Quantifying Metabolic Velocity: Tirzepatide’s Role in Adipose Tissue Remodeling Research
Explore the advanced biochemistry of Tirzepatide research, focusing on its dual GIP/GLP-1 receptor agonism and its impact on adipose tissue remodeling and metabolic velocity in murine models.
Introduction to Metabolic Velocity and Dual-Incretin Agonism
In the landscape of metabolic research, the concept of "metabolic velocity" describes the rate at which an organism can transition between metabolic states while maintaining homeostatic control over glucose and lipid flux. For decades, researchers focused on single-pathway interventions, primarily targeting the Glucagon-like Peptide-1 (GLP-1) receptor. However, the emergence of dual-agonist polypeptides like Tirzepatide has shifted the scientific paradigm toward multi-receptor integration, specifically the synergy between GLP-1 and Glucose-dependent Insulinotropic Polypeptide (GIP).
Tirzepatide represents a "twincretin" architecture—a 39-amino acid synthetic peptide based on the native GIP sequence, modified with a C20 fatty diacid moiety to extend its half-life. While traditional research molecules like Semaglutide strictly target the GLP-1 receptor, Tirzepatide introduces a biased signaling approach that prioritizes GIP receptor activation over GLP-1 (potency ratio of approximately 3:1 in favor of GIP). This distinction is critical in murine and clinical models for understanding how adipose tissue remodeling occurs beyond simple caloric restriction.
The Molecular Architecture: Why GIP Integration Matters
The biochemistry of Tirzepatide is rooted in its ability to mimic the structural properties of both GIP and GLP-1. In peptide synthesis, the inclusion of aminoisobutyric acid (Aib) residues at positions 2 and 13 provides significant resistance to Dipeptidyl Peptidase-4 (DPP-4) degradation. Understanding this synthetic stability is paramount for researchers calculating dosing intervals in chronic metabolic studies.
GIP Receptor (GIPR): The Metabolic Gatekeeper
For years, GIPR was considered a "pro-lipogenic" receptor, with some researchers arguing that its inhibition might prevent obesity. However, recent data suggests that GIPR agonism in the central nervous system (CNS) and white adipose tissue (WAT) actually enhances insulin sensitivity and facilitates healthy lipid storage, preventing ectopic fat deposition in the liver or skeletal muscle. This process, often referred to as "adipose tissue remodeling," is where Tirzepatide diverges from its predecessors.
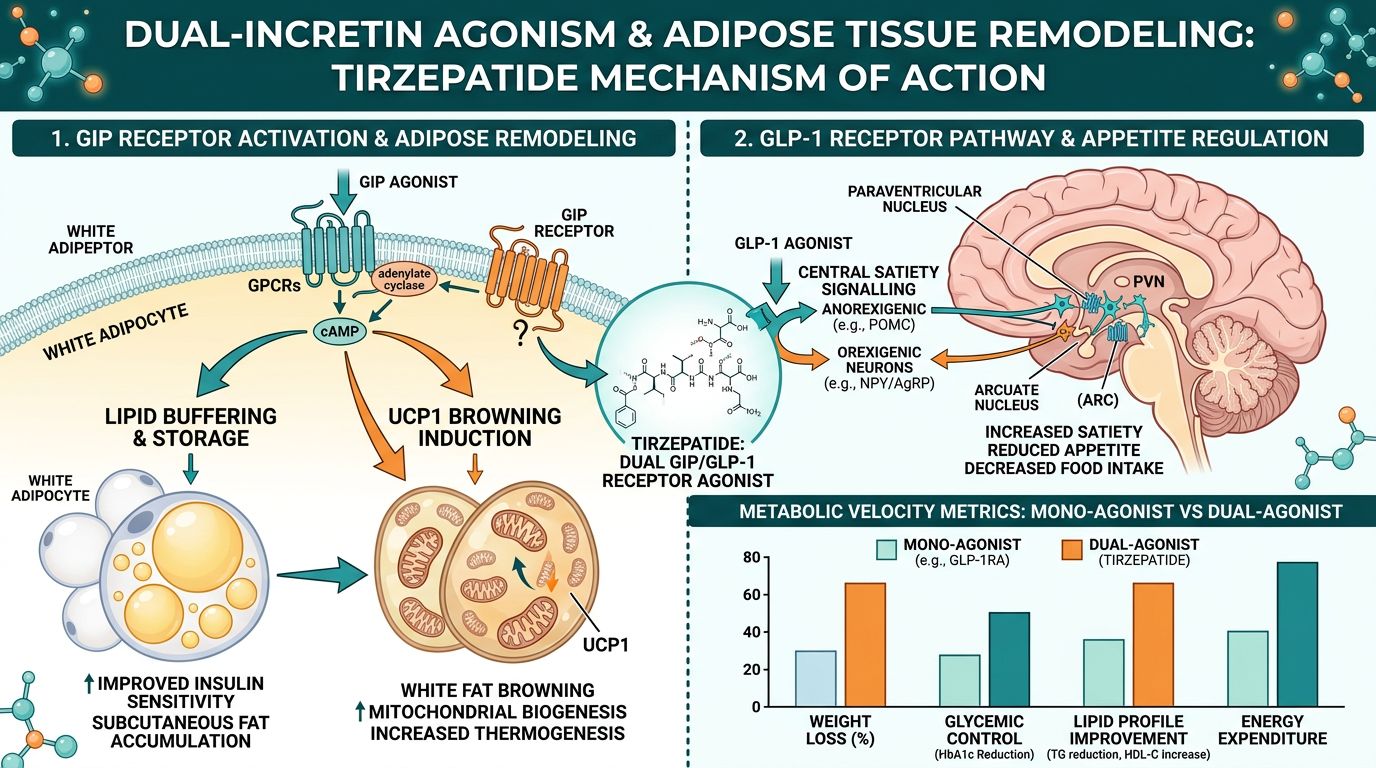
