Bioenergetic Shifts: How Retatrutide Redefines Incretin Research Parameters
Explore the scientific shift from mono-agonism to triple-agonism as Retatrutide integrates glucagon receptor activation to redefine energy expenditure and lipid oxidation parameters in metabolic research.
Introduction: The Third Axis of Metabolic Regulation
In the rapidly evolving landscape of metabolic research, the transition from mono-agonism to multi-agonism represents a paradigm shift in how we understand and manipulate energy homeostasis. While Semaglutide established the potency of GLP-1 receptor agonism and Tirzepatide introduced the synergistic effects of GIP integration, Retatrutide (LY3437943) introduces a critical third component: the Glucagon receptor (GCGR) agonist. This "triple agonist" approach does not merely add to the existing effects of its predecessors; it fundamentally redefines the bioenergetic parameters of incretin research by addressing the energy expenditure side of the thermodynamic equation.
For researchers, the inclusion of glucagon agonism represents a bold departure from traditional insulin-centric models. Glucagon, often mischaracterized solely as a hyperglycemic agent, is now recognized as a potent regulator of lipid metabolism and thermogenesis. By balancing GLP-1, GIP, and Glucagon activities in a single 39-amino-acid peptide, Retatrutide allows for a comprehensive interrogation of metabolic flux, offering insights into non-shivering thermogenesis, hepatic lipid oxidation, and the mitigation of the metabolic slowdown typically associated with caloric restriction.
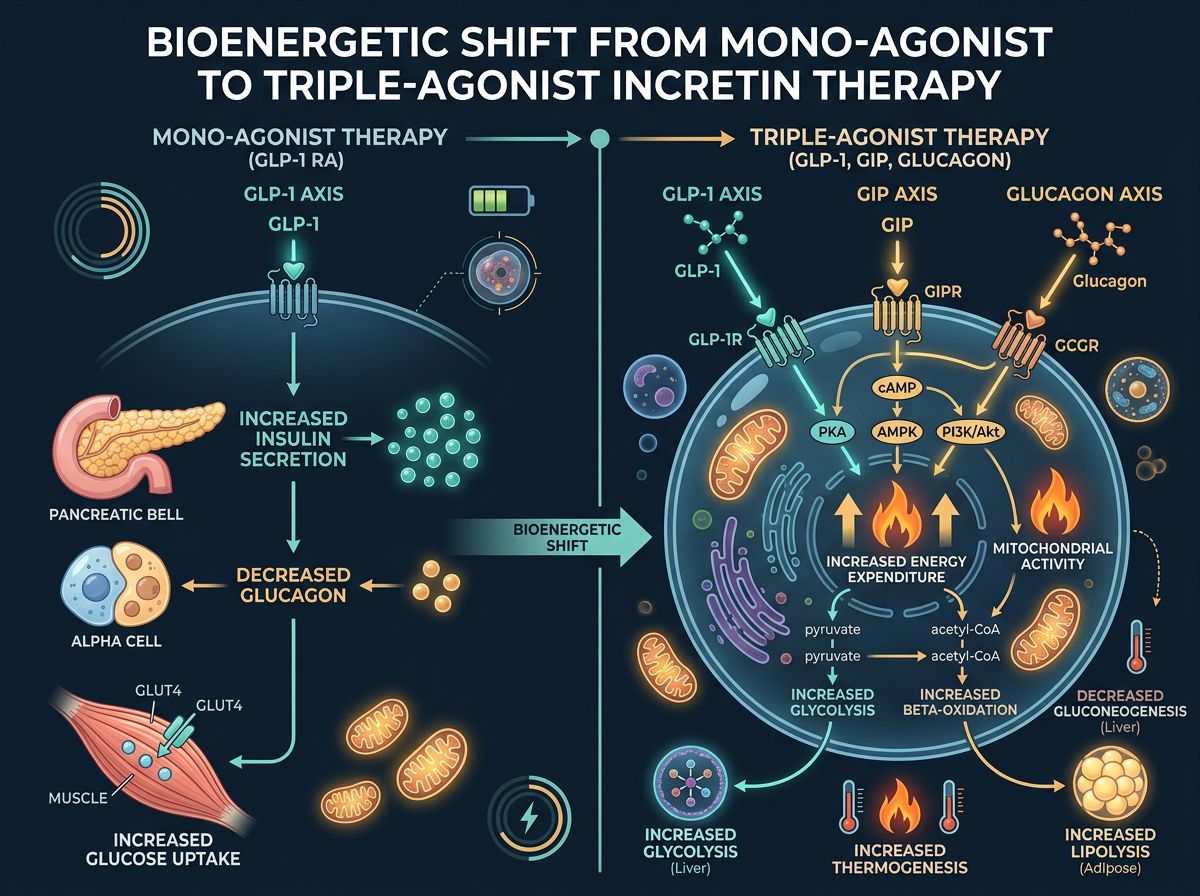
The Structural Biochemistry of Retatrutide
Retatrutide is an oxyntomodulin-based triple agonist peptide. To achieve its specific potency profile across three distinct receptors, the peptide backbone underwent rigorous engineering at our peptide synthesis facilities. The sequence is based on the GIP backbone but incorporates specific substitutions that facilitate multi-receptor engagement:
- Aib (Alpha-aminoisobutyric acid) at position 2: Enhances metabolic stability against Dipeptidyl peptidase-4 (DPP-4) degradation.
