Actin Sequestration and GH Flux: Evaluating the Parallel Mechanisms of TB-500 and Hexarelin in Healing Research
This article provides a rigorous mechanistic comparison of TB-500 and Hexarelin, exploring the differences between actin-driven tissue remodeling and GH-driven anabolic signaling in high-load vs. low-load research protocols.
Introduction: The Dual Axis of Tissue Regeneration
In the landscape of regenerative medicine and peptide therapeutics, the mechanisms governing tissue repair are often bifurcated into two distinct categories: structural reorganization and metabolic amplification. The former involves the physical movement of cells and the reconstruction of the extracellular matrix (ECM), while the latter dictates the energy availability and anabolic signaling required to fuel these processes. When evaluating peptide protocols for accelerated healing in research settings, distinguishing between these pathways is crucial for optimizing experimental outcomes.
This article provides a rigorous mechanistic comparison of two pivotal peptides that exemplify these distinct yet complementary pathways: TB-500 (synthetic Thymosin Beta-4) and Hexarelin. While TB-500 operates primarily through actin sequestration—essentially controlling the cellular mechanics of migration and cytoskeletal remodeling—Hexarelin functions through the modulation of Growth Hormone (GH) flux and direct interaction with cardiac receptors.
We will explore the physiological nuances of high-load versus low-load administration protocols in animal models, analyzing how researchers manipulate the "Actin-GH Axis" to influence healing trajectories in musculoskeletal, cardiac, and neurological tissues.
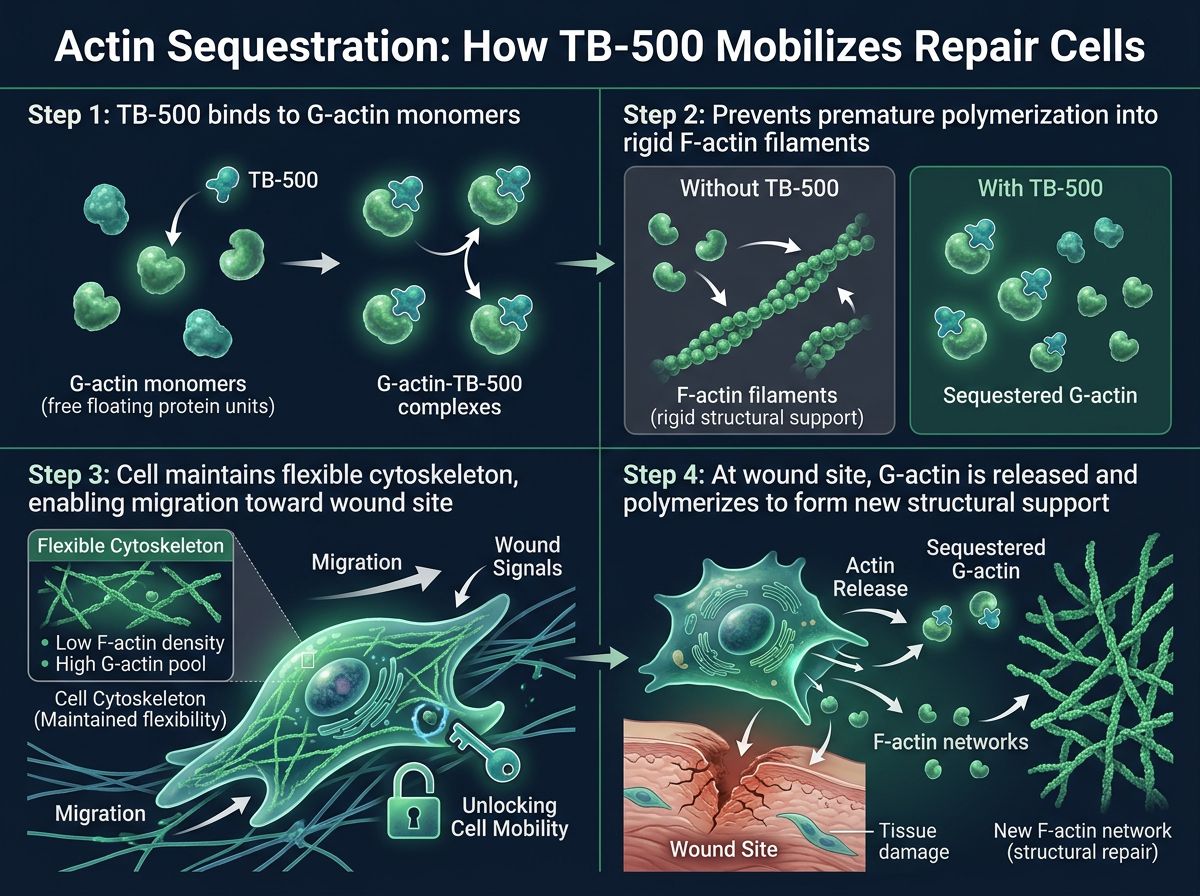
Part I: TB-500 and the Mechanics of Actin Sequestration
The Biological Imperative of Cell Motility
To understand the efficacy of TB-500 in healing models, one must first appreciate the role of the cytoskeleton. In any injury model, whether it be a skeletal muscle tear or a myocardial infarction, the immediate requirement for repair is the migration of reparative cells—macrophages, fibroblasts, and endothelial cells—to the site of injury. If these cells cannot move efficiently, healing is stalled.