Quantifying Adipose Tissue Remodeling
In research models of diet-induced obesity (DIO), the administration of Tirzepatide produces a distinct phenotypic change in adipose morphology. While single-agonist GLP-1 molecules primarily reduce fat mass through appetite suppression, Tirzepatide active research shows an acceleration in the following areas:
- Lipid Buffering Capacity: Increasing the ability of subcutaneous adipose tissue to safely store triglycerides, thereby reducing circulating free fatty acids.
- Thermogenesis and "Browning": Induction of UCP1 expression in white adipocytes, effectively converting energy-storage cells into energy-expenditure cells (beige fat).
- Anti-inflammatory Modulation: Reducing the infiltration of M1-polarized macrophages into adipose tissue, a primary driver of systemic insulin resistance.
Comparative Efficacy: Tirzepatide vs. Older Incretins
When compared to single-agonists or other research peptides like AOD9604 or HGH Fragment 176-191, Tirzepatide demonstrates a synergistic effect that exceeds the sum of its parts. The table below outlines the metabolic velocity differences observed in comparative murine studies.
| Parameter | GLP-1 Mono-agonist (Semaglutide) | Dual Agonist (Tirzepatide) | Metabolic Velocity Impact |
|---|---|---|---|
| Weight Loss Origin | Mainly Hypothalamic (Appetite) | Appetite + Adipose Remodeling | Higher (Multi-modal) |
| Insulin Sensitivity (HOMA-IR) | Moderate Improvement | Significant Improvement | High (GIP-mediated sensitivity) |
| Visceral Fat Reduction | Dose-Dependent | Accelerated | Enhanced Proteolysis |
| Energy Expenditure | Stable or Slight Decrease | Increase (Browning induction) | Positive Metabolic Shift |
Optimizing Insulin Sensitivity in Murine Models
The "Metabolic Velocity" of Tirzepatide is most evident when analyzing the kinetics of glucose clearance. GIP receptor activation appears to potentate the GLP-1 response, particularly in the pancreatic beta cells. In research observing islet architecture, Tirzepatide has been shown to preserve beta-cell mass more effectively than GLP-1 alone.
Furthermore, Tirzepatide research often intersects with mitochondrial health. Researchers looking to maximize fat oxidation in research subjects may combine incretin mimetics with mitochondrial-targeted peptides such as MOTS-c or SS-31. This allows for a two-pronged approach: Tirzepatide mobilizes lipid stores and improves insulin signaling, while MOTS-c ensures the liberated fatty acids are efficiently oxidized within the mitochondria.
