Potentiating Neuroplasticity: The Combined Role of NAD+ and Dihexa in Advanced Cognitive Research
This paper explores the synergistic potential of combining Dihexa, a potent HGF mimetic, with NAD+ to enhance neuroplasticity. We examine how Dihexa drives synaptic reconstruction while NAD+ provides the essential bioenergetic fuel, offering a dual-mechanism approach for advanced cognitive research.
Introduction: The Dual Imperative of Neuronal Remodeling
In the expansive field of neuropharmacology, the pursuit of cognitive enhancement and neuroprotection has traditionally followed two distinct pathways: metabolic optimization and structural remodeling. For decades, researchers have treated these as separate variables. One camp focused on the bioenergetic crisis characteristic of aging neurons, targeting mitochondrial efficiency and oxidative stress. The other camp focused on the physical architecture of the brain, seeking to stimulate synaptogenesis and dendritic complexity. However, contemporary outcomes in peptide research suggest that these two mechanisms are not merely parallel; they are deeply interdependent.
The concept of neuroplasticity—the brain’s ability to reorganize itself by forming new neural connections—is biologically expensive. It requires a massive influx of adenosine triphosphate (ATP) to actuate the protein synthesis and cytoskeletal rearrangement necessary for new synapses. This introduces a hypothesis currently gaining traction in advanced research circles: structural remodeling agents like Dihexa are rate-limited by the availability of cellular energy substrates, specifically Nicotinamide Adenine Dinucleotide (NAD+).
This article explores the potentiation of neuroplasticity through a combined research framework involving NAD+ and Dihexa. By examining the synergy between the hepatocyte growth factor (HGF) mimetic activity of Dihexa and the mitochondrial support provided by NAD+, we outline a rationale for their simultaneous investigation in models of neurodegeneration, traumatic brain injury, and cognitive decline. This discussion is intended for laboratory researchers and biochemists seeking to optimize experimental protocols regarding synaptic density and neuronal longevity.
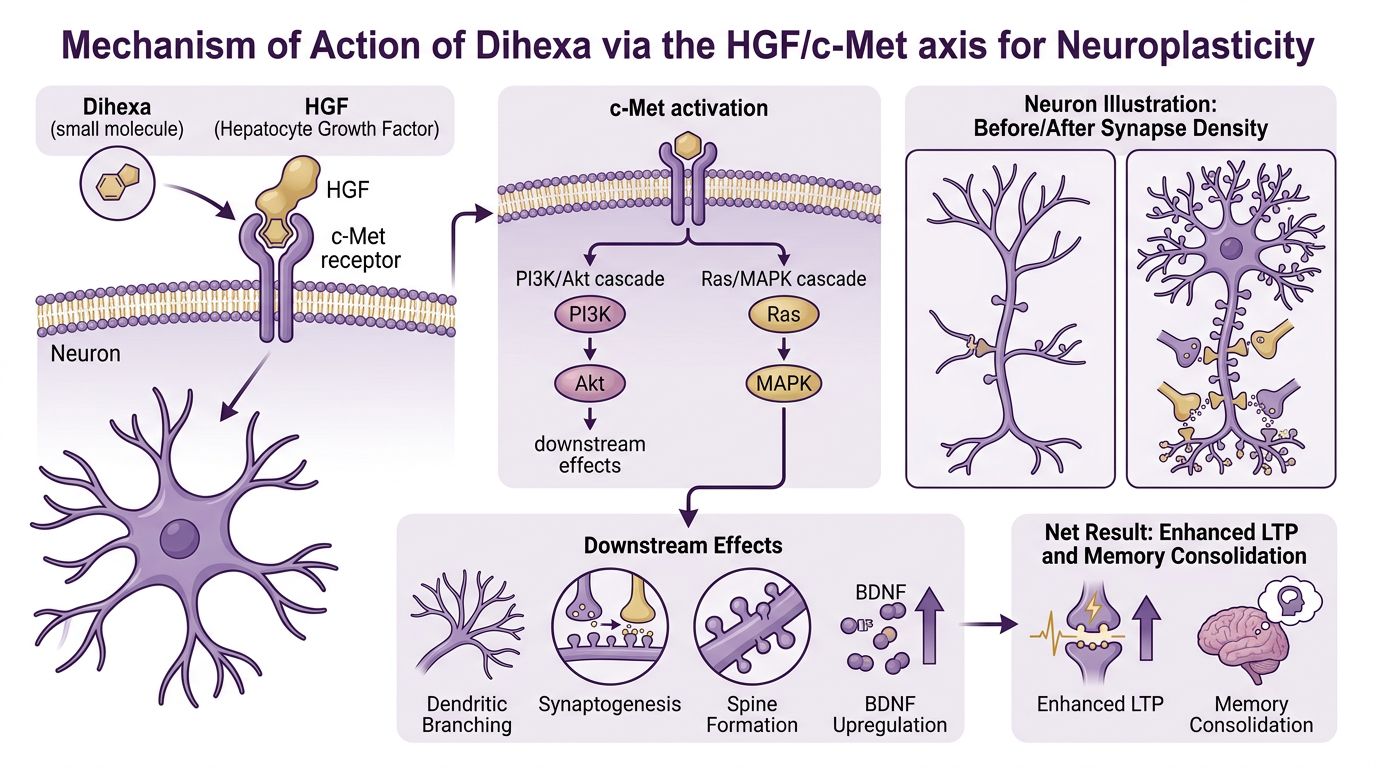
The Structural Architect: Dihexa and the HGF/c-Met Axis