Cellular motility relies heavily on actin, a globular protein that polymerizes to form filaments. The transition between Globular actin (G-actin, the monomer) and Filamentous actin (F-actin, the polymer) creates the propulsive force that drives cell movement. However, uncontrolled polymerization can lead to rigid, immobile cellular structures.
Mechanism of Action: The Thymosin Beta-4 Analog
TB-500 is a synthetic fraction of the naturally occurring protein Thymosin Beta-4. Its primary mechanism of action is actin sequestration. By binding to G-actin monomers, TB-500 maintains a large pool of non-polymerized actin within the cell. This might seem counterintuitive—preventing polymerization—but it is actually a regulatory necessity.
By sequestering G-actin, TB-500 prevents spontaneous, chaotic polymerization. When the cell receives a signal to migrate toward a wound, TB-500 releases the G-actin locally, allowing for rapid, directional polymerization. This effectively "turbocharges" the cell's ability to crawl through the extracellular matrix to reach damaged tissue.
Angiogenesis and Vascular Remodeling
Beyond cytoskeletal restructuring, TB-500 has been identified as a potent pro-angiogenic factor. Research indicates that it promotes the expression of Vascular Endothelial Growth Factor (VEGF) and facilitates the migration of endothelial cells to form new capillaries. In ischemic injury models, such as cardiac ischemia, the presence of TB-500 has been linked to the preservation of cardiomyocytes and the collateralization of blood vessels, reducing the total area of infarct.
Part II: Hexarelin and the Modulation of GH Flux
Beyond Standard Secretagogues
While TB-500 manages the structural logistics of repair, Hexarelin addresses the anabolic signaling required for protein synthesis. Hexarelin is a synthetic hexapeptide belonging to the growth hormone secretagogue (GHS) class. Unlike Growth Hormone Releasing Hormone (GHRH) analogs like CJC-1295 or Sermorelin, Hexarelin mimics the action of ghrelin, functioning as a selective agonist of the GHS-R1a receptor.
The Physiology of GH Flux
The term "GH Flux" refers to the pulsatile amplitude and frequency of Growth Hormone release from the anterior pituitary. In research subjects, particularly aged models, GH flux is naturally diminished, slowing the rate of protein synthesis and lipolysis. Hexarelin is noted for its ability to induce massive, acute spikes in plasma GH levels, regardless of endogenous somatostatin tone.
This surge in GH triggers the hepatic release of Insulin-like Growth Factor 1 (IGF-1), which is the primary mediator of anabolic effects in peripheral tissues. Elevated IGF-1 signaling accelerates the transcription of proteins necessary for muscle hypertrophy, collagen synthesis, and bone density accretion.
The CD36 Receptor: A Unique Mechanism
What sets Hexarelin apart from other secretagogues like Ipamorelin or GHRP-2 is its high affinity for the CD36 receptor (Cluster of Differentiation 36), also known as the fatty acid translocase. This interaction is distinct from the GH axis.
In cardiac research, CD36 activation by Hexarelin has been shown to exert direct cardioprotective effects, reducing apoptosis in cardiomyocytes and inhibiting fibrosis following reperfusion injury. This makes Hexarelin a dual-threat peptide: it drives systemic anabolism via GH/IGF-1 while stimulating local tissue survival pathways in the heart via CD36.
