Optimizing Protein Accretion: Technical Protocols for GHRP and GHRH Combinations in Myogenesis Research
A technical guide for researchers on standardizing dosing and timing of GHRP/GHRH combinations to maximize protein synthesis and myogenesis in laboratory settings.
Introduction: The Physiology of Myogenesis and Peptide Synergism
In the field of regenerative biology and musculoskeletal research, the optimization of protein accretion—the process by which protein synthesis exceeds breakdown—is a primary objective. While resistance stimulus and amino acid availability are the foundational substrates of myogenesis (muscle tissue formation), the hormonal environment dictates the rate and magnitude of this growth. Central to this regulatory environment is the Growth Hormone (GH) and Insulin-Like Growth Factor 1 (IGF-1) axis.
For researchers investigating peptide therapeutics, the goal is not merely to introduce exogenous GH, which can lead to downregulation of endogenous production, but to amplify the organism's natural pulsatile release. This is achieved through the strategic combination of Growth Hormone Releasing Hormones (GHRH) and Growth Hormone Releasing Peptides (GHRP). Understanding the technical interplay between these two classes of peptides allows for the development of protocols that maximize nitrogen retention and satellite cell proliferation while minimizing pituitary desensitization.
This article serves as a technical guide for optimizing protein accretion in research settings, focusing on the specific dosing, timing, and combinatorial logic of pairing compounds like CJC-1295 and Ipamorelin. By replicating natural physiology through precise administration, research can elucidate the upper limits of anabolic signaling.
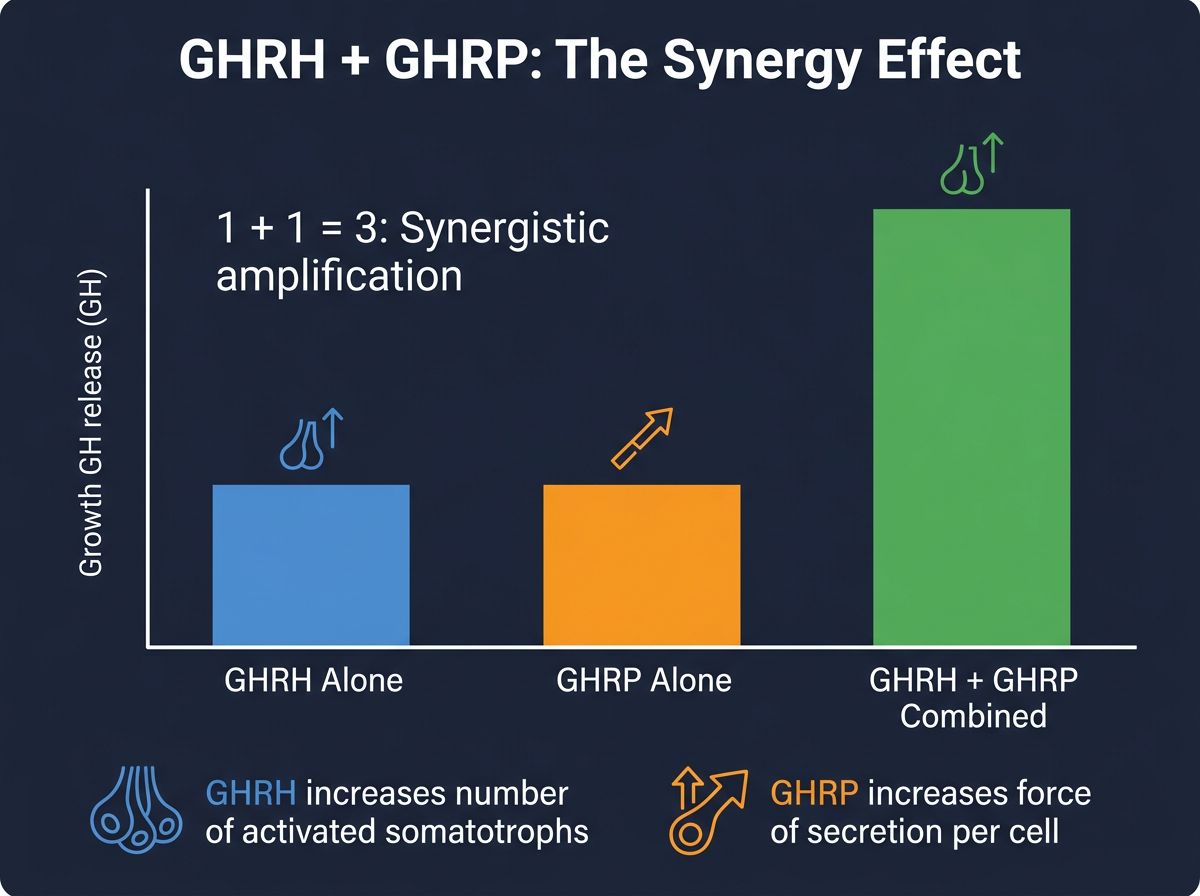
The Biological Basis of the "Pulse": Why Combinations are Superior
To optimize myogenesis, one must understand that Growth Hormone secretion is not linear; it is pulsatile. Evidence suggests that the pattern of GH exposure is just as critical for tissue growth as the total volume of GH released. Continuous exposure to GH (bleeding) can lead to receptor downregulation, whereas distinct, high-amplitude pulses trigger the liver to secrete IGF-1 effectively.
