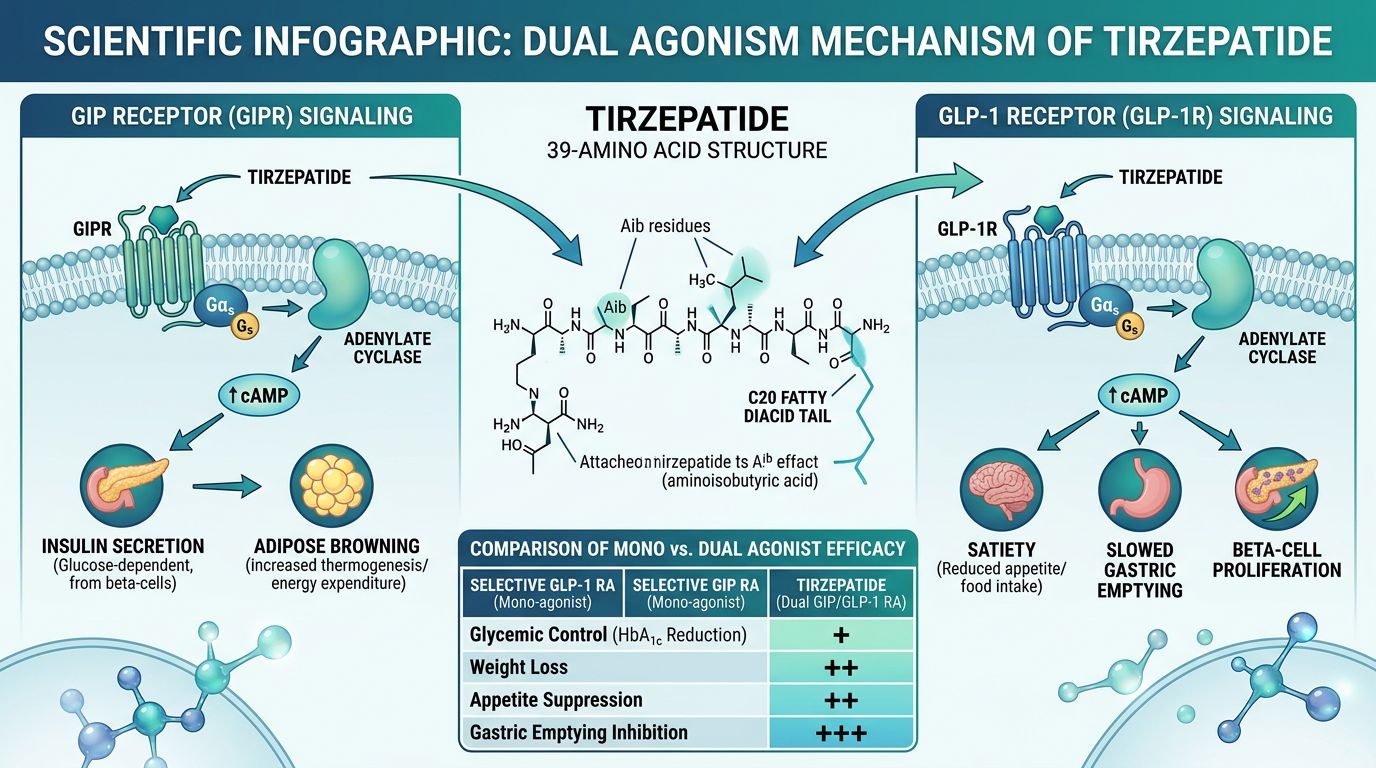
The Mechanism of Action: Dual Receptor Synergy
The distinct advantage of Tirzepatide lies in its ability to simultaneously recruit two distinct yet complementary signaling pathways. While GLP-1 has long been known to inhibit gastric emptying and suppress appetite, the addition of GIP receptor agonism introduces novel mechanisms for lipid handling and central energy balance.
GLP-1 Receptor Agonism
Activation of the GLP-1R in the pancreatic beta-cells stimulates glucose-dependent insulin secretion. In the central nervous system, GLP-1R signaling in the hypothalamus and hindbrain promotes satiety and reduces reward-driven feeding behavior. Furthermore, GLP-1 inhibits glucagon secretion from alpha-cells during periods of hyperglycemia.
GIP Receptor Agonism: The Metabolic "Master Key"
Historically, the role of GIP was debated, as its insulinotropic effect is often diminished in subjects with chronic metabolic dysfunction. However, research into GIPR agonism has revealed several critical functions that complement GLP-1:
- Adipose Tissue Buffering: GIP enhances the "buffering" capacity of white adipose tissue. It encourages postprandial lipid storage in subcutaneous fat, which prevents ectopic lipid deposition in the liver and skeletal muscle—a primary driver of insulin resistance.
- Nausea Mitigation: Early evidence suggests that GIP signaling in the brain may mitigate some of the gastrointestinal side effects typically associated with high-dose GLP-1 mono-agonists, allowing for higher therapeutic ceilings.