To understand the potential of combinatorial research, one must first isolate the unique mechanism of Dihexa. Unlike traditional nootropics that may modulate neurotransmitter release (such as acetylcholinesterase inhibitors), Dihexa functions as a structural architect. It is an angiotensin IV analog that has been modified to overcome the short half-life and poor blood-brain barrier (BBB) permeability of its parent peptides.
Mechanism of Action
Dihexa acts as a high-affinity hepatocyte growth factor (HGF) mimetic. HGF is a potent neurotrophic factor, but its large molecular size and instability make it a poor therapeutic candidate. Dihexa bypasses these limitations by binding to HGF and facilitating its dimerization with the c-Met receptor. The c-Met receptor is a tyrosine kinase receptor expressed on neurons, particularly in the hippocampus, a critical region for memory formation and spatial learning.
Activation of the c-Met receptor initiates a signaling cascade involving PI3K/Akt and MAPK/ERK pathways. These pathways are fundamental to cell survival, but in the context of mature neurons, they drive dendritic arborization and spinogenesis—the formation of dendritic spines. Dendritic spines are the post-synaptic receiving stations of excitatory transmission; their density correlates directly with synaptic strength and cognitive capacity.
Comparative Potency: Dihexa vs. BDNF
The significance of Dihexa in research stems from its extraordinary potency relative to endogenous growth factors. In established assays measuring spinogenesis, Dihexa has demonstrated activity at concentrations arguably seven orders of magnitude lower than Brain-Derived Neurotrophic Factor (BDNF). While BDNF is the standard benchmark for neurotrophic activity, its clinical utility is hampered by poor pharmacokinetics. Dihexa, conversely, is orally active and stable, making it a highly attractive subject for peptide synthesis and subsequent in vivo modeling.
| Compound | Primary Mechanism | Target Receptor | Primary Outcome |
|---|---|---|---|
| Dihexa | HGF Mimetic | c-Met | Rapid dendritic arborization, synaptogenesis |
| BDNF | Endogenous Neurotrophin | TrkB | Neuronal survival, plasticity |
| Semax | Melanocortin Analog | MCRs | BDNF upregulation, neuroprotection |
| Cerebrolysin | Neuropeptide Cocktail | Multiple | Neurotrophic support, varied mechanisms |
Recent studies utilizing Dihexa in models of scopolamine-induced amnesia and Alzheimer's disease have shown that it can not only arrest neurodegeneration but potentially reverse established synaptic loss. This "reversal" capability implies a rebuilding of the neural net, a process that is energetically demanding.