The pituitary gland releases GH under the influence of two opposing forces:
- Stimulation: Mediated by endogenous GHRH from the hypothalamus.
- Inhibition: Mediated by Somatostatin, which blocks GH release.
The superiority of combining a GHRH analogue with a GHRP lies in their distinct mechanisms of action. GHRH analogues stimulate the pituitary directly to release hormone stores. However, if Somatostatin tone is high, this release is blunted. GHRPs (ghrelin-mimetics) act functionally as Somatostatin inhibitors while simultaneously stimulating the pituitary via a different receptor (GHS-R1a).
When administered together, the result is synergistic rather than additive. A GHRH alone might produce a score of 2. A GHRP alone might produce a score of 4. Yet, combined, they often produce a GH pulse score of 10 or greater. This non-linear amplification is critical for maximizing protein accretion efficiency in research subjects.
The Primary Agents in Myogenesis Research
Before establishing protocols, it is essential to define the variables. The choice of peptide significantly alters the half-life and side-effect profile of the study.
| Class | Peptide | Primary Characteristic | Half-Life |
|---|---|---|---|
| GHRH | Mod GRF 1-29 (CJC-1295 No DAC) | Short-acting, mimics natural pulse | ~30 minutes |
| GHRH | CJC-1295 (With DAC) | Long-acting, elevated basal levels | ~6-8 days |
| GHRH | Tesamorelin | Highly potent, specific lipolytic effects | ~30-40 minutes |
| GHRP | Ipamorelin | Selective, no cortisol/prolactin spike | ~2 hours |
| GHRP | GHRP-6 | Strong appetite stimulation | ~2-3 hours |
| GHRP | GHRP-2 | High release, moderate cortisol risk | ~2-3 hours |
| Secretagogue | MK-677 (Ibutamoren) | Oral, sustained 24hr release | ~24 hours |
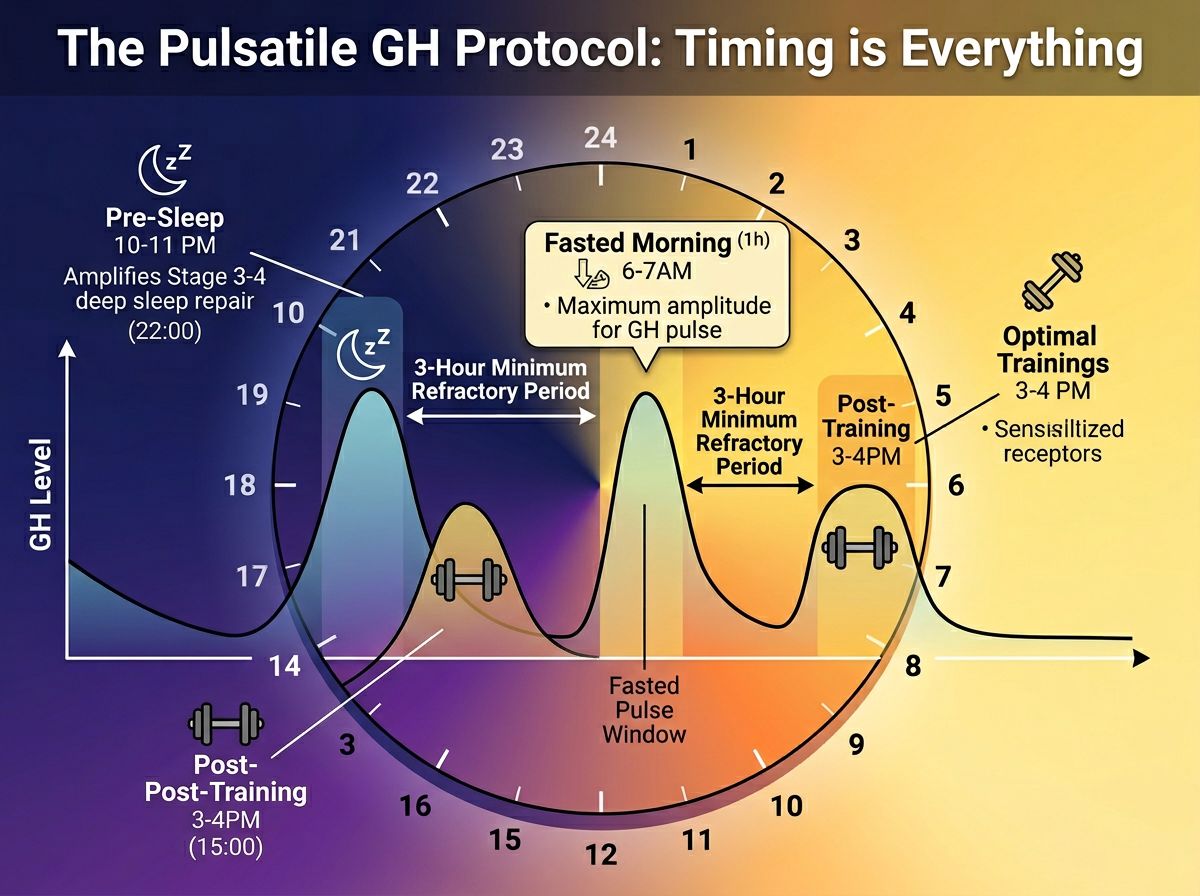
Protocol 1: The Specificity Protocol (Precision Pulse)
This protocol is considered the "Gold Standard" for pure protein accretion research where side effects such as water retention, lethargy, or hunger must be controlled. It utilizes reagents with short half-lives to create a massive, instantaneous spike in GH followed by a rapid return to baseline. This mimics youthful physiology.
Reagents
- GHRH: Mod GRF 1-29 (CJC-1295 No DAC)
- GHRP: Ipamorelin
