Mitigating Myocellular Atrophy: Synergistic Research Applications of Semaglutide and Ibutamoren (MK-677)
This deep-dive article explores the synergistic potential of combining Semaglutide and MK-677 to prevent muscle loss during rapid weight reduction. We examine the mechanisms of action, the physiology of myocellular atrophy, and the theoretical framework for using GH secretagogues to preserve lean mass in caloric deficit research models.
Introduction: The Dual-Edge of Therapeutic Caloric Restriction
In the contemporary landscape of metabolic research, the advent of Glucagon-Like Peptide-1 (GLP-1) receptor agonists has fundamentally shifted the paradigm of weight management protocols. Agents such as Semaglutide have demonstrated efficacy in inducing substantial weight loss, often rivaling outcomes seen in bariatric surgery. However, as with any potent intervention affecting systemic energy homeostasis, a critical variable remains the composition of the mass lost. In clinical trials and animal models, rapid weight reduction is frequently accompanied by a reduction in lean body mass (LBM), specifically myocellular atrophy.
The preservation of skeletal muscle tissue is paramount, not merely for locomotion but because muscle acts as a primary reservoir for glucose disposal and a determinant of basal metabolic rate (BMR). The research community is increasingly focused on combinatorial approaches that utilize the lipolytic potency of GLP-1 agonists while simultaneously employing anabolic or anti-catabolic agents to shield muscle tissue. Among the most promising candidates for this synergistic role is MK-677 (Ibutamoren), a non-peptide, orally active agonist of the ghrelin receptor and a growth hormone secretagogue.
This article provides an in-depth analysis of the physiological mechanisms underpinning both compounds, the kinetics of muscle loss during hypophagia, and the rationale for investigating their co-administration. We will explore how regulating the Growth Hormone (GH)/Insulin-Like Growth Factor 1 (IGF-1) axis via MK-677 may serve as a functional counterweight to the catabolic environment induced by Semaglutide-mediated caloric deficits.
Physiological Mechanisms of Action
To understand the potential synergy, one must first dissect the independent pathways through which these research peptides operate. While they ultimately impact body composition, their vectors of action are diametrically opposed in terms of appetite signaling yet complementary in metabolic outcome.
Semaglutide: The Anorexigenic Powerhouse
Semaglutide acts as a long-acting analog of native GLP-1, a gut-derived incretin hormone. Its modification—specifically, the substitution of albumin-binding fatty acid chains—allows for resistance to degradation by dipeptidyl peptidase-4 (DPP-4), extending its half-life significantly compared to endogenous GLP-1.
Research indicates that Semaglutide exerts its effects through both peripheral and central mechanisms:
- Central Nervous System (CNS) Signaling: Semaglutide crosses the blood-brain barrier, interacting with GLP-1 receptors in the hypothalamus (specifically the arcuate nucleus) and the hindbrain. This signaling pathway enhances satiety and diminishes hunger, leading to valid, voluntary hypophagia (reduction in food intake).
- Gastric Motility: By delaying gastric emptying, Semaglutide prolongs the postprandial sensation of fullness, flattening glucose spikes and moderating nutrient absorption rates.
- Glycemic Control: It stimulates glucose-dependent insulin secretion while suppressing glucagon release, optimizing the metabolic handling of carbohydrates.
MK-677 (Ibutamoren): The Anabolic Secretagogue
Ibutamoren distinguishes itself from other Growth Hormone Secretagogues (GHS) like Ipamorelin or GHRP-6 by being a non-peptide small molecule that mimics the action of ghrelin, the "hunger hormone." However, unlike ghrelin, which promotes adiposity, MK-677 utