Myogenic Regulatory Factors: Comparing the Efficacy of GHRP-2 and GHRP-6 in Lean Mass Preservation Research
Explore the distinct mechanisms of GHRP-2 and GHRP-6 in preserving lean muscle mass during caloric restriction. This detailed analysis compares their effects on myogenic regulatory factors, appetite signaling, and the IGF-1 axis to determine the superior research candidate.
Introduction to Peptide-Mediated Muscle Preservation
In the realm of regenerative medicine and metabolic research, the challenge of preserving lean muscle mass during periods of caloric deficit or catabolic stress remains a primary focus. Muscle tissue is metabolically expensive; under conditions of starvation, injury, or severe dieting, the body often prioritizes the breakdown of myofibrillar protein to provide amino acids for gluconeogenesis. This evolutionary survival mechanism, while effective for short-term energy balance, is detrimental to long-term metabolic health and physical function.
Research into mitigating this catabolic response has largely centered on the Growth Hormone (GH) and Insulin-Like Growth Factor 1 (IGF-1) axis. Within this axis, synthetic Growth Hormone Secretagogues (GHS)—specifically GHRP-2 and GHRP-6—have emerged as significant subjects of study. These peptides, though structurally similar, exhibit distinct pharmacokinetic profiles and varying impacts on appetite, cortisol secretion, and the transcription of Myogenic Regulatory Factors (MRFs).
This article provides a comprehensive scientific analysis of the comparative efficacy of GHRP-2 and GHRP-6. We will explore how these peptides influence the molecular machinery of muscle protein synthesis, specifically focusing on their interaction with myogenic genes during caloric restriction.
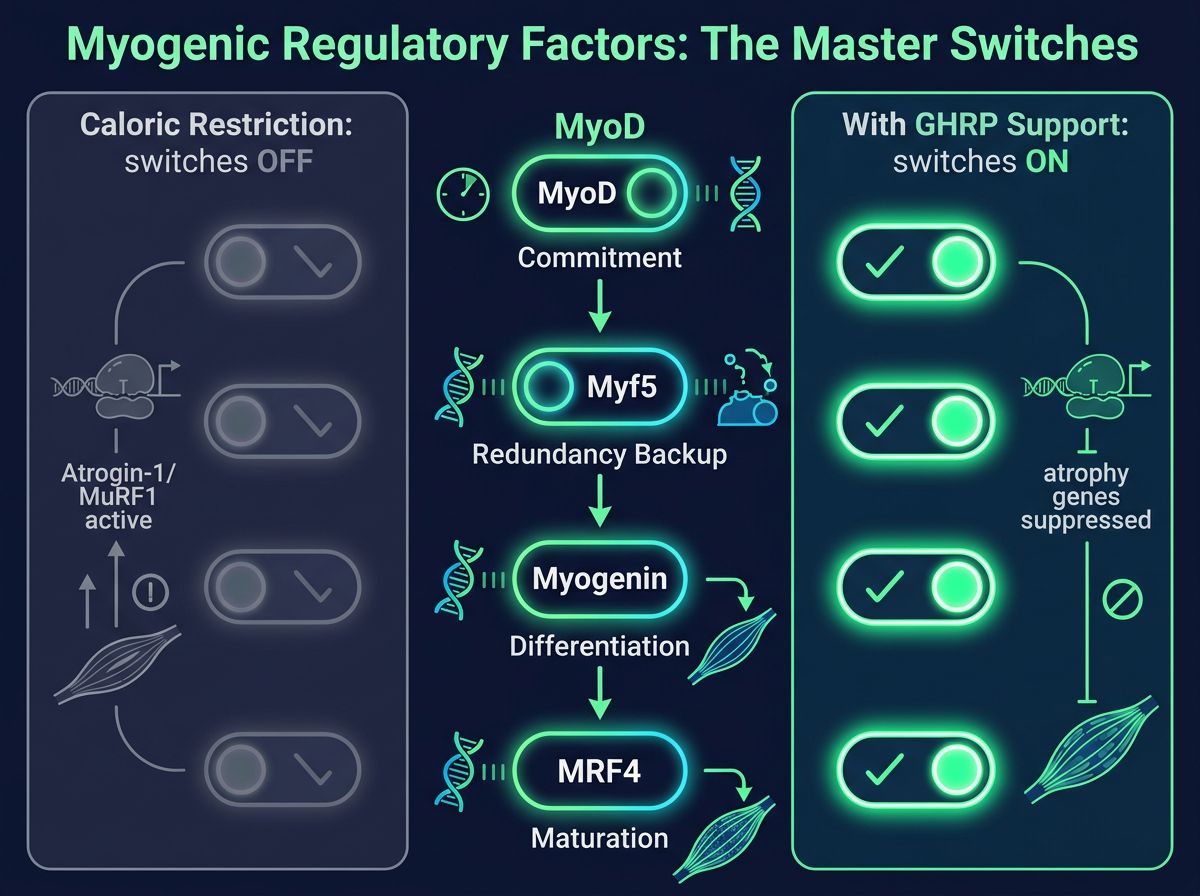
The Molecular Basis: Myogenic Regulatory Factors (MRFs)
To understand how GHRPs preserve muscle, one must first understand the genetic drivers of muscle growth. Myogenic Regulatory Factors (MRFs) are a family of transcription factors that orchestrate myogenesis—the formation and repair of skeletal muscle. They are the "master switches" that convert cellular signals into structural protein synthesis.
The primary MRFs include:
- MyoD (Myoblast Determination Protein 1): Acts early in the pathway, committing mesenchymal stem cells (satellite cells) to the myogenic lineage. It is essential for the activation of satellite cells following micro-trauma.
- Myf5 (Myogenic Factor 5): Works in tandem with MyoD. Redundancy between MyoD and Myf5 ensures that muscle determination occurs even if one factor is compromised.
- Myogenin: Functions downstream of MyoD/Myf5, driving the differentiation of myoblasts into multinucleated myotubes. It is critical for the formation of functional muscle fibers.
- MRF4 (Muscle Regulatory Factor 4): Involved in both differentiation and the maturation/maintenance of adult muscle fibers.
During caloric restriction or catabolic states, the expression of these factors—particularly MyoD and Myogenin—is typically downregulated. Simultaneously, ubiquitin-ligases such as Atrogin-1 and MuRF1 are upregulated, tagging muscle proteins for degradation. The research hypothesis surrounding GHRPs suggests that by amplifying the pulsatile release of GH and subsequently IGF-1, these peptides may maintain MRF expression and inhibit ubiquitin-proteasome activity, effectively "shielding" muscle tissue from catabolism.
The Mechanism of Action: GHS-R1a Activation
Both GHRP-2 and GHRP-6 function as agonists of the Ghrelin Receptor, formally known as the Growth Hormone Secretagogue Receptor 1a (GHS-R1a). This receptor is located primarily in the pituitary gland and the hypothalamus.
Intracellular Signaling
When GHRP-2 or GHRP-6 binds to GHS-R1a, it triggers a G-protein coupled cascade involving Phospholipase C (PLC). This catalyzes the hydrolysis of phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol triphosphate (IP3) and diacylglycerol (DAG). The subsequent release of intracellular calcium (Ca2+) combined with the inhibition of somatostatin (the hormone that blocks GH release) results in a powerful, pulsatile release of Growth Hormone.
While both peptides utilize this pathway, their binding affinities and secondary effects differ significantly, influencing their utility in research focused on lean mass preservation.
GHRP-6: Structurally Broad, Metabolically Active
As a first-generation hexapeptide, GHRP-6 established the baseline for synthetic secretagogues. Its effects extend beyond simple somatotroph stimulation.
Distinct Research Characteristics of GHRP-6
- Broad Receptor Affinity: GHRP-6 binds to GHS-R1a but also affects other receptor subtypes, potentially explaining its broader physiological effects.
- Appetite Stimulation: GHRP-6 is a potent hunger inducer. In animal models, it mimics the action of ghrelin primarily in the hypothalamus, triggering orexigenic (appetite-simulating) neurons. While this may seem counterintuitive for "dieting," it is relevant in cachexia (wasting syndrome) research where increasing caloric intake is vital alongside hormonal support.