The choice of Ipamorelin is distinct here because, unlike other GHRPs, it does not stimulate adrenocorticotropic hormone (ACTH) or cortisol significantly. High cortisol is catabolic (muscle wasting) and directly counteracts protein accretion.
Dosing Strategy: The Saturation Rule
Research indicates that the pituitary saturation dose for these peptides is approximately 1mcg per kg of body weight for the subject. Doses exceeding this yield diminishing returns (a flattened dose-response curve).
Standard Dosage Calculation:
- 100mcg Mod GRF 1-29
- 100mcg Ipamorelin
- Administered primarily via subcutaneous route.
Timing for Accretion
To maximize myogenesis, the timing of administration in relation to feeding and metabolic state is paramount. Insulin and Free Fatty Acids (FFAs) are negative regulators of GH release. High blood glucose or high circulating lipids will blunt the somatotroph response.
- The Fasted Morning Pulse: Administered immediately upon waking or 30 minutes prior to first feeding. This capitalizes on low insulin levels to maximize the pulse amplitude.
- The Post-Stimulus Pulse: Administered post-training or physical exertion. Physical stress naturally sensitizes the body to repair signals. However, this must be timed before the post-workout nutritional intake to avoid the insulin blunt.
- The Pre-Sleep Pulse: The most critical administration for hypertrophy. The majority of natural GH release and tissue repair occurs during Stage 3 and 4 Slow Wave Sleep (SWS). Enhancing this natural spike facilitates the mTOR pathway during the body's primary repair window.
Protocol 2: The Hypertrophy & Appetite Protocol
In research scenarios involving cachexia (muscle wasting) or where caloric surplus is difficult to achieve, the specific protocol above may be insufficient. Here, the goal is to drive protein synthesis while simultaneously stimulating ghrelin receptors to increase caloric intake.
Reagents
Mechanism Difference
GHRP-6 is a potent hunger inducer. By stimulating the ghrelin receptor in the hypothalamus, it signals a demand for fuel. This is synergistic with the anabolic drive of the GH pulse. If the subject consumes adequate protein and amino acids during this window, the substrate availability for protein accretion is maximized. Researchers must note that GHRP-6 and GHRP-2 can elevate prolactin and cortisol slightly, so this protocol requires careful monitoring using verified data from COA documents to ensure compound purity and dosage accuracy.