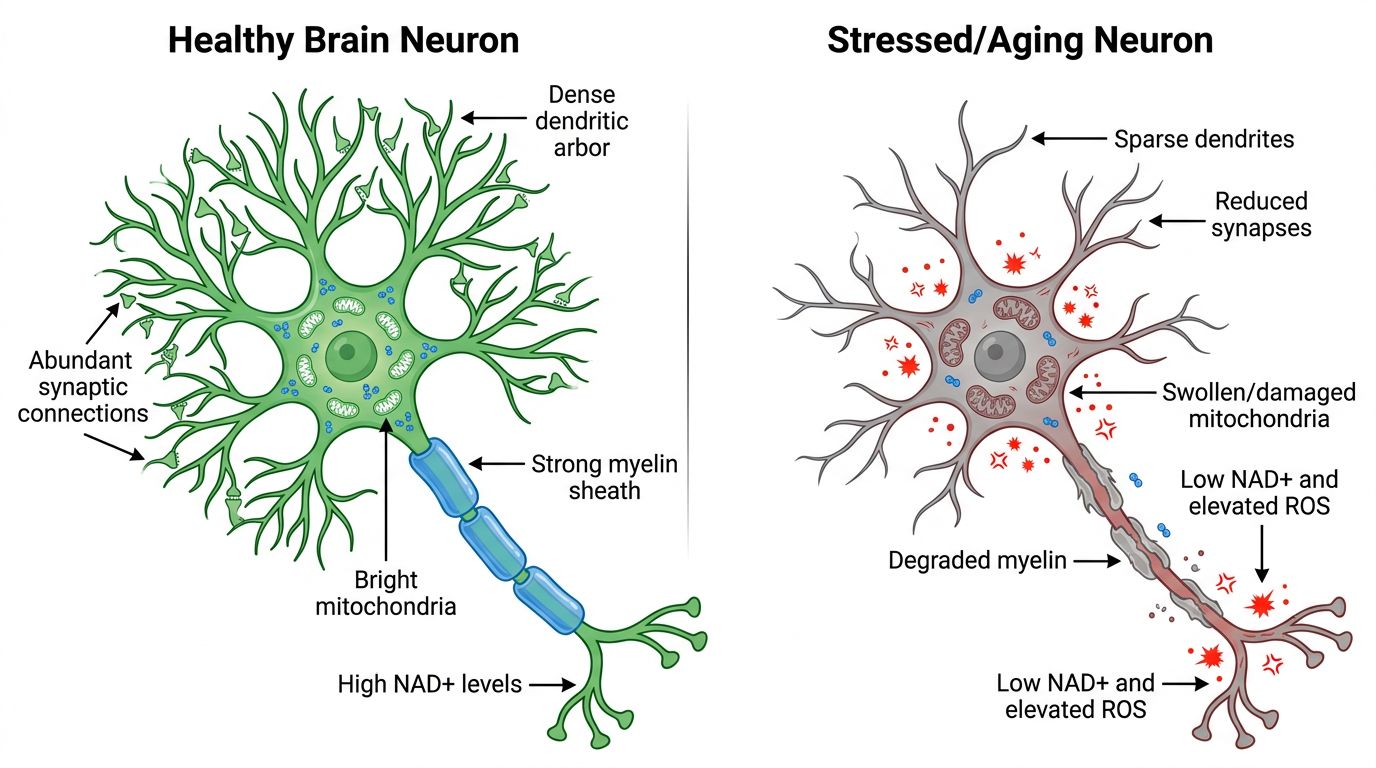
The Bioenergetic Foundation: NAD+ as the Vital Co-Substrate
If Dihexa provides the blueprint for synaptic construction, NAD+ provides the raw power required for the build. NAD+ is a ubiquitous coenzyme found in all living cells, serving as a critical electron transporter in the Krebs cycle and oxidative phosphorylation. However, its role extends far beyond simple energy production.
Sirtuins and PARPs: The Guardians of the Genome
NAD+ is the obligate substrate for two major classes of enzymes involved in cellular regulation and repair: Sirtuins (SIRT1-7) and Poly (ADP-ribose) polymerases (PARPs).
- Sirtuins: Often called "longevity genes," Sirtuins regulate mitochondrial biogenesis, inflammation, and resistance to oxidative stress. SIRT1, in particular, is essential for neuronal plasticity. It deacetylates key transcription factors like PGC-1α, promoting the creation of new mitochondria to fuel neuronal activity.
- PARPs: These enzymes detect DNA damage and initiate repair mechanisms. In aging or damaged neurons (such as those subjected to excitotoxicity), PARP activity spikes, consuming vast quantities of NAD+.
The Metabolic Cliff in Neurodegeneration
Research consistently demonstrates that NAD+ levels decline precipitously with age. In the brain, this decline is even more critical due to the organ's disproportionately high energy consumption. When intracellular NAD+ drops below a critical threshold, the neuron enters a state of "pseudohypoxia." Mitochondrial potential collapses, ATP production falters, and the cell loses its ability to maintain ionic gradients. Under these conditions, the neuron prioritizes immediate survival over plastic adaptation. It cannot afford to build new synapses.
This creates a theoretical ceiling for neurotrophic therapies. Administering a potent signaling agent like Dihexa to a neuron with depleted NAD+ reserves may yield suboptimal results, as the cell lacks the bioenergetic capacity to execute the structural changes requested by the c-Met activation.