- Systemic Energy Expenditure: GIPR expression in the central nervous system may play a role in regulating energy expenditure and metabolic rate, though more research is needed to delineate these pathways fully.
The "Imbalanced" Agonism Phenomenon
Tirzepatide is described as an "imbalanced" agonist because it possesses a potency for GIPR comparable to the native hormone, whereas its GLP-1R potency is approximately 20 times weaker than native GLP-1. This design is intentional; by leading with GIP signaling, the peptide leverages the metabolic stability of the GIP system while providing sufficient GLP-1 signaling to modulate appetite and glucose without overwhelming the system with GI distress.
Research Findings: Tirzepatide vs. Mono-Agonists
Comparative studies have consistently demonstrated that Tirzepatide outperforms traditional GLP-1 agonists in several metabolic markers. The following table summarizes research data comparing Tirzepatide (at higher doses) with selective GLP-1 agonists like Semaglutide.
| Parameter | Tirzepatide (Dual Agonist) | Semaglutide (Mono-Agonist) | Mechanism of Difference |
|---|---|---|---|
| Weight Reduction | 15% - 22.5% | 10% - 15% | Synergistic central satiety and adipose tissue modulation. |
| HbA1c Reduction | Up to 2.4 - 3.0% | Up to 1.8% | Combined insulinotropic effect of GIP and GLP-1. |
| Lipid Profile | Significant reduction in VLDL and TG | Moderate reduction | GIP-mediated lipid clearance in adipose tissue. |
| Insulin Sensitivity | Markedly improved | Improved | Reduced ectopic fat and hepatosteatosis. |
Complementary Research Pathways
In addition to glucose and weight management, researchers are examining how Tirzepatide interacts with other peptide systems to enhance metabolic health. For instance, combining incretin therapy with mitochondrial stimulants like MOTS-c or SS-31 may offer insights into cellular energy restoration and muscle-to-metabolism signaling.
For research focuses on muscle preservation during rapid weight loss, scientists often look toward the Growth Hormone Secretagogue (GHS) family, such as Ipamorelin or Tesamorelin, to maintain lean mass while the dual-agonist enhances lipolysis. Furthermore, the role of anti-inflammatory peptides like KPV or BPC-157 in mitigating systemic inflammation associated with metabolic syndrome is a burgeoning area of study.
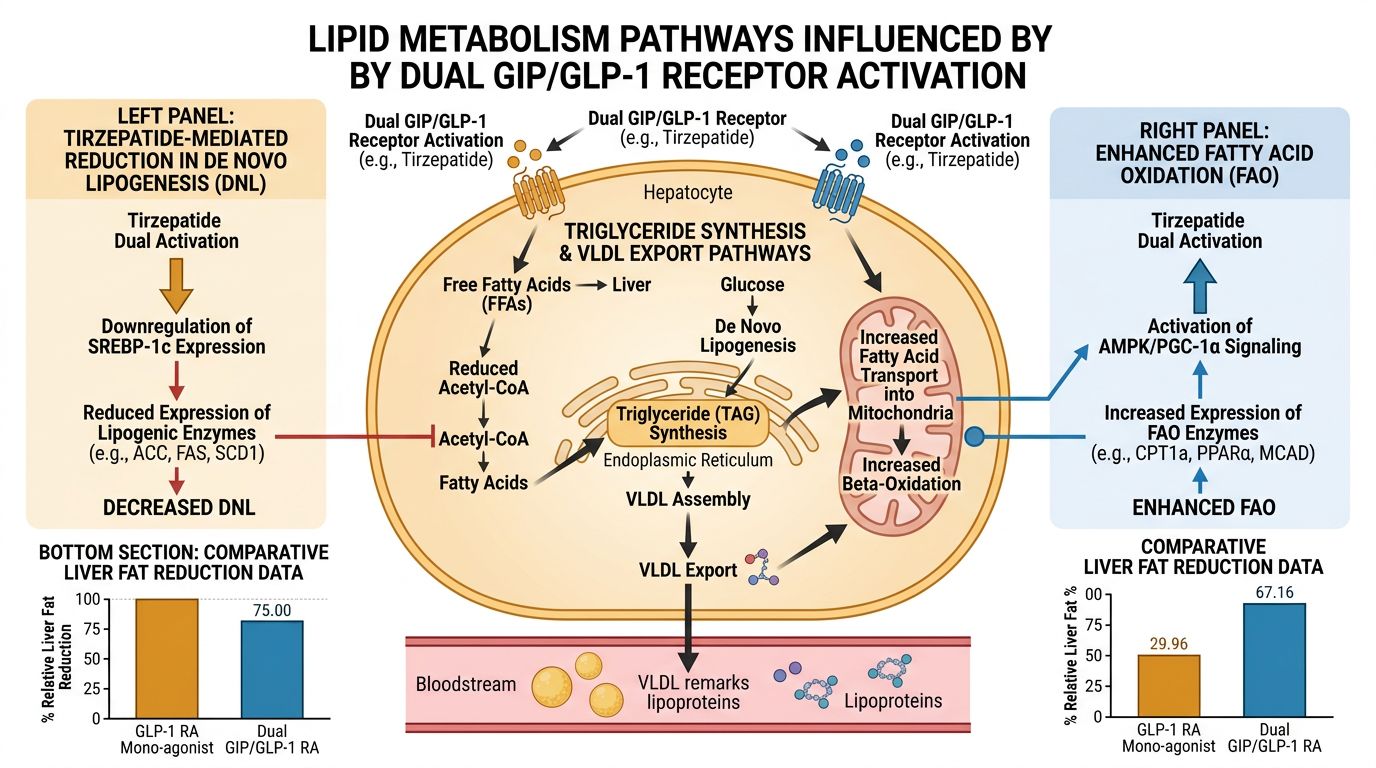
Advanced Metabolic Homeostasis: Impact on Lipid Metabolism
One of the most profound impacts of Tirzepatide is its effect on non-alcoholic fatty liver disease (NAFLD) and systemic lipid handling. While GLP-1 primarily acts by reducing caloric intake, the GIP component of Tirzepatide directly influences adipose tissue biology. It promotes the clearance of triglycerides from the blood and facilitates their storage in "healthy" depots. This reduces the burden of free fatty acids (FFAs) on the liver, thereby improving hepatic insulin sensitivity.
Researchers investigating lipid-lowering compounds like 5-amino-1mq often find that Tirzepatide provides a systemic foundation upon which targeted metabolic inhibitors can be studied. The dual-agonist effectively "resets" the metabolic baseline, allowing for more precise observation of secondary chemical interventions.
The Future of Multi-Agonism
