Angiogenic Signaling: The Wolverine Stack’s Role in Neovascularization and Blood Flow Dynamics
Explore the molecular synergy of BPC-157 and TB-500, the "Wolverine Stack," and how this combination drives neovascularization and tissue repair in research models.
Introduction to Advanced Angiogenic Research
The pursuit of regenerative medicine has long focused on the critical challenge of revascularization. In scientific circles, the synergistic application of BPC-157 and TB-500—often colloquially referred to as the "Wolverine Stack"—has emerged as a focal point for investigating accelerated tissue repair and neovascularization. While both peptides individually possess potent biological activity, their combined administration exhibits a unique pharmacological profile that target multiple pathways of the angiogenic cascade.
Angiogenesis, the physiological process through which new blood vessels form from pre-existing vessels, involves a complex interplay of growth factors, extracellular matrix (ECM) remodeling, and endothelial cell migration. When tissue undergoes trauma or ischemia, the restoration of blood flow is the rate-limiting step in recovery. This article explores the deep molecular mechanisms by which this dual-peptide approach facilitates these processes, focusing on the modulation of the Vascular Endothelial Growth Factor (VEGF) pathway, the recruitment of endothelial progenitor cells, and the stabilization of the microvascular environment.
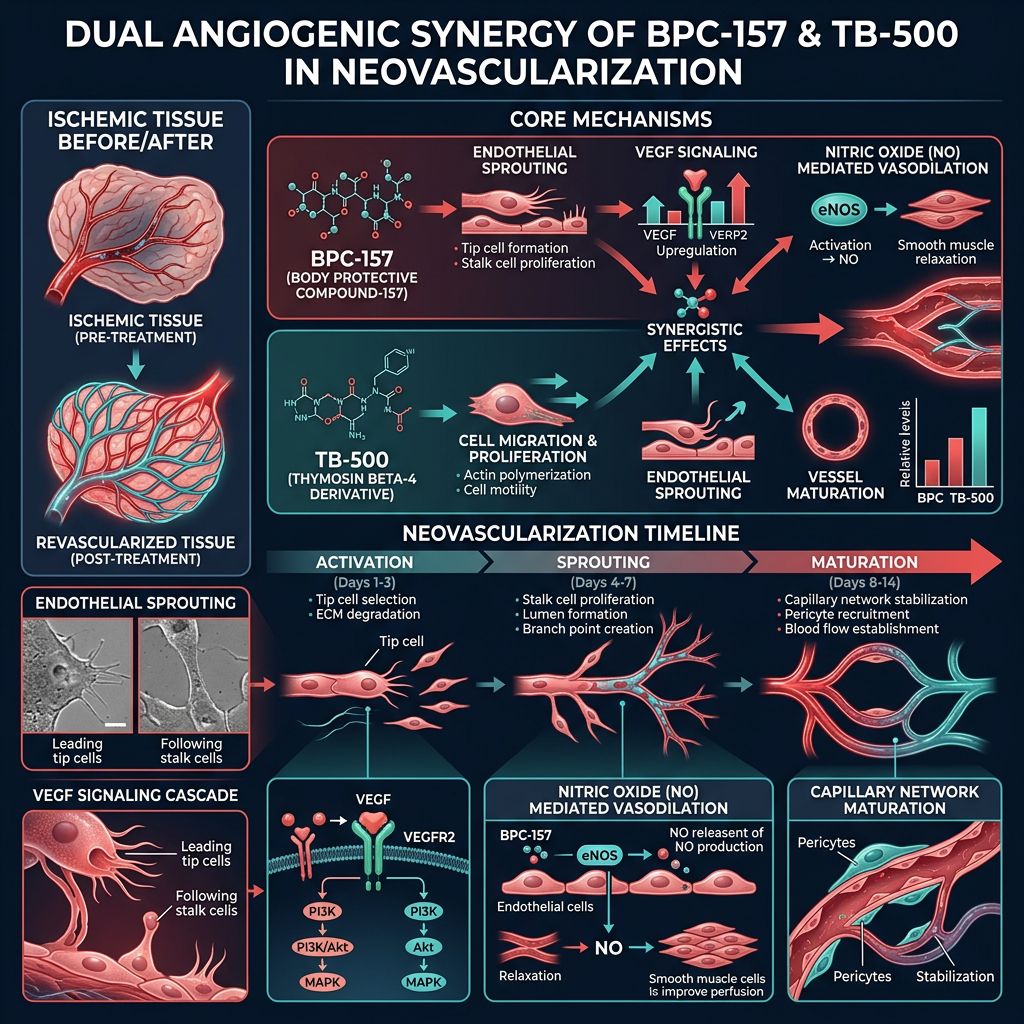 BPC-157 and TB-500 in endothelial sprouting, VEGF signaling, and vessel maturation" style="max-width:100%; height:auto; border-radius:8px;" />
BPC-157 and TB-500 in endothelial sprouting, VEGF signaling, and vessel maturation" style="max-width:100%; height:auto; border-radius:8px;" />Mechanisms of Action: The Dual-Peptide Synergy
BPC-157: The Master of Gastric and Vascular Integrity
BPC-157 (Body Protective Compound-157) is a 15-amino acid pentadecapeptide derived from human gastric juice. Its primary role in research is its "cytoprotective" effect. Unlike many growth factors that have a narrow therapeutic window, BPC-157 has shown the ability to stabilize the brain-gut axis and protect vascular endothelium even under conditions of high oxidative stress. In the context of angiogenesis, BPC-157 acts primarily by upregulating the expression of VEGFR2 (Vascular Endothelial Growth Factor Receptor 2), which is the primary receptor mediating the pro-angiogenic signals of VEGF-A.