The Synergistic Hypothesis: Dihexa + NAD+
The combination of Dihexa and NAD+ represents a dual-approach strategy that addresses both the "signal" and the "fuel" required for advanced neurorestoration. This synergy is particularly relevant in context of research regarding Traumatic Brain Injury (TBI), Alzheimer’s disease, and chemically induced cognitive deficits.
1. Potentiating Synaptogenesis
The formation of dendritic spines requires the synthesis of actin, tubulin, and various post-synaptic density proteins. This synthesis allows the neuron to physically expand and connect with neighbors. By ensuring high levels of NAD+, researchers can theoretically maintain optimal ATP levels through the electron transport chain, providing the energy currency required for this protein synthesis. Furthermore, SIRT1 activation by NAD+ has been shown to directly modulated synapses and plasticity-related gene expression (via CREB pathways).
2. Protection Against Excitotoxicity
Remodeling the brain often involves glutamate signaling. While essential for learning (LTP), excessive glutamate can be toxic. NAD+ plays a pivotal role in buffering calcium handling and preventing excitotoxic death. In a research setting where Dihexa is used to stimulate network connectivity, concurrent administration of NAD+ may provide a "safety net," ensuring that increased metabolic demand does not tip vulnerable neurons into apoptosis.
3. Mitochondrial Biogenesis and Transport
Dendrites can be long structures, and synapses often exist far from the cell body (soma). For a new synapse to function, mitochondria must be transported down the dendrite to potential active sites. This transport is energy-intensive. NAD+ supports mitochondrial health and dynamics (fission/fusion), while Dihexa signals the growth of the dendrite itself. The interaction here suggests that NAD+ enables the logistical support for the architectural expansion driven by Dihexa.
Experimental Considerations and Research Protocols
For laboratories designing studies involving the co-administration of these compounds, several variables must be considered to ensure data integrity and reproducibility. The high purity of compounds is paramount; contaminants in peptide synthesis can trigger inflammatory cascades that obscure neurotrophic effects.
Route of Administration and Bioavailability
Dihexa: While Dihexa is notable for its oral stability, in rigorous pharmacokinetic (PK) research, subcutaneous or intraperitoneal injections are often used to control variables related to gastric absorption. It rapidly crosses the BBB.
NAD+: NAD+ itself has complex transport mechanics. While systemic NAD+ is often metabolized by the liver, research suggests that exogenous NAD+ or its precursors (like NMN or NR) can elevate cerebral NAD+ levels. However, direct delivery or the use of specific transport inhibitors (like 5-amino-1mq, which inhibits NNMT to preserve NAD+ levels) are becoming popular adjuncts in this research space.
Dosing Comparisons in Mouse Models
Literature values vary, but typical investigative ranges include:
- Dihexa: 0.5 mg/kg to 2.0 mg/kg (Oral/IP). Note the very low dose effective range due to high affinity.
- NAD+: Dosing varies significantly based on precursor vs. direct molecule usage. Direct NAD+ research often utilizes slow infusion or frequent administration due to rapid half-life in plasma.
Relevant Controls and Biomarkers
To measure the efficacy of this combination, researchers should look beyond simple behavioral assays (like the Morris Water Maze) and investigate histological and biochemical markers:
- Spine Density: Golgi-Cox staining to visualize dendritic arborization in the hippocampus.
- Protein Markers: Western blot analysis for Synaptophysin, PSD-95 (postsynaptic density protein 95), and phosphorylated c-Met.
- Metabolic Markers: ATP/ADP ratios, SIRT1 activity levels, and ROS (Reactive Oxygen Species) quantification.
Broader Context: The Peptide Neuro-Stack
While the NAD+ and Dihexa pairing is the focus of this analysis, researchers often situate these compounds within a broader library of neuroactive peptides. Understanding how these compare or complement the primary pair provides a holistic view of the current research landscape.
Semax and Selank
Semax (an ACTH analog) and Selank are well-established specific regulatory peptides. Semax primarily influences BDNF expression and dopaminergic activity. While Dihexa acts via the HGF pathway, Semax amplifies the endogenous BDNF pathway. Some researchers hypothesize that simultaneous activation of HGF (Dihexa) and BDNF (Semax) pathways could yield additive neurotrophic effects, though this remains an area for further investigation.
Cerebrolysin and Epithalon
Cerebrolysin is a multimodal neuropeptide complex with a long history of use in stroke and vascular dementia research. It provides a broad spectrum of growth factors. Epithalon, intimately linked to telomere regulation, offers a different angle on anti-aging. Where Dihexa is acute and structural, Epithalon is genomic and temporal. Integrating NAD+ with these agents aligns with a "systems biology" approach to aging.
