Semaglutide vs. Retatrutide: Comparative Efficacy in Mediating Adipose Tissue Plasticity
An in-depth scientific comparison of Semaglutide and Retatrutide, focusing on their distinct mechanisms in mediating adipose tissue plasticity and thermogenesis.
Introduction to Incretin Mimetics and Adipose Tissue Plasticity
In the evolving landscape of metabolic research, the transition from mono-agonism to multi-receptor poly-pharmacology represents a paradigm shift in how we approach adiposity and systemic metabolic dysfunction. Adipose tissue is no longer viewed merely as a passive storage site for triglycerides, but as a dynamic endocrine organ capable of significant "plasticity"—the ability to remodel its structure, metabolic rate, and signaling profile in response to hormonal stimuli.
Two of the most prominent molecules in current metabolic research are Semaglutide, a potent Glucagon-Like Peptide-1 (GLP-1) receptor agonist, and Retatrutide, a pioneering triple agonist targeting GLP-1, Glucose-Dependent Insulinotropic Polypeptide (GIP), and Glucagon (GCG) receptors. This article explores the comparative efficacy of these peptides in mediating adipose tissue remodeling and their broader implications for researchers studying metabolic syndrome.
Semaglutide: The Gold Standard of Selective GLP-1 Agonism
Semaglutide has long served as the benchmark for incretin-based therapy. Its primary mechanism of action involves the activation of GLP-1 receptors in the pancreas (potentiating glucose-dependent insulin secretion) and the hypothalamus (enhancing satiety and reducing caloric intake).
Adipose-Specific Effects of Semaglutide
While much of the weight loss associated with Semaglutide is attributed to central nervous system (CNS) mediated appetite suppression, research indicates significant direct and indirect effects on adipose tissue:
- Inhibition of Adipogenesis: GLP-1 signaling may inhibit the differentiation of pre-adipocytes into mature, lipid-storing adipocytes.
- Reduction of Pro-inflammatory Cytokines: Semaglutide has been shown to decrease markers like TNF-alpha and IL-6 within visceral fat deposits, thereby improving systemic insulin sensitivity.
- Lipolysis Modulation: While GLP-1 is not a primary lipolytic driver, the resulting negative energy balance facilitates the mobilization of free fatty acids from white adipose tissue (WAT).
For more details on the manufacturing of these high-purity peptides, researchers are encouraged to review our peptide synthesis protocols and quality control standards.
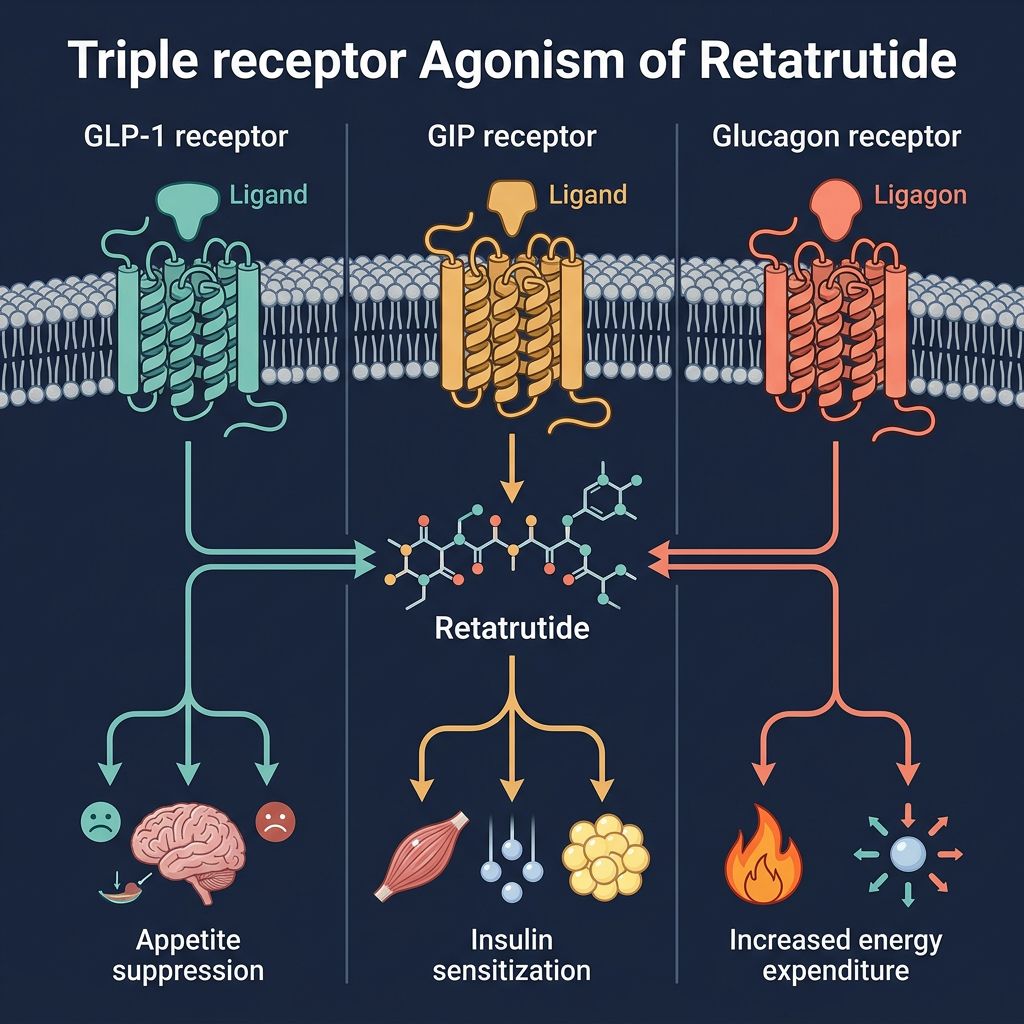
Retatrutide: The Triple Agonist Revolutionary
Retatrutide (LY3437943) introduces a more complex pharmacological profile. By simultaneously activating three distinct pathways, it seeks to overcome the biological plateaus often observed with mono-therapy. The synergy between GLP-1, GIP, and Glucagon receptors targets adipose tissue plasticity through multiple orthogonal axes.
The Role of GIP in Lipid Storage
The GIP receptor (GIPR) is highly expressed in adipose tissue. Unlike GLP-1, GIP is thought to play a direct role in adipose tissue "buffering," improving the health of fat cells so they can store energy safely without spilling over into ectopic sites like the liver or skeletal muscle. In the presence of GLP-1 agonism, GIPR activity appears to enhance the metabolic efficiency of fat depots.
The Glucagon Component: Driving Thermogenesis
Perhaps the most significant differentiator for Retatrutide is the inclusion of Glucagon Receptor (GCGR) agonism. Traditionally viewed solely as a hyperglycemic hormone, glucagon is now recognized as a potent stimulant of energy expenditure. It promotes "browning" of white adipose tissue and increases mitochondrial uncoupling in brown adipose tissue (BAT).