TB-500: The Actin-Sequestering Powerhouse
TB-500 is a synthetic version of the naturally occurring peptide Thymosin Beta-4 (Tβ4). Its primary mechanism involves G-actin sequestration. By regulating the polymerization of actin, TB-500 facilitates cellular motility. This is crucial for angiogenesis because endothelial cells must physically migrate into ischemic or damaged zones to form new capillary sprouts. Furthermore, TB-500 is known to induce the production of matrix metalloproteinases (MMPs), enzymes that break down the ECM to clear a path for new vessel growth.
Comparison of Biological Roles
| Feature | BPC-157 | TB-500 (Thymosin Beta-4) |
|---|---|---|
| Primary Function | Upregulation of VEGF receptors/Nitric Oxide signaling | Actin-binding and cell migration |
| Tissue Focus | Ligaments, Tendons, Gastric mucosa, Endothelium | Muscle, Heart, Skin, Cornea |
| Molecular Weight | ~1,419 Da | ~4,963 Da |
| Mechanism of Repair | Activation of EGR-1 and VEGFR2 pathways | G-actin sequestration and MMP upregulation |
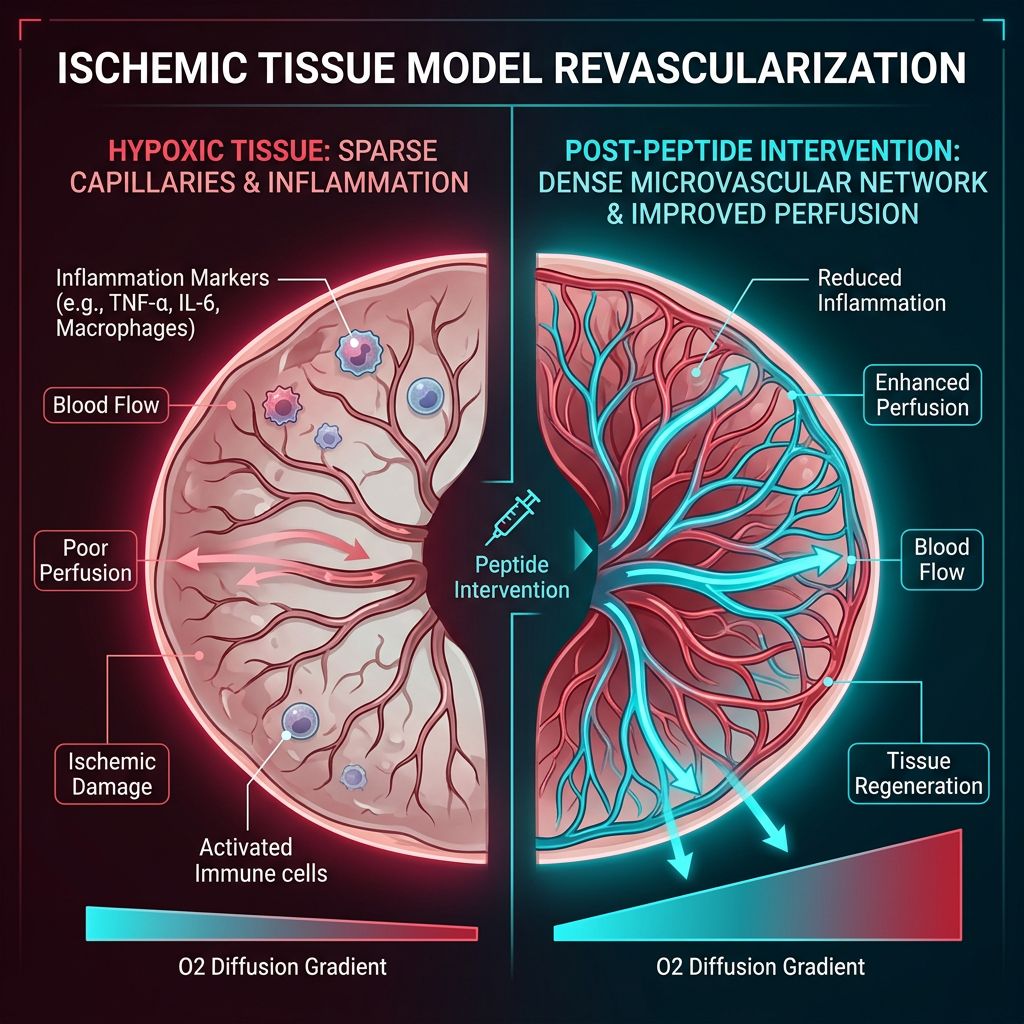
Neovascularization in Ischemic Tissue Models
Ischemia, or the restriction of blood supply to tissues, results in oxygen and nutrient deprivation, leading to cellular necrosis. The "Wolverine Stack" is hypothesized to overcome the "ischemic block" by two distinct but complementary routes. In research settings involving hindlimb ischemia or myocardial infarction models, the BPC-157 + TB-500 Blend has shown a superior ability to restore perfusion compared to monotherapy.
The Nitric Oxide (NO) Pathway
BPC-157 strongly influences the Nitric Oxide (NO) system. It acts as a modulator, ensuring that NO levels remain within a homeostatic range. This leads to vasodilation, which improves immediate blood flow to the damaged area. By maintaining endothelial integrity, BPC-157 prevents the catastrophic "no-reflow" phenomenon often seen after reperfusion of ischemic tissue.
Endothelial Cell Recruitment
While BPC-157 prepares the vascular bed, TB-500 actively recruits endothelial cells to the site of injury. Tβ4 has been observed to stimulate the differentiation of epicardial progenitor cells into endothelial cells and smooth muscle cells. This is particularly relevant in cardiovascular research where the regeneration of the coronary microvasculature is essential for long-term recovery. For complex regenerative needs, researchers often look toward the BPC-157 + TB-500 + GHK-Cu Blend to incorporate the copper-peptide’s ability to remodel collagen while the others drive vascularization.