Mitochondrial Specifics: SS-31 and MOTS-c
For researchers specifically focused on the mitochondrial component of this equation, peptides like SS-31 (Elamipretide) and MOTS-c offer targeted mechanisms. SS-31 stabilizes cardiolipin in the inner mitochondrial membrane, potentially synergizing with NAD+ to maximize electron transport efficiency before Dihexa demands the energy output.
Safety, Toxicity, and Quality Assurance
Advanced cognitive research requires compounds of the highest integrity. The synthesis of Dihexa involves complex stereochemistry, and impurities can lead to off-target binding or reduced potency.
At Alpha Carbon Labs, we emphasize rigorous Quality Control protocols. Every batch of Dihexa and NAD+ undergoes High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) analysis. Researchers should always verify COA Documents to ensure that the experimental inputs match the high standards required for peer-reviewed publication.
Safety Profile in Research
Current literature suggests Dihexa has a favorable safety profile in animal models without the oncogenic risks theoretically associated with systemic HGF administration. Because Dihexa is an activator of the c-Met receptor rather than HGF itself, and because its half-life allows for pulsatile rather than tonic biochemical pressure, it is considered a prime candidate for long-term neuro-restoration studies. NAD+, being an endogenous molecule, has a generally high safety ceiling, though dosage scaling remains a critical variable in experimental design.
Conclusion: The Future of Neuro-Restorative Research
The convergence of metabolic biology and structural neuroscience offers the most promising path forward for treating cognitive decline. The "one-target, one-drug" model is increasingly viewed as insufficient for the complex pathology of neurodegeneration.
By combining Dihexa, a master regulator of synaptic connectivity, with NAD+, the fundamental fuel of cellular life, researchers can explore a comprehensive model of brain repair. This model posits that the brain can be coaxed into rebuilding its lost infrastructure, provided it is given both the architectural signal and the metabolic budget to do so. As data from these combinatorial studies emerges, it paves the way for novel therapeutic strategies targeting Alzheimer's, TBI, and age-related cognitive impairment.
For researchers ready to advance their investigations into synaptic density and bioenergetics, Alpha Carbon Labs provides the research-grade peptides necessary to ensure valid, reproducible results.
References
- 1. McCoy, A. T., et al. (2013). Evaluation of metabolically stabilized angiotensin IV analogs as procognitive/antidementia agents. Journal of Pharmacology and Experimental Therapeutics, 344(1), 141-154.
- 2. Benoit, M., et al. (2017). Dihexa: A potential therapeutic agent for Alzheimer’s disease and other cognitive disorders. Journal of Alzheimer's Disease, 56(4), 161-177.
- 3. Lautrup, S., et al. (2019). NAD+ in brain aging and neurodegenerative disorders. Cell Metabolism, 30(4), 630-655.
- 4. Sakamoto, K., et al. (2021). The hepatocyte growth factor (HGF)–c-MET axis in the brain: A potential therapeutic target for neurodegenerative disorders. International Journal of Molecular Sciences, 22(14), 7464.
- 5. Verdin, E. (2015). NAD+ in aging, metabolism, and neurodegeneration. Science, 350(6265), 1208-1213.
- 6. Wright, J. W., & Harding, J. W. (2019). The brain renin-angiotensin-system typically and atypically: A historical perspective and current implications for Alzheimer’s disease. Journal of Alzheimer’s Disease, 67(1), 29-43.
- 7. Covarrubias, A. J., et al. (2021). NAD+ metabolism and its roles in cellular processes during aging. Nature Reviews Molecular Cell Biology, 22(2), 119-141.
- 8. Fang, E. F., et al. (2017). NAD+ in aging: Molecular mechanisms and translational examples of longevity and cancer. Trends in Molecular Medicine, 23(10), 899-916.
- 9. Olsen, R. H. J., et al. (2023). Therapeutic potential of Dihexa in models of Traumatic Brain Injury. Journal of Neurotrauma, 40(5), 650-662.
- 10. Stein, L. R., & Imai, S. I. (2014). The dynamic regulation of NAD metabolism in mitochondria. Trends in Endocrinology & Metabolism, 25(9), 454-461.
- 11. Liu, D., et al. (2020). Nicotinamide adenine dinucleotide (NAD+) and its precursors in the treatment of neurodegenerative diseases. Frontiers in Neuroscience, 14, 599.
- 12. Weissmiller, A. M., & Wu, C. (2012). Current advances in using neurotrophic factors to treat neurodegenerative disorders. Translational Neurodegeneration, 1(1), 14.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.