- Fatty acid acylation (C20 diacid): Positioned at Lysine-17 via a linker, this modification allows for high-affinity albumin binding, extending the half-life to approximately 6 days, suitable for once-weekly research protocols.
- Chimeric sequence optimization: The amino acid sequence is tuned to provide a potency ratio that favors GIP and GLP-1 while maintaining a calculated "breath" of Glucagon activity to maximize fat oxidation without inducing pathological hyperglycemia.
Potency Comparison Table
| Peptide | GLP-1R Activity | GIPR Activity | GCGR Activity |
|---|---|---|---|
| Semaglutide | Full Agonist | None | None |
| Tirzepatide | Full Agonist | Full Agonist | None |
| Retatrutide | Full Agonist | Full Agonist (High) | Partial Agonist (Optimized) |
Mechanism of Action: Beyond Appetite Suppression
The primary mechanism of action for traditional GLP-1 RAs like Semaglutide revolves around the slowing of gastric emptying and the modulation of hypothalamic satiety signals. While Retatrutide retains these pathways, the activation of the Glucagon receptor introduces a secondary and tertiary layer of bioenergetic manipulation.
1. Glucagon-Mediated Energy Expenditure
Glucagon signaling in the liver and potentially in brown adipose tissue (BAT) stimulates pathways that increase the basal metabolic rate (BMR). In murine and human models, GCGR activation promotes the transcription of genes involved in thermogenesis, such as UCP1 (Uncoupling Protein 1). By uncoupling oxidative phosphorylation from ATP production, energy is dissipated as heat, thereby increasing "caloric burn" independent of physical activity. This is a critical distinction from GLP-1 monotherapy, which typically results in a compensatory drop in metabolic rate as weight is lost.
2. Hepatic Lipid Oxidation and Autophagy
One of the most striking findings in Retatrutide research is its impact on Intrahepatic Triglyceride (IHTG) content. Glucagon actvation enhances mitochondrial fatty acid oxidation (FAO) in the liver. By activating the cAMP-PKA pathway, Retatrutide shifts the liver from a state of lipid storage to a state of lipid utilization. Research has shown that triple agonism can lead to a near-total normalization of hepatic fat content in models of NAFLD (Non-Alcoholic Fatty Liver Disease).
3. GIP: The Metabolic Buffer
The role of GIP in the Retatrutide complex is to act as a metabolic stabilizer. GIP receptor (GIPR) agonism has been shown to protect against the potentially diabetogenic effects of glucagon while synergizing with GLP-1 to improve insulin sensitivity in white adipose tissue. This triad creates a "safe" environment for glucagon activity, allowing for high rates of lipolysis without the adverse elevation in blood glucose that usually accompanies pure glucagon signaling.