Part III: Divergent Pathways, Convergent Goals
To visualize the separation of duties between these two peptides, consider the different requirements of healing a tendon injury vs. reversing cardiac damage. The following table summarizes their primary and secondary mechanisms.
| Feature | TB-500 (Actin Sequestration) | Hexarelin (GH Flux/CD36) |
|---|---|---|
| Primary Mechanism | Binds G-actin to regulate polymerization and cell motility. | Agonizes GHS-R1a to stimulate high-amplitude GH release. |
| Secondary Mechanism | Upregulates VEGF; modulates inflammation via NF-kB downregulation. | Binds CD36 receptor; promotes cardiac cell survival and anti-fibrosis. |
| Metabolic Effect | Neutral; focuses on cytoskeletal dynamics. | Anabolic; promotes lipolysis and protein synthesis via IGF-1. |
| Key Tissues | Endothelium, Myocytes, Tenocytes, Cornea. | Pituitary, Myocardium, Skeletal Muscle, Adipose Tissue. |
| Half-Life Influence | Longer systemic effects; accumulates at injury sites. | Short half-life; relies on acute signaling pulses. |
Part IV: Analyzing High-Load vs. Low-Load Protocols
In the scientific literature involving animal models, dosing protocols are rarely static. Researchers typically employ "High-Load" (loading phase) or "Low-Load" (maintenance) strategies depending on whether the objective is acute trauma repair or chronic tissue support. The behavior of TB-500 and Hexarelin differs significantly under these varying loads.
TB-500: The Saturation Model
Research into Thymosin Beta-4 suggests that its concentration in plasma acts as a reservoir. Because TB-500 does not rely on a pulsatile receptor trigger (like GHS-R1a), but rather on steady-state availability for actin binding, "High-Load" protocols are often utilized in the acute phase of injury research.
- High-Load Rationale: In the first 7-14 days post-injury, the demand for actin monomers is highest as cell recruitment is active. Studies employ frequent administration to saturate the local environment, ensuring that actin availability is never the rate-limiting step for repair.
- Maintenance Rationale: Once the remodeling phase is established (angiogenesis is underway), the requirement for massive cell migration drops. Lower maintenance doses are used in long-term studies to support tissue maturation without wasting substrate.
Hexarelin: The Desensitization Challenge
Hexarelin usage in research is governed by the principle of receptor desensitization. Unlike the relatively linear dose-response of TB-500, the GHS-R1a receptor can downregulate rapidly in the presence of continuous or excessive agonist exposure.
- High-Load Rationale: High-dose or high-frequency Hexarelin administration is effective only for very short durations in research (e.g., acute cardiac events). The goal is to maximize the immediate ejection of GH stores and activate CD36 receptors during a critical window of ischemia.
- Cycling Requirement: Prolonged exposure to Hexarelin without a "washout" period leads to attenuated GH release. Therefore, long-term healing protocols involving Hexarelin often utilize intermittent dosing schedules (e.g., 2 days on, 1 day off) or are cycled with lower-affinity secretagogues.
Part V: Specific Therapeutic Contexts in Research
1. Cardiac Fibrosis and Remodeling
The synergy between actin sequestration and GH flux is perhaps most critical in cardiac research. Following a myocardial infarction, the heart undergoes "remodeling," a euphemism for the replacement of contractile tissue with non-functional scar tissue (fibrosis).
TB-500's Role: It promotes the survival of "sleeping" or stunned cardiomyocytes and encourages the growth of new blood vessels into the ischemic zone. By modulating actin, it helps preserve the structural integrity of the ventricular wall.
Hexarelin's Role: Through the CD36 pathway, Hexarelin actively combats the formation of atherosclerotic plaques and reduces the inflammatory cytokine storm that precipitates fibrosis. Furthermore, the increase in IGF-1 supports the contractile strength of the remaining heart muscle.
2. Tendinopathy and Ligament Repair
Tendons are notoriously slow to heal due to poor vascularity. This presents a transport problem for nutrients and a migration problem for reparative cells.
The Combined Hypothesis: Researchers often investigate the co-administration of agents like BPC-157 + TB-500 Blend alongside growth factors. In the specific context of Hexarelin, the mechanism provides the systemic anabolic drive (collagen synthesis) while TB-500 ensures that tenocytes (tendon cells) can migrate into the tear to deposit that collagen. Without TB-500, the cells may not reach the injury core; without Hexarelin/GH, the cells may lack the metabolic drive to synthesize sufficient matrix.
3. Neurological Regeneration
While often overshadowed by peptides like Semax or Cerebrolysin, both TB-500 and Hexarelin show promise in neuroprotection.