Advanced Pathways: The Role of MGF and Insulin Sensitivity
While the GHRH/GHRP stack stimulates the systemic release of IGF-1 from the liver, local muscular repair involves a splice variant of IGF-1 known as Mechano Growth Factor (MGF). In advanced research protocols, the systemic elevation of IGF-1 via the pituitary axis is often studied alongside local factors.
Furthermore, maintaining insulin sensitivity is crucial. Chronic elevation of GH can induce temporary insulin resistance (diabetogenic effect), which prevents amino acids from entering muscle cells efficiently. This is why pulsatile protocols (using Mod GRF 1-29) are generally favored over continuous bleed protocols (CJC-1295 DAC) for myogenesis. The "valleys" between the pulses allow insulin sensitivity to reset.
For research looking at long-term elevation of IGF-1 without daily injections, MK-677 provides a compelling oral alternative. However, researchers usually observe higher water retention with this compound, which may confound measurements of lean tissue accretion via scale weight, necessitating DEXA scans for accurate data.
Technical Considerations for Reconstitution and Storage
The efficacy of a protein accretion study is entirely dependent on the stability of the peptides used. GHRH, in particular, is fragile.
- Lyophilization: Peptides must remain lyophilized (freeze-dried) until use.
- Bacteriostatic Water: Reconstitution should be done with bacteriostatic water (0.9% benzyl alcohol) to maintain sterility for multi-dose vials.
- The "Fragility" Factor: Mod GRF 1-29 is susceptible to enzymatic degradation. Once reconstituted, it should be kept at 4°C and used within 15-20 days. Do not shake the vial vigorously; gentle rotation is sufficient to dissolve the peptide structure.
For laboratories sourcing these compounds, referencing quality control standards is defining. The presence of trifluoroacetic acid (TFA) salts or incomplete peptide chains can alter receptor binding affinity, skewing research data.
Protein Synthesis Measurements: How to Quantify Success
In a research setting, "feeling" stronger is not a metric. To scientifically validate the efficacy of a GHRP/GHRH protocol for protein accretion, specific biomarkers and physical measurements are employed.
1. Nitrogen Balance
A positive nitrogen balance is the clinical definition of an anabolic state. By measuring nitrogen intake (protein) vs. nitrogen excretion (urea), researchers can determine if the GH pulses are effectively reducing protein breakdown (proteolysis) or increasing synthesis.
2. IGF-1 Serum Levels
Measuring serum GH is difficult due to its pulsatile nature—missing the pulse by 20 minutes results in a near-zero reading. Instead, researchers measure IGF-1 levels. IGF-1 has a longer half-life and serves as a stable proxy for total GH secretion over the last 24-48 hours. A successful protocol involving Sermorelin or CJC-1295 should result in a statistically significant elevation of baseline IGF-1.
3. DEXA Body Composition
Dual-Energy X-ray Absorptiometry (DEXA) remains the gold standard for differentiating between lean muscle mass accrual and fluid retention. This is particularly vital when using compounds like MK-677 or GHRP-2, where water weight can mask true tissue accretion.
Safety Profiles and Desensitization
A common hurdle in myogenesis research is the "tachyphylaxis" or desensitization of the pituitary gland. If the somatotrophs are bombarded with constant signals to release GH, they eventually become refractory.
The 3-Hours Rule: To prevent desensitization, research protocols generally dictate a minimum of 3 hours between administrations of GHRP/GHRH combinations. This refractory period allows the pituitary to resynthesize GH stores and resets the receptor sensitivity.
Prolactin Management: When utilizing GHRP-2, GHRP-6, or Hexarelin, researchers must monitor prolactin levels. Elevated prolactin can suppress testosterone, which is counter-productive to myogenesis. Ipamorelin does not carry this risk, which is why it is preferred for longer-duration studies.
The Role of Cortisol
Cortisol is catabolic. It initiates the breakdown of muscle tissue to free up amino acids for gluconeogenesis. While GH is lipolytic (fat burning) and anabolic (muscle building), a spike in cortisol accompanying a GH pulse can negate the protein accretion benefits. This differentiates the "dirty" GHRPs (Hexarelin, high-dose GHRP-2) from the "clean" GHRPs (Ipamorelin). For pure myogenesis, minimizing the cortisol response is as important as maximizing the GH response.
Emerging Research: Mitochondrial Support
Newer frontiers in protein accretion research look beyond the GH axis to mitochondrial health. Muscle tissue is metabolically expensive; it requires significant ATP to maintain. Research into mitochondrial peptides like SS-31 or MOTS-c suggests that improving mitochondrial efficiency may raise the "ceiling" for muscle growth by improving the energy handling of the cell. Combining a GHRP/GHRH stack with mitochondrial support represents a comprehensive approach to cellular bioenergetics.