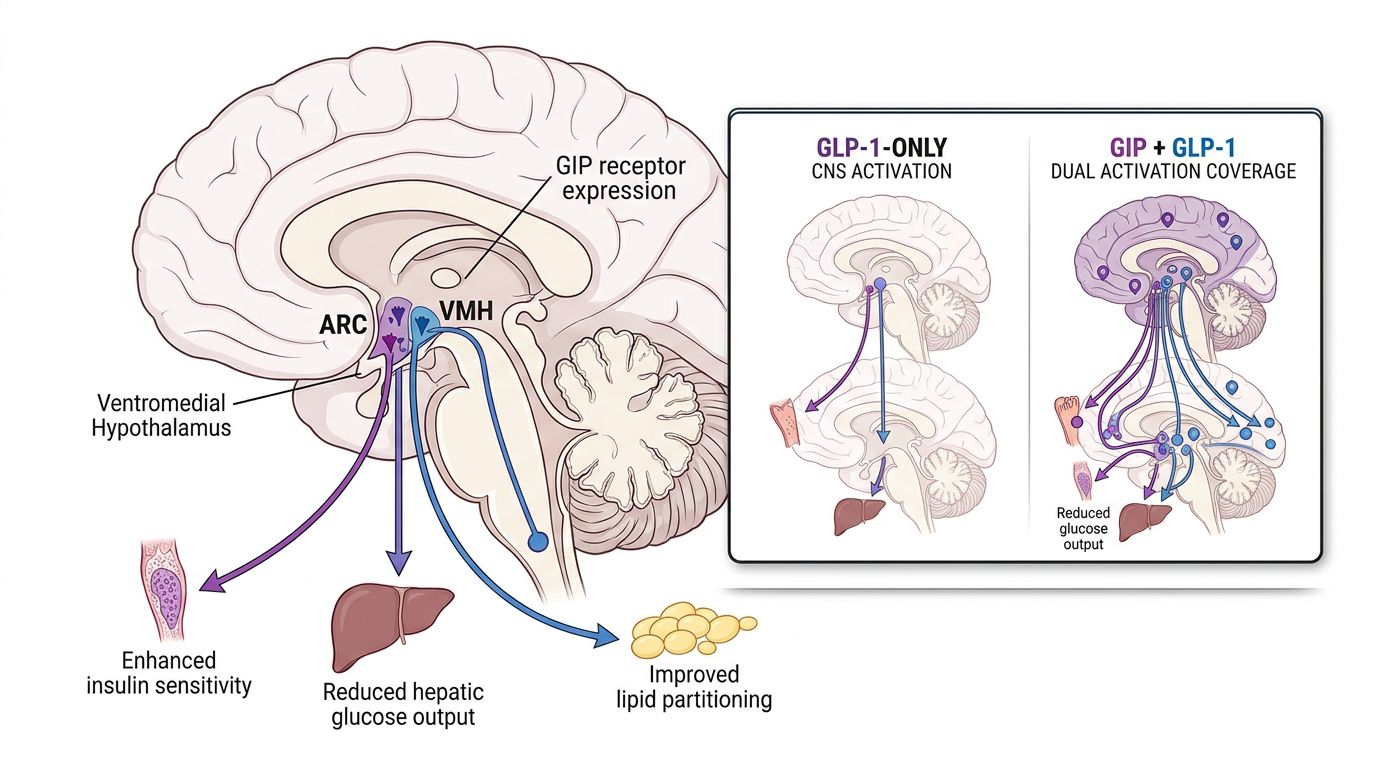
The Role of GIP in the CNS
One of the most profound discoveries in the study of Tirzepatide is the density of GIP receptors in the hypothalamus. While GLP-1 is known for its role in satiety within the nucleus tractus solitarius (NTS), GIPR signaling appears to modulate the rewarding aspects of food intake in the area postrema. By targeting both pathways, Tirzepatide achieves a more complete suppression of "hedonic hunger," allowing research models to achieve caloric deficits that were previously unsustainable.
Advanced Synergies: Integrating Tirzepatide in Comprehensive Research
For researchers evaluating metabolic syndrome beyond simple weight loss, Tirzepatide is frequently studied alongside peptides that manage auxiliary symptoms. For example, during significant adipose reduction, systemic inflammation can be an obstacle. Utilizing BPC-157 or KPV in tandem may help modulate the inflammatory response associated with rapid metabolic shifts.