The success of Tirzepatide has paved the way for triple-agonists. Emerging research into compounds like Retatrutide—which adds Glucagon receptor agonism to the GIP/GLP-1 backbone—suggests that even greater metabolic shifts may be possible. Additionally, the development of oral or prolonged-delivery formulations continues to be a high-priority area for peptide engineering.
Other novel metabolic researchers are exploring the role of SLU-PP-332 (an ERR agonist) and AICAR (an AMPK activator) in conjunction with incretin mimetics to simulate the physiological effects of exercise alongside caloric modulation.
Safety, Stability, and Research Considerations
While Tirzepatide demonstrates a favorable safety profile in clinical trials, researchers must account for the peptide's potent biological activity. Proper storage and reconstitution are essential to prevent degradation. As a multifaceted peptide, its effects on the hypothalamic-pituitary-adrenal (HPA) axis and the enteric nervous system are still being characterized.
Research protocols typically involve monitoring markers of pancreatic health (amylase/lipase) and gallbladder function, as rapid weight loss of any kind can influence biliary stasis. Furthermore, the peptide's impact on muscle proteolysis is an essential consideration, often addressed in studies by including myotropic agents like IGF-1LR3 or MGF to ensure that weight loss is predominantly adipose-derived.
Conclusion
Tirzepatide represents a monumental shift from mono-therapy to multi-pathway modulation. By harnessing the dual agonism of GIP and GLP-1, it provides a more robust and biologically harmonious approach to metabolic homeostasis. For researchers, Tirzepatide is not just a tool for weight or glucose management; it is a gateway to understanding the complex interplay between different endocrine signals and their collective influence on human physiology.
As the scientific community continues to explore the potential of dual and triple agonists, the importance of high-purity research materials cannot be overstated. Alpha Carbon Labs provides researchers with the essential compounds—from Tirzepatide and Semaglutide to next-generation metabolic research peptides—necessary to advance our understanding of metabolic health.
References
- 1. Jastreboff, A. M., et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine, 387(3), 205-217.
- 2. Frias, J. P., et al. (2021). Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine, 385(6), 503-515.
- 3. Nauck, M. A., & D'Alessio, D. A. (2022). Tirzepatide, a dual GIP/GLP-1 receptor co-agonist, the state of the art. Molecular Metabolism, 63, 101545.
- 4. Finan, B., et al. (2013). Unicellular peptide multi-agonism: a new paradigm for the treatment of metabolic diseases. Diabetologia, 56(8), 1667-1680.
- 5. Samms, R. J., et al. (2020). GIPR Agonism Mediates Weight Loss by Effects on Central Reward and Satiety Centers. JCI Insight, 5(11).
- 6. Müller, T. D., et al. (2022). The Incretin System: From Clinical Application to Future Therapeutic Vistas. Nature Reviews Endocrinology, 18, 549–569.
- 7. Willard, F. S., et al. (2020). Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight, 5(17).
- 8. Heise, T., et al. (2022). Tirzepatide Improves Gastrointestinal Transit Time and Bolus-Induced Gastric Secretion. Diabetes, Obesity and Metabolism, 24(5).
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.