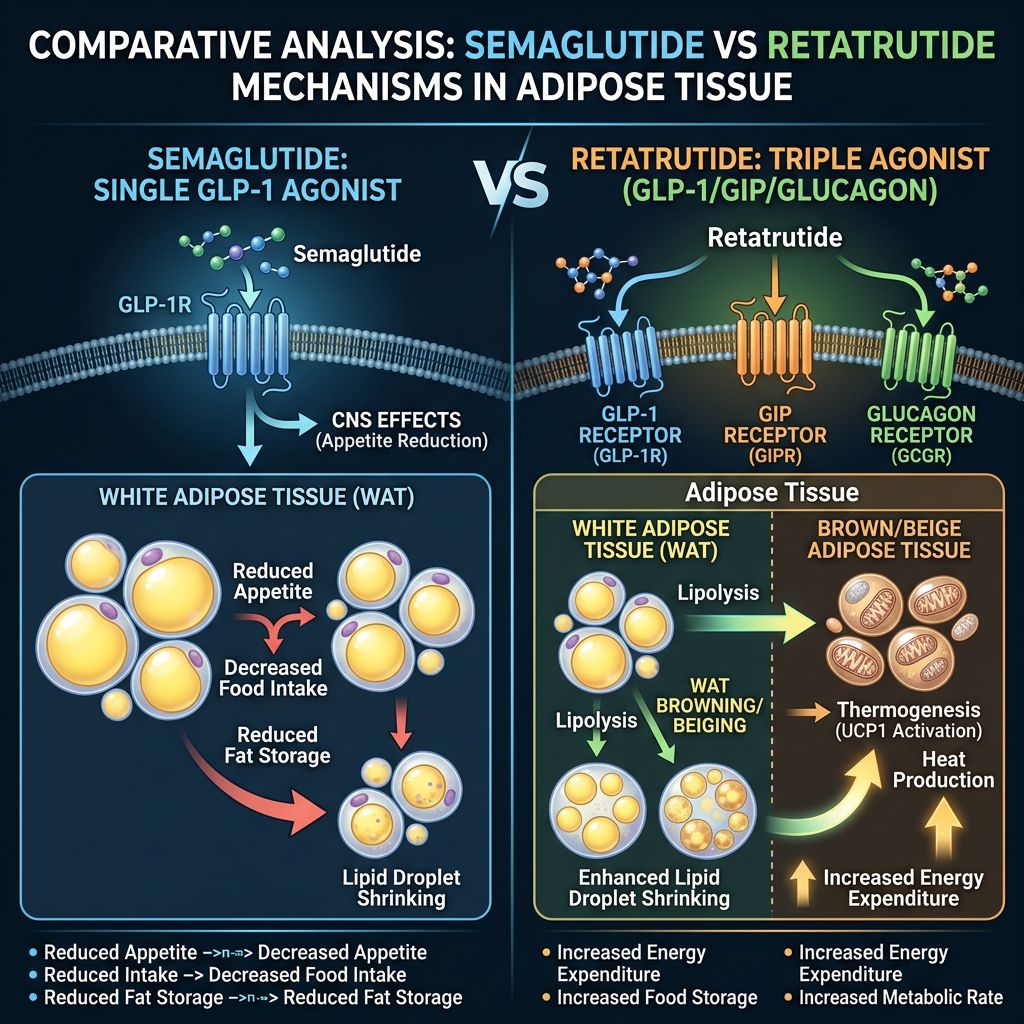
Comparative Analysis: Mechanistic Divergence
The following table summarizes the physiological differences in how these two peptides approach metabolic regulation:
| Feature | Semaglutide | Retatrutide |
|---|---|---|
| Receptor Targets | Selective GLP-1R | GLP-1R / GIPR / GCGR |
| Primary Weight Loss Driver | Appetite Suppression (CNS) | Appetite Suppression + Increased Basal Metabolic Rate |
| Adipose Tissue Plasticity | Mainly through caloric deficit | Direct thermogenic "browning" via GCGR |
| Hepatic Fat Reduction | Significant (Indirect) | Highly Potent (Direct GCGR signaling) |
| Glucagon Response | Suppressed | Mimicked (Agonism) |
Efficacy in Clinical and Pre-clinical Models
Data from the STEP clinical trials for Semaglutide showed an average weight loss of approximately 14.9% to 17.4% over 68 weeks. In contrast, phase 2 data for Retatrutide demonstrated mean weight reductions of up to 24.2% at only 48 weeks, suggesting a significantly faster and more profound impact on total body adiposity.
Mitochondrial Health and Cellular Efficiency
Researchers investigating cellular metabolism often combine these studies with mitochondrial-focused peptides. For instance, SS-31 and MOTS-c are frequently used alongside incretin mimetics to study how protecting mitochondrial integrity can prevent the metabolic "crash" often associated with rapid weight loss. Retatrutide’s ability to stimulate glucagon-mediated hepatic lipid oxidation makes it a particularly interesting candidate for co-administration studies with mitochondrial peptides.