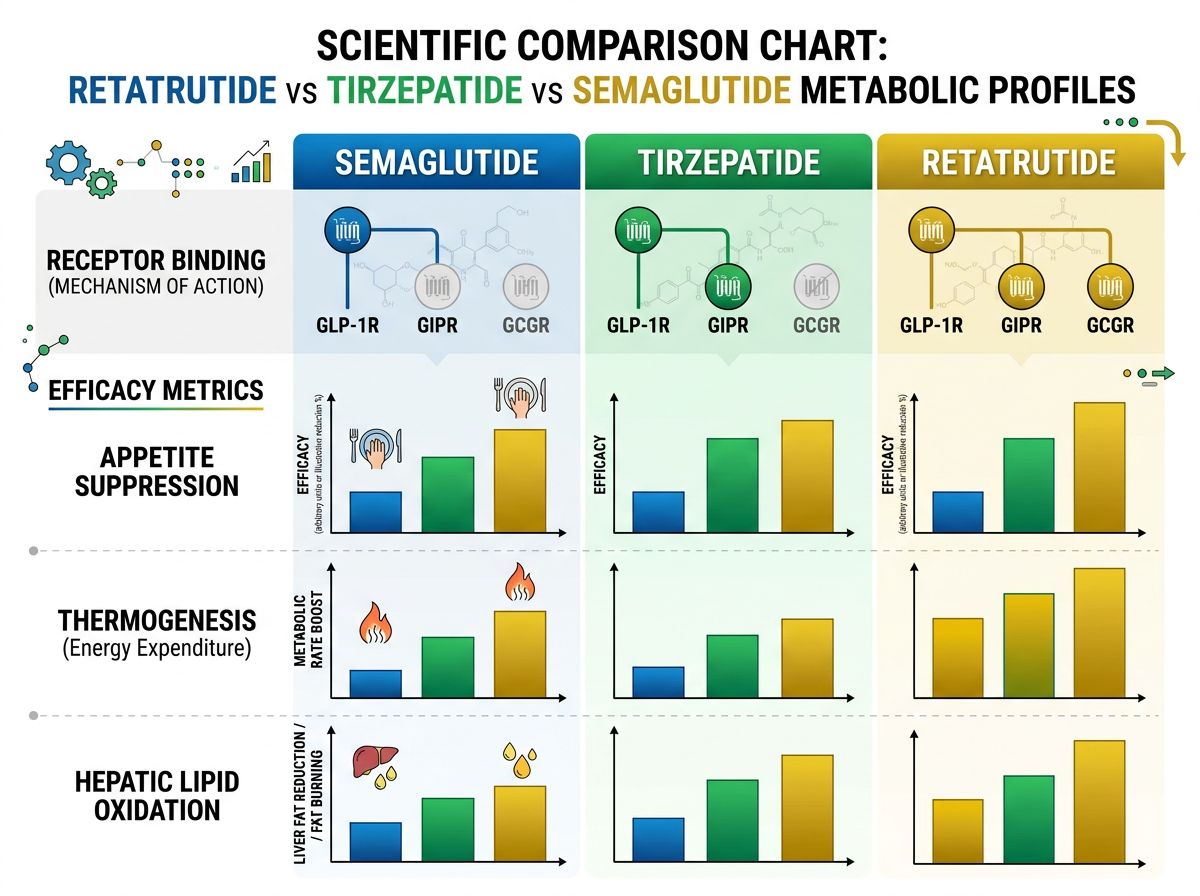
Comparative Analysis: Retatrutide vs. Tirzepatide and Semaglutide
In clinical research cohorts, Retatrutide has demonstrated a weight loss trajectory that exceeds even the highly effective Tirzepatide. The reason lies in the metabolic "floor." When a biological system undergoes significant weight loss, it typically triggers an adaptive thermogenesis response—essentially, the body becomes more efficient to prevent further weight loss. Retatrutide's glucagon component appears to counteract this adaptation.
Lipid Profile Shifts
Unlike AOD9604, which targets lipolysis through a growth hormone fragment mechanism, Retatrutide leverages systematic incretin signaling to alter the whole-body lipid profile. Researchers have observed significant reductions in LDL cholesterol, VLDL, and triglycerides that outpace GLP-1 mono-agonists. This suggests that the bioenergetic shift is not just about weight, but about a fundamental restructuring of lipid metabolism.
Advanced Research Applications: Beyond Weight Loss
While the focus on Retatrutide often centers on weight reduction, its unique bioenergetic profile opens several other avenues for scientific investigation:
Neuroprotective Research
Similar to Dihexa or Semax, there is growing interest in how metabolic peptides influence neuro-inflammation. The improved insulin sensitivity and reduced systemic inflammation provided by triple agonism may have secondary effects on cognitive health, particularly in models of metabolic-associated cognitive decline.