Additionally, for models where muscle preservation is a primary endpoint alongside fat loss, combining Tirzepatide with Tesamorelin or IGF-1LR3 allows for the investigation of nutrient partitioning—directing calories away from fat storage and toward muscle protein synthesis. You can review the purity of these compounds via our COA documents to ensure experimental consistency.
Mechanism of Action: Step-by-Step Lipid Flux
- Activation: Tirzepatide binds to GLP-1 and GIP receptors on the cell membrane of adipocytes and pancreatic cells.
- Incretin Effect: Glucose-dependent insulin secretion increases, reducing post-prandial glucose spikes.
- Glucagon Modulation: Unlike pure GLP-1 agonists, the GIP component of Tirzepatide may modulate glucagon secretion in a manner that prevents hypoglycemia while maintaining catabolic pressure on adipose tissue.
- Lipolysis vs. Lipogenesis: Activation of GIPR in white adipose tissue increases the blood flow and the clearance of chylomicrons, facilitating a "healthier" metabolism of dietary fats.
Safety and Rigidity in Research Environments
As with all powerful metabolic research tools, quantifying the safety profile is as important as measuring efficacy. In murine models, the primary side effects are gastrointestinal; however, these are often transient. Researchers must ensure they are utilizing pharmaceutical-grade lyophilized peptides to avoid impurities that could skew metabolic data. Information on our manufacturing standards can be found on our Quality Control page.
In high-velocity metabolic studies, monitoring for dehydration and electrolye balance is essential, especially when combining Tirzepatide with other metabolic accelerators like 5-amino-1mq or AICAR. These combinations can significantly alter ATP/AMP ratios, necessitating careful observation of intracellular energy flux.
Future Directions: Triple Agonism and Beyond
The success of Tirzepatide has paved the way for "tri-agonist" research, where Glucagon receptor (GCGR) agonism is added to the GIP/GLP-1 backbone. Peptides like Retatrutide are currently the subject of intense scrutiny for their ability to increase metabolic velocity even further by directly stimulating hepatic fat oxidation. However, for many research protocols, the dual-agonism provided by Tirzepatide remains the gold standard for clinical-to-research translatability.
Researchers may also find interest in the development of long-acting oral versions or co-formulations with next-generation peptides like Cagrilintide (an amylin analogue) to further optimize satiety signals and gastric emptying rates.
Conclusion
Tirzepatide is more than a weight-loss molecule; it is a sophisticated tool for understanding the plasticity of adipose tissue and the complexity of the incretin system. By integrating GIP and GLP-1 agonism, it offers a "metabolic velocity" that traditional therapies cannot match. For researchers focused on insulin sensitivity, lipid metabolism, and the prevention of metabolic decay, Tirzepatide provides a robust framework for study.
Alpha Carbon Labs provides high-purity Tirzepatide and related research compounds to facilitate advanced metabolic studies. Through rigorous testing and a commitment to scientific transparency, we support the research community in uncovering the next generation of metabolic interventions.
References
- 1. Frias JP, et al. (2021). Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine.
- 2. Cosin-Roger J, et al. (2021). The dual GIP and GLP-1 receptor agonist tirzepatide improves pancreatic islet function and insulin sensitivity in adults with type 2 diabetes. Nature Metabolism.
- 3. Samms RJ, et al. (2020). GIPR Agonism Mediates Weight-Independent Insulin Sensitization by Effector Mechanisms in Adipocyte and Hepatocyte. Diabetes.
- 4. Finan B, et al. (2013). Unimolecular dual incretins maximize metabolic benefits in rodents, monkeys, and humans. Science Translational Medicine.
- 5. Moyers JS, et al. (2022). Tirzepatide is a potent and selective dual GIP and GLP-1 receptor agonist with an unbalanced activity. JCI Insight.
- 6. Heise T, et al. (2022). Tirzepatide reduces appetite, energy intake, and fat mass in people with type 2 diabetes. Diabetes Care.
- 7. Baggio LL, Drucker DJ. (2021). Glucagon-like peptide-1 receptors in the brain: Controlling food intake and body weight. Journal of Clinical Investigation.
- 8. Zhang Q, et al. (2022). The GIP receptor Agonist Tirzepatide Induces White Adipose Tissue Browning. Molecular Metabolism.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.