Advanced Modulation of the Inflammatory Response
A significant hurdle in tissue engineering and repair is the "cytokine storm" or chronic inflammation that follows injury. Excessive inflammation leads to fibrosis (scar tissue) rather than functional regeneration. BPC-157 and TB-500 both exhibit potent anti-inflammatory properties, but through different vectors.
- BPC-157: Inhibits the expression of pro-inflammatory cytokines such as TNF-α and IL-6. It also appears to counteract the systemic inflammatory response syndrome (SIRS) in several animal models.
- TB-500: Reduces the infiltration of inflammatory cells (neutrophils and macrophages) into the site of injury, thereby limiting secondary tissue damage. It also facilitates the transition from the inflammatory phase of healing to the proliferative phase.
For research specifically focused on the inflammatory component of gastrointestinal or systemic repair, the use of KPV in conjunction with BPC-157 is frequently explored, though the BPC-157 + TB-500 combination remains the gold standard for musculoskeletal and vascular applications.
Applications in Musculoskeletal Repair
The "Wolverine Stack" earned its name primarily through its dramatic effects on "hypovascular" tissues—tissues like tendons and ligaments that naturally receive very little blood flow. These tissues possess a poor intrinsic healing capacity because nutrients cannot reach the site of injury efficiently.
Tendon-to-Bone Healing
In studies of Achilles tendon ruptures, BPC-157 has been found to promote the outgrowth of tendon fibroblasts and increase the expression of growth hormone receptors on these cells. When combined with the migratory stimulus of TB-500, researchers observe faster collagen deposition and more organized fiber alignment. High-purity peptides are essential for these studies to ensure that observed outcomes are not skewed by impurities; researchers can verify these standards through our Quality Control processes and COA Documents.
Muscle Regeneration
Muscle tears often lead to the formation of scar tissue, which reduces the contractile strength of the muscle. TB-500's role in G-actin regulation ensures that satellite cells (muscle stem cells) can migrate to the site of the tear and fuse into new myofibers. BPC-157 supports this by ensuring the capillary density in the regenerating muscle is sufficient to handle the metabolic demands of the new tissue.