Beyond Adiposity: Cardiovascular and Renal Plasticity
Metabolic health is inextricably linked to vascular and renal function. Semaglutide has established a strong record for cardiovascular risk reduction (MACE). Retatrutide, while newer, is being scrutinized for its effects on heart rate (due to GCGR and GIPR activity) and its potentially superior ability to clear intra-organ fat, which could have protective effects on renal health similar to ARA-290 in inflammatory models.
Synergistic Research Applications
In research environments focused on body composition, authors often look at peptides that protect lean mass while maximizing fat loss. While Semaglutide and Retatrutide are masters of fat mass reduction, they are sometimes studied in conjunction with growth hormone secretagogues like Ipamorelin or Tesamorelin to observe the preservation of skeletal muscle during rapid weight descent.
Furthermore, for studies involving "stubborn" localized fat deposits or the transition of WAT to BAT, researchers may look at AOD9604 or 5-amino-1mq as adjunctive tools to enhance the lipolytic effects seen with triple agonism.
Current Research Landscape: Mazdutide and Cagrilintide
The field is moving fast. Beyond the Semaglutide vs. Retatrutide debate, researchers are now looking at dual agonists like Mazdutide (GLP-1/GCG) and long-acting amylin analogues like Cagrilintide. The goal remains the same: achieving "metabolic flexibility," where the body can efficiently switch between fuel sources while maintaining low levels of systemic inflammation.
Safety and Tolerability in Research Protocols
Both peptides exhibit a classic "incretin" side effect profile, primarily gastrointestinal (nausea, vomiting, delayed gastric emptying). However, Retatrutide’s triple agonism requires careful monitoring of heart rate and glycemic variability. Researchers must ensure they are using high-quality materials; Alpha Carbon Labs provides comprehensive COA documents for all batches to ensure experimental reproducibility.
Conclusion
Semaglutide remains the cornerstone of GLP-1 research, offering a proven, safe, and effective pathway for reducing adiposity through caloric regulation. However, Retatrutide represents the "next frontier," leveraging the metabolic firepower of Glucagon and GIP to directly alter the plasticity and thermogenic capacity of adipose tissue. For the researcher, the choice between these two molecules depends on whether the goal is pure GLP-1 receptor mapping or the exploration of complex, multi-organ metabolic synergy.
As the primary source for research-grade peptides, Alpha Carbon Labs supports these investigations by providing the highest purity Semaglutide, Retatrutide, and Tirzepatide for laboratory use.
References
- 1. Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002.
- 2. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. N Engl J Med. 2023;389(6):514-526.
- 3. Knerr PJ, et al. Next generation GLP-1, GIP, and glucagon receptor agonists: Emerging therapeutics for obesity and type 2 diabetes. Peptides. 2022;157:170884.
- 4. Müller TD, et al. Glucagon-like peptide 1 (GLP-1). Mol Metab. 2019;30:72-130.
- 5. Heise T, et al. Tirzepatide versus semaglutide 2.0 mg in patients with type 2 diabetes: a randomized, double-blind phase 2 study (SURPASS-6). Lancet Diabetes Endocrinol. 2023.
- 6. Evers A, et al. Glucagon receptor signaling: a fatty acid based approach for the treatment of metabolic diseases. J Lipid Res. 2017;58(5):1018-1029.
- 7. Cersosimo E, et al. The role of glucagon in health and disease. In: Endotext [Internet]. 2019.
- 8. Finan B, et al. A novel trimodal co-agonist of GLP-1, GIP, and glucagon receptors for metabolic disease. Nat Med. 2015;21(1):27-36.
- 9. Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes. Mol Metab. 2018;18:3-14.
- 10. Sun B, et al. Discovery and characterization of retatrutide, a triple GIP, GLP-1, and glucagon receptor agonist. Cell Metab. 2023.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.