- Cytoprotection: Unique to GHRP-6 is its demonstrated ability to protect cells from distinct injuries, independent of the IGF-1 axis. Research suggests it may reduce inflammation and oxidative stress in cardiac and hepatic tissues.
In the context of muscle preservation, GHRP-6 induces a massive spike in GH. However, its intense stimulation of gastric motility and hunger can complicate research protocols requiring controlled caloric intake. It is often compared to MK-677 (Ibutamoren) regarding appetite effects, though MK-677 has a much longer half-life.
GHRP-2 (Pralmorelin): Potency and Specificity
GHRP-2 is a second-generation hexapeptide. Structural modifications have made it a more specific agonist for the GHS-R1a receptor compared to its predecessor.
Distinct Research Characteristics of GHRP-2
- Higher GH Secretion: Head-to-head disparate studies repeatedly demonstrate that GHRP-2 elicits a stronger release of Growth Hormone per microgram than GHRP-6. In some assays, the release is nearly double that of GHRP-6.
- Reduced Orexis: While GHRP-2 still stimulates appetite, the effect is significantly attenuated compared to GHRP-6. This makes GHRP-2 a preferred candidate for research involving caloric restriction where variable caloric intake must be minimized.
- Prolactin and Cortisol: A notable trade-off is that GHRP-2 stimulates the release of Prolactin and Cortisol (ACTH axis) to a greater degree than GHRP-6, particularly at saturation doses. While typically remaining within physiological stress limits, this is a variable researchers must account for.
For high-purity research materials, Alpha Carbon Labs provides stringent quality control data to ensure the integrity of these comparative observations.
Comparative Efficacy in Muscle Protein Synthesis
The core of the efficacy debate lies in how these peptides influence the balance between Anabolism (Protein Synthesis) and Catabolism (Protein Breakdown).
1. The IGF-1 / PI3K / Akt / mTOR Pathway
Both peptides ultimately work by elevating hepatic production of IGF-1. IGF-1 binds to receptors on the muscle cell membrane, activating the PI3K/Akt pathway. This activation leads to the phosphorylation of mTOR (Target of Rapamycin), which is arguably the central regulator of protein synthesis.
When mTOR is activated, it triggers downstream effectors like p70S6K, which initiate the translation of mRNA into new muscle proteins. Because GHRP-2 stimulates a higher peak of GH, it theoretically drives a stronger momentary pulse of IGF-1, potentially triggering this anabolic cascade more aggressively than GHRP-6.
2. Inhibition of FoxO Transcription Factors
Muscle loss during caloric restriction is largely driven by the FoxO family of transcription factors, which activate the "atropy genes" (Atrogin-1 and MuRF1). The Akt pathway, activated by the GH/IGF-1 surge from GHRP administration, phosphorylates FoxO, trapping it in the cytoplasm and preventing it from entering the nucleus.
In this capacity, both GHRP-2 and GHRP-6 are anti-catabolic. However, due to the dose-dependent nature of this signaling, GHRP-2's higher potency may offer a stronger "brake" on the catabolic machinery.