Organ Protection and Fibrosis
The impact of Retatrutide on the liver (specifically the resolution of steatosis) suggests potential applications in fibrotic research. Researchers studying peptides like BPC-157 for tissue repair may find that Retatrutide provides a complementary systemic environment by reducing the lipotoxic load on peripheral organs.
Cardiovascular Bioenergetics
By stimulating the GCGR, Retatrutide may influence cardiac chronotropy and inotropy. While this requires careful monitoring in research settings, it provides a unique model for studying the heart's metabolic flexibility and its shift between glucose and fatty acid oxidation.
Safety and Quality Control in Peptide Research
Given the high potency of triple-agonist peptides, the integrity of the research material is paramount. At Alpha Carbon Labs, we ensure that every batch of Retatrutide undergoes rigorous testing. Researchers should always consult the COA documents and our quality control protocols to verify purity and peptide content.
The complexity of triple agonism means that impurities or incorrect peptide folding (iso-D-aspartate formation, etc.) can lead to off-target effects or loss of receptor specificity. Ensuring a 99%+ purity profile is non-negotiable for obtaining reproducible data in metabolic studies.
Current Research Landscape: Phase 2 and Beyond
Recent data published in the New England Journal of Medicine highlighted that Retatrutide achieved weight loss of up to 24.2% over a 48-week period at the highest research doses. This level of efficacy was previously thought to be attainable only via bariatric surgery. For researchers, these results underscore the importance of the glucagon receptor as a lever for metabolic intervention.
Furthermore, the data on Mazdutide (another GLP-1/glucagon co-agonist) and Cagrilintide (an amylin analogue often paired with GLP-1) suggests that the future of metabolic research is inherently multi-modal. Retatrutide, however, remains the "gold standard" for triple agonism due to its inclusion of the GIP receptor, which serves as the essential balancer for the glucagon-GLP-1 axis.
Practical Considerations for Researchers
When designing protocols involving Retatrutide, several factors must be considered to maintain scientific rigor:
- Dose Escalation: Due to the gastrointestinal effects of GLP-1 and the metabolic stimulating effects of glucagon, a gradual dose-escalation protocol is standard in the literature to minimize "off-target" stress on the research subject.
- Hydration and Electrolytes: The زيادة (increase) in metabolic rate and potential diuretic effect of glucagon agonism may require researchers to monitor hydration levels more closely than with Semaglutide alone.
- Comparative Controls: To isolate the effect of the glucagon component, researchers often use Tirzepatide as a control group, as it contains the GLP-1 and GIP components but lacks the GCGR activation.
Conclusion
Retatrutide represents a landmark achievement in peptide engineering and metabolic science. By integrating glucagon receptor agonism, it moves research beyond the "satiety-only" model of weight loss into a more comprehensive "bioenergetic" model. This allows for the simultaneous reduction of caloric intake and the optimization of caloric expenditure, alongside potent effects on hepatic health and lipid metabolism.
As we continue to explore the capabilities of peptides like Retatrutide, Survodutide, and Tesamorelin, the focus of metabolic research is shifting toward "metabolic flexibility"—the ability of the body to efficiently switch and utilize different fuel sources. Retatrutide is, currently, the most sophisticated tool available for the study of this phenomenon.
For high-purity research materials and detailed technical data on the latest in incretin research, explore our full catalog of metabolic peptides and review our latest COA data.
References
- 1. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. New England Journal of Medicine. 2023.
- 2. Knerr PJ, et al. Next generation GLP-1/GIP/glucagon triple agonists for the treatment of obesity and type 2 diabetes. Cell Metabolism. 2023.
- 3. Coskun T, et al. LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: From bench to bedside. Cell Metabolism. 2022.
- 4. Müller TD, et al. Glucagon-like peptide 1 (GLP-1). Molecular Metabolism. 2019.
- 5. Heppner KM, et al. Glucagon regulation of energy expenditure. Physiology & Behavior. 2015.
- 6. Finan B, et al. A strategically designed tri-agonist multispecificity for the treatment of metabolic syndrome. Science Translational Medicine. 2014.
- 7. Capozzi ME, et al. The Glucose-Dependent Insulinotropic Polypeptide Receptor Is Essential for the Glucoregulatory Role of the Glucagon Receptor. Diabetes. 2019.
- 8. Sanyal AJ, et al. Retatrutide in Patients with Non-alcoholic Fatty Liver Disease: A Randomized Phase 2 Trial. Hepatology. 2023.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.