Integrating Metabolic and Mitochondrial Support
While the angiogenic signaling provided by BPC-157 and TB-500 is robust, the metabolic demand of building new tissue is immense. To enhance the research outcomes of the "Wolverine Stack," many scientists integrate mitochondrial peptides. For example, MOTS-c or SS-31 can be used to optimize mitochondrial bioenergetics, providing the ATP necessary for the cellular migration and protein synthesis driven by TB-500 and BPC-157.
Similarly, the use of NAD+ precursors or direct NAD+ research has shown promise in maintaining the youthful epigenetic state of endothelial cells, making them more responsive to the cues provided by TB-500. This multi-layered approach mimics the complex biological environment required for true "Wolverine-like" regeneration.
Research Design and Experimental Considerations
When designing research protocols involving these peptides, the concentration and dosing frequency are paramount. Most research models utilize a subcutaneous or intramuscular administration route to ensure systemic distribution or localized concentration respectively. The stability of these peptides is also a factor; for information on the chemical stability and manufacturing standards, visit our Peptide Synthesis page.
Synergistic Combinations Table
| Combination | Primary Research Objective |
|---|---|
| BPC-157 + TB-500 | Musculoskeletal repair and neovascularization |
| BPC-157 + TB-500 + GHK-Cu | Dermal repair and remodeling of fibrotic tissue |
| BPC-157 + Ipamorelin | GH-mediated tissue growth and structural repair |
| TB-500 + ARA-290 | Neuropathic repair and vascular protection |
Emerging Research: Neuroprotection and the Blood-Brain Barrier
Newer studies suggest that the angiogenic properties of the Wolverine Stack extend to the central nervous system. Ischemic stroke models have shown that BPC-157 can protect the blood-brain barrier (BBB) and reduce cerebral edema. When combined with the neuro-migratory effects of TB-500, there is potential for stimulating neurovascular units. Researchers interested in cognitive and neurological repair may also consider investigating Cerebrolysin or Dihexa, which operate through different neurotrophic pathways but share the goal of structural restoration.
Safety, Purity, and Laboratory Standards
The efficacy of angiogenic research is highly dependent on peptide purity. The presence of trifluoroacetic acid (TFA) salts, bacterial endotoxins, or truncated peptide sequences can trigger non-specific inflammatory responses that confound histological data. Alpha Carbon Labs provides researchers with the high-purity sequences necessary for precise experimentation. The use of BPC-157 + TB-500 Blend from a verified source ensures that the ratio of peptides is consistent, allowing for reproducible data across different study cohorts.
Conclusion: The Future of Revascularization
The "Wolverine Stack" represents more than just a combination of two popular research compounds. It represents a sophisticated approach to biological engineering where one peptide (BPC-157) stabilizes the vascular infrastructure and receptor sensitivity, while the other (TB-500) drives the cellular mechanics required for tissue expansion. As we move closer to solving the riddles of organ regeneration and chronic wound healing, these peptides will undoubtedly remain at the forefront of angiogenic signaling research.
For researchers seeking to explore these dynamics in their own laboratory environments, choosing the right tools is the first step toward discovery. Explore our full range of research-grade peptides and technical documentation to support your next breakthrough in regenerative science.
References
- 1. Sikiric, P., et al. (2010). Focus on ulcerative colitis: stable gastric pentadecapeptide BPC 157. Current Pharmaceutical Design.
- 2. Hasan, L., et al. (2021). The pleiotropic effects of Thymosin beta 4 in angiogenesis and tissue regeneration. Frontiers in Physiology.
- 3. Chang, C. H., et al. (2014). The promoting effect of pentadecapeptide BPC 157 on tendon-to-bone healing. Journal of Applied Physiology.
- 4. Malekmakan, L., et al. (2022). Thymosin Beta-4: A potential therapeutic target for cardiovascular diseases. Journal of Cardiovascular Development and Disease.
- 5. Seiwerth, S., et al. (2018). BPC 157 and Blood Vessels. Current Pharmaceutical Design.
- 6. Philp, D., & Kleinman, H. K. (2010). Thymosin β4: A multifunctional regenerative peptide. Vitamins and Hormones.
- 7. Huang, T., et al. (2015). Body protective compound-157 enhances alkali-burn wound healing in vivo. Burns.
- 8. Brcic, L., et al. (2009). Modulatory effect of gastric pentadecapeptide BPC 157 on angiogenesis in muscle and tendon. Journal of Physiology and Pharmacology.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.