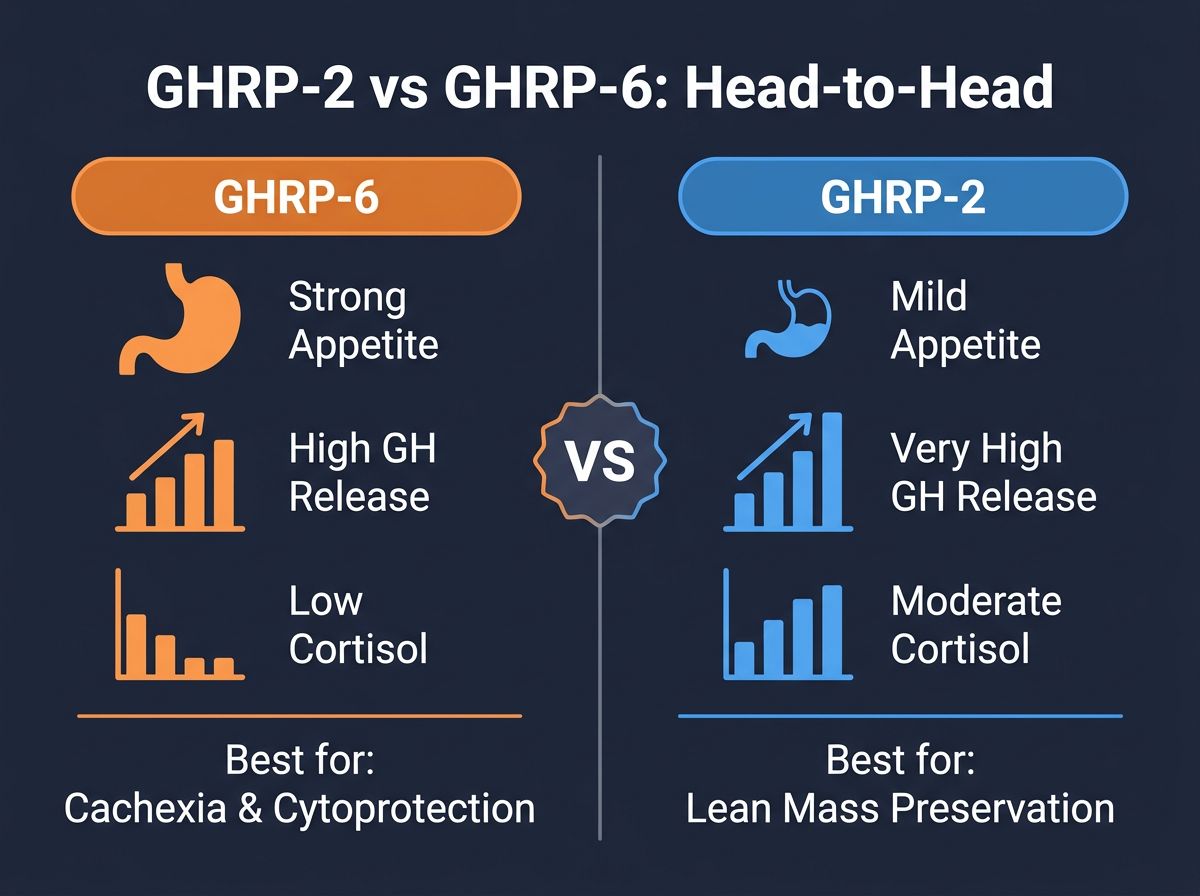
Table 1: Comparative Profile of GHRP-2 and GHRP-6
| Parameter | GHRP-6 | GHRP-2 |
|---|---|---|
| GH Release Potency | High | Very High (approx. 1.3x - 2x stronger) |
| Appetite Stimulation | Strong (Orexigenic) | Identifyable but Mild |
| Cortisol/Prolactin Response | Minimal | Moderate (Dose-dependent) |
| Half-Life | ~20-30 Minutes | ~20-30 Minutes |
| Primary Research Utility | Cachexia, Gastric Motility, Cytoprotection | Pure GH secretion, Lean Mass Retention |
Desensitization and Synergistic Combinations
A critical limitation of GHRP mono-therapy in research is the phenomenon of tachyphylaxis, or receptor desensitization. The GHS-R1a receptor internalizes rapidly upon constant stimulation. Consequently, research protocols often utilize pulsatile administration schedules rather than continuous exposure.
Furthermore, efficacy is drastically improved when GHRPs are combined with Growth Hormone Releasing Hormone (GHRH) analogues. Peptides such as CJC-1295 or Sermorelin bind to a different receptor (GHRH-R), increasing the number of GH-secreting cells activated, while the GHRP increases the force of that secretion.
Research indicates that the combination of CJC-1295 and GHRP-2 creates a synergistic effect where the total GH release is greater than the sum of the individual parts. This combination is particularly potent for preserving lean tissue because it mimics the body's natural pulsatile rhythm more effectively than bolus GH administration.
Caloric Restriction: The Critical Context
Why is the distinction between GHRP-2 and GHRP-6 so vital during caloric restriction?
The Ghrelin Paradox
During starvation, natural ghrelin levels rise to protect the organism by stimulating hunger and GH release (to mobilize fat for fuel). However, chronic elevation can lead to GH resistance. By introducing exogenous GHRPs, researchers attempt to override this resistance.