TB-500 has been shown to improve oligodendrocyte differentiation—critical for remyelination of nerves. Hexarelin offers neuroprotection through ghrelin-receptor pathways that inhibit neuronal apoptosis and reduce oxidative stress in the brain. The contrasting mechanisms offer a multi-faceted approach to traumatic brain injury (TBI) models: structural repair of the neural sheath (TB-500) combined with metabolic support for neuronal firing (Hexarelin).
Part VI: Safety, Purity, and Synthesis Considerations
When conducting research with potent biological signaling agents, the purity of the peptide substrate is paramount. Impurities in peptide synthesis, such as trifluoroacetic acid (TFA) salts or incomplete peptide chains, can induce immunogenic responses that confound research data.
Specifically for Hexarelin, stereochemical purity is vital to ensure proper locking into the GHS-R1a and CD36 receptors. For TB-500, oxidation can alter its actin-binding affinity. Alpha Carbon Labs maintains rigorous standards for peptide synthesis, utilizing High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry to verify sequence and purity data. Researchers should always consult COA documents prior to initiating any subject trials to ensure the integrity of the mechanistic variables being tested.
Potential Adverse Mechanisms
While generally well-tolerated in animal models, high-flux GH protocols (Hexarelin) carry theoretical risks of insulin resistance if maintained indefinitely without breaks, due to GH's antagonism of insulin action. Conversely, while TB-500 promotes angiogenesis, this mechanism theoretically warrants caution in subjects with proliferative retinopathies or active neoplasms, where vessel growth is contraindicated.
 BPC-157, TB-500, and Hexarelin covering blood flow, migration, and material supply" style="max-width: 100%; border-radius: 8px;" />
BPC-157, TB-500, and Hexarelin covering blood flow, migration, and material supply" style="max-width: 100%; border-radius: 8px;" />Part VII: Theoretical Synergies with Other Agents
The research landscape frequently explores "stacks" or combinations to cover multiple signaling pathways.
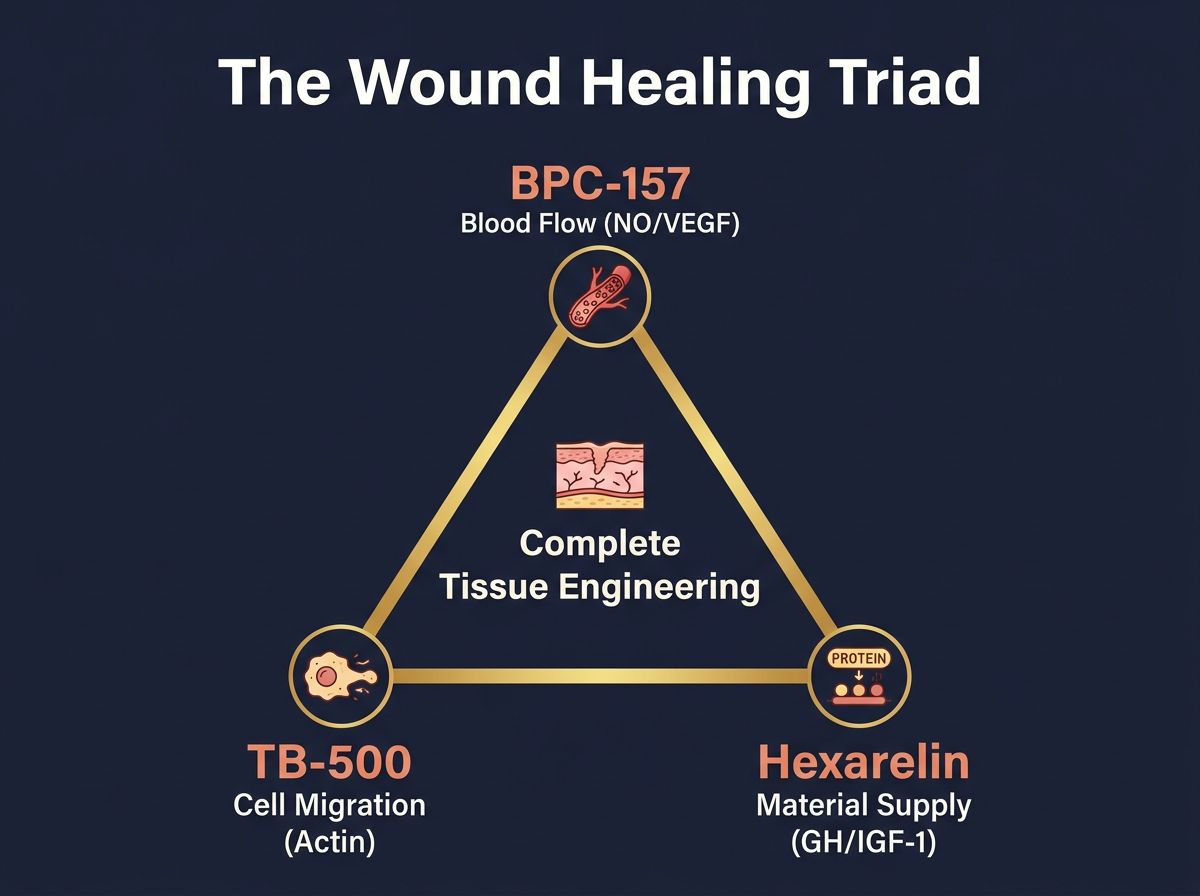
The "Wound Healing Triad"
A common theoretical framework in literature involves the combination of:
- BPC-157: For acute nitric oxide modulation and gastric/tendon protection.