Conclusion
Optimizing protein accretion in a research environment requires more than simply introducing growth factors; it requires the precise replication of physiological rhythms. The combination of a GHRH (specifically CJC-1295 No DAC) and a GHRP (specifically Ipamorelin) offers the most targeted approach to elevating IGF-1 levels without the confounding variables of cortisol elevation or insulin resistance.
By adhering to saturation dosing (1mcg/kg), respecting the 3-hour refractory period, and timing administrations around low-insulin windows, researchers can effectively study the mechanisms of myogenesis. It is through these rigorous, standardized protocols that the full potential of peptide therapeutics in muscle wasting diseases and sarcopenia can be understood. As always, the foundation of reproducible results lies in the purity of the reagents; ensure all peptides act in accordance with current peptide synthesis standards.
References
- Bowers, C. Y. (1998). Growth hormone-releasing peptide (GHRP). Cellular and Molecular Life Sciences CMLS, 54(12), 1316-1329.
- Veldhuis, J. D., & Bowers, C. Y. (2003). Integrating GHS into the GH–IGF-I axis. International Journal of Obesity, 27, S86-S98.
- Popovic, V., et al. (2000). GHRH and GHRP-6 for testing the pituitary-GH reserve. Journal of Clinical Endocrinology & Metabolism, 85(7), 2618-2620.
- Raun, K., et al. (1998). Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology, 139(5), 552-561.
- Teichman, S. L., et al. (2006). Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a therapeutic peptide analog of GH-releasing hormone. The Journal of Clinical Endocrinology & Metabolism, 91(3), 799-805.
- Svensson, J., et al. (1998). Two-month treatment of obese subjects with the oral growth hormone secretagogue MK-677 increases cortisol, GH, and IGF-I. The Journal of Clinical Endocrinology & Metabolism, 83(2), 362-369.
- Veldhuis, J. D., et al. (2001). Mechanisms of GHRH and GHRP interaction in the human. Endocrine Reviews, 22(5), 629-652.
- Phung, L. T., et al. (2000). The effects of growth hormone-releasing peptide-2 (GHRP-2) on the release of growth hormone and growth performance in swine. Domestic Animal Endocrinology, 18(3), 279-291.
- Hartman, M. L., et al. (1995). Pulsatile GH release in human subjects: metabolic aspects. American Journal of Physiology-Endocrinology and Metabolism, 269(3), E453.
- Velloso, C. P. (2008). Regulation of muscle mass by growth hormone and IGF-I. British Journal of Pharmacology, 154(3), 557-568.
References
- 1. Bowers, C. Y. (1998). Growth hormone-releasing peptide (GHRP). Cellular and Molecular Life Sciences CMLS, 54(12), 1316-1329.
- 2. Veldhuis, J. D., & Bowers, C. Y. (2003). Integrating GHS into the GH–IGF-I axis. International Journal of Obesity, 27, S86-S98.
- 3. Popovic, V., et al. (2000). GHRH and GHRP-6 for testing the pituitary-GH reserve. Journal of Clinical Endocrinology & Metabolism, 85(7), 2618-2620.
- 4. Raun, K., et al. (1998). Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology, 139(5), 552-561.
- 5. Teichman, S. L., et al. (2006). Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a therapeutic peptide analog of GH-releasing hormone. The Journal of Clinical Endocrinology & Metabolism, 91(3), 799-805.
- 6. Svensson, J., et al. (1998). Two-month treatment of obese subjects with the oral growth hormone secretagogue MK-677 increases cortisol, GH, and IGF-I. The Journal of Clinical Endocrinology & Metabolism, 83(2), 362-369.
- 7. Veldhuis, J. D., et al. (2001). Mechanisms of GHRH and GHRP interaction in the human. Endocrine Reviews, 22(5), 629-652.
- 8. Phung, L. T., et al. (2000). The effects of growth hormone-releasing peptide-2 (GHRP-2) on the release of growth hormone and growth performance in swine. Domestic Animal Endocrinology, 18(3), 279-291.
- 9. Hartman, M. L., et al. (1995). Pulsatile GH release in human subjects: metabolic aspects. American Journal of Physiology-Endocrinology and Metabolism, 269(3), E453.
- 10. Velloso, C. P. (2008). Regulation of muscle mass by growth hormone and IGF-I. British Journal of Pharmacology, 154(3), 557-568.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.