If the research subject (animal model) is under strict caloric restriction, the intense hunger pangs induced by GHRP-6 can stimulate significant stress responses (corticosterone in rodents) if food is denied. This stress elevation can be catabolic, potentially negating the anabolic benefits of the peptide. Therefore, GHRP-2 is often cited as the superior tool for lean mass preservation in strictly calorie-controlled models because it provides the GH stimulus with less orexigenic conflict.
Lipolysis and Fat Oxidation
Preserving lean mass is only half the equation; the other is the utilization of adipose tissue. GH is a potent lipolytic agent. It stimulates hormone-sensitive lipase (HSL) to break down triglycerides into free fatty acids. Both GHRP-2 and GHRP-6 facilitate this shift from glucose burning to fat burning, which spares amino acids from being oxidized for energy. This "protein sparing" effect is the fundamental mechanism by which lean mass is preserved.
Safety Profile and Research Considerations
While discussing efficacy, one must address the physiological side effects noted in literature.
Cortisol and Prolactin
Elevated cortisol is catabolic to muscle tissue—it achieves the exact opposite of the researcher's goal. As mentioned, GHRP-2 stimulates more cortisol than GHRP-6. However, studies indicate that this elevation usually remains within the normal diurnal range and does not reach the pathological levels seen in Cushing's syndrome. Nevertheless, for research subjects with pre-existing adrenal sensitivity, Ipamorelin might be considered as an alternative, as it selectively releases GH with almost zero impact on cortisol or prolactin.
Glucose Metabolism
Both peptides can transiently increase blood glucose and reduce insulin sensitivity shortly after administration, a direct effect of high GH levels. Chronic use without cycling can lead to insulin resistance. However, in the context of caloric restriction (where glucose is already scarce), this effect is generally mitigated and the lipolytic benefits take precedence.
Synthesis and Purity in Research
The validity of any study comparing myogenic factors hinges on the purity of the peptides used. Contaminants or degradation products can trigger immunogenic responses or fail to bind receptors correctly, skewing data regarding MyoD or Myogenin expression. At Alpha Carbon Labs, we emphasize the importance of distinct peptide synthesis protocols. Understanding the difference between lyophilized stability and reconstituted degradation is vital for laboratory handling.
For detailed verification of peptide integrity, researchers are encouraged to review COA documents and methodologies to ensure that the GHRP-2 or GHRP-6 employed meets strict HPLC purity standards.
Conclusion: Selecting the Right Tool for Mass Preservation
In the comparative analysis of GHRP-2 and GHRP-6 regarding myogenic regulatory factors and lean mass preservation, the choice depends heavily on the specific parameters of the caloric restriction model.
GHRP-6 serves as a broad-spectrum secretagogue. Its utility is highest in models where increasing food intake is desirable (e.g., combatting cachexia) or where its unique cytoprotective properties are required to mitigate tissue damage from ischemia.
GHRP-2, conversely, stands out as the more precise instrument for maximal GH secretion. Its ability to generate significant GH pulses with reduced appetite stimulation makes it the superior candidate for strictly maintaining lean mass during aggressive dieting or caloric deficits. By driving the IGF-1 axis more potently, it theoretically offers greater upregulation of MyoD and Myogenin and stronger inhibition of the ubiquitin-proteasome system.
Ultimately, both peptides act as powerful signals to the myogenic machinery, shifting the metabolic priority toward protein synthesis and lipid oxidation. Understanding the nuances of their secondary effects allows researchers to fine-tune their protocols for optimal myogenic support.