- TB-500: For actin-mediated cell migration and vascular growth.
- High-Flux GH Secretagogue (Hexarelin): For maximal protein accretion.
This triad addresses the three pillars of tissue engineering: blood flow, cell presence, and material synthesis.
The "Metabolic/Mitochondrial" Axis
For research focusing on energetic capacity and muscle recovery, Hexarelin is sometimes studied alongside MOTS-c or SS-31. While Hexarelin drives the hormonal signal for growth, mitochondrial peptides improve the cellular energy (ATP) efficiency required to execute that growth, suggesting a potential for synergistic enhancement of endurance and recovery parameters.
Conclusion
The distinction between actin sequestration and GH flux represents a fundamental divide in regenerative biology: the difference between moving the workers to the construction site and supplying them with the energy to build. TB-500 acts as the logistical manager, ensuring that G-actin is available for immediate cell mobilization and cytoskeletal reorganization. Hexarelin acts as the systemic signal amplifier, utilizing the GH/IGF-1 axis and CD36 pathways to drive anabolic retention and cardioprotection.
Current research regarding high-load versus low-load protocols suggests that while TB-500 benefits from saturation strategies during acute injury phases, Hexarelin requires careful pulsatile administration to avoid receptor downregulation. By understanding these parallel yet distinct mechanisms, researchers can design more sophisticated experiments that target specific failures in the healing cascade, moving beyond generic "growth" protocols toward precision regenerative medicine.
Alpha Carbon Labs is dedicated to supplying the highest purity research peptides for the advancement of biological science. Our commitment to quality control ensures that your data is derived from accurate, verified chemical pathways.
References
- 1. Goldstein, A. L., et al. "Thymosin β4: actin-sequestering protein tunes cell cytoskeletal reorganization." Trends in Biochemical Sciences 30.8 (2005): 444-449.
- 2. Mao, Y., et al. "Hexarelin treatment prevents cardiac remodeling and dysfunction in spontaneously hypertensive rats." European Journal of Pharmacology 515.1-3 (2005): 124-130.
- 3. Philp, D., et al. "Thymosin β4 promotes angiogenesis, wound healing, and hair follicle development." Mechanisms of Development 120.1 (2003): 77-85.
- 4. Korbonits, M., et al. "Ghrelin—a hormone with multiple functions." Frontiers in Neuroendocrinology 25.1 (2004): 27-68.
- 5. Sosa, I., et al. "Thymosin beta 4 is a pivotal modulator of focal adhesion formation and migration in endothelial cells." Journal of Cellular Physiology 204.3 (2005): 937-947.
- 6. Tanda, M. L., et al. "Growth hormone secretagogues and the cardiac tissue: an update." Peptides 28.7 (2007): 1461-1467.
- 7. Crockford, D., et al. "Thymosin beta4: structure, function, and biological properties supporting current and future clinical applications." Annals of the New York Academy of Sciences 1194.1 (2010): 179-189.
- 8. Sigala, S., et al. "Evidence for the presence of receptors for growth hormone secretagogues on human endothelial cells." Journal of Endocrinological Investigation 32.11 (2009): 908-912.
- 9. Smart, N., et al. "Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization." Nature 445.7124 (2007): 177-182.
- 10. Bodart, V., et al. "Identification of a new growth hormone-releasing peptide receptor in the heart." Circulation Research 87.8 (2000): 703-709.
- 11. Huff, T., et al. "β-Thymosins, small acidic peptides with multiple functions." The International Journal of Biochemistry & Cell Biology 33.3 (2001): 205-220.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.