References
- Bowers, C. Y. (1998). Growth hormone-releasing peptide (GHRP). Cellular and Molecular Life Sciences, 54(10), 1316-1329. DOI: 10.1007/s000180050257
- Laferrère, B., Abraham, C., Russell, C. D., & Bowers, C. Y. (2005). Growth hormone releasing peptide-2 (GHRP-2), like ghrelin, increases food intake in healthy men. The Journal of Clinical Endocrinology & Metabolism, 90(2), 611-614. DOI: 10.1210/jc.2004-1719
- Velloso, C. P. (2008). Regulation of muscle mass by growth hormone and IGF-I. British Journal of Pharmacology, 154(3), 557-568. DOI: 10.1038/bjp.2008.153
- Laron, Z. (2001). Insulin-like growth factor 1 (IGF-1): a growth hormone. Molecular Pathology, 54(5), 311. DOI: 10.1136/mp.54.5.311
- Sigalos, J. T., & Pastuszak, A. W. (2018). The safety and efficacy of growth hormone secretagogues. Sexual Medicine Reviews, 6(1), 45-53. DOI: 10.1016/j.sxmr.2017.02.004
- Granado, M., et al. (2010). Effects of GHRP-2 on the specific growth hormone-releasing factor receptor in the pituitary of the rat. Journal of Endocrinology, 207(2), 221-230. DOI: 10.1677/JOE-10-0226
- Muller, A. F., et al. (2015). Ghrelin and its analogues, GHRP-6 and GHRP-2, interaction with the regulation of growth hormone secretion. Physiological Reviews, 95(4), 1121-1153. DOI: 10.1152/physrev.00011.2015
- Schiaffino, S., & Mammucari, C. (2011). Regulation of skeletal muscle growth by the IGF1-Akt-mTOR pathway. Skeletal Muscle, 1(1), 4. DOI: 10.1186/2044-5040-1-4
- Glass, D. J. (2005). Skeletal muscle hypertrophy and atrophy signaling pathways. The International Journal of Biochemistry & Cell Biology, 37(10), 1974-1984. DOI: 10.1016/j.biocel.2005.04.018
- Sabnis, G. S., et al. (2007). Myogenic regulatory factors: their role in muscle fiber determination and development. Frontiers in Bioscience, 12, 2901-2911. DOI: 10.2741/2281
- Yamamoto, D., et al. (2008). GHRP-2, a synthetic ghrelin agonist, increases food intake and body weight in cachectic mice. Biochemical and Biophysical Research Communications, 369(3), 856-861. DOI: 10.1016/j.bbrc.2008.02.100
References
- 1. Bowers, C. Y. (1998). Growth hormone-releasing peptide (GHRP). Cellular and Molecular Life Sciences, 54(10), 1316-1329.
- 2. Laferrère, B., Abraham, C., Russell, C. D., & Bowers, C. Y. (2005). Growth hormone releasing peptide-2 (GHRP-2), like ghrelin, increases food intake in healthy men. The Journal of Clinical Endocrinology & Metabolism, 90(2), 611-614.
- 3. Velloso, C. P. (2008). Regulation of muscle mass by growth hormone and IGF-I. British Journal of Pharmacology, 154(3), 557-568.
- 4. Laron, Z. (2001). Insulin-like growth factor 1 (IGF-1): a growth hormone. Molecular Pathology, 54(5), 311.
- 5. Sigalos, J. T., & Pastuszak, A. W. (2018). The safety and efficacy of growth hormone secretagogues. Sexual Medicine Reviews, 6(1), 45-53.
- 6. Granado, M., et al. (2010). Effects of GHRP-2 on the specific growth hormone-releasing factor receptor in the pituitary of the rat. Journal of Endocrinology, 207(2), 221-230.
- 7. Muller, A. F., et al. (2015). Ghrelin and its analogues, GHRP-6 and GHRP-2, interaction with the regulation of growth hormone secretion. Physiological Reviews, 95(4), 1121-1153.
- 8. Schiaffino, S., & Mammucari, C. (2011). Regulation of skeletal muscle growth by the IGF1-Akt-mTOR pathway. Skeletal Muscle, 1(1), 4.
- 9. Glass, D. J. (2005). Skeletal muscle hypertrophy and atrophy signaling pathways. The International Journal of Biochemistry & Cell Biology, 37(10), 1974-1984.
- 10. Sabnis, G. S., et al. (2007). Myogenic regulatory factors: their role in muscle fiber determination and development. Frontiers in Bioscience, 12, 2901-2911.
- 11. Yamamoto, D., et al. (2008). GHRP-2, a synthetic ghrelin agonist, increases food intake and body weight in cachectic mice. Biochemical and Biophysical Research Communications, 369(3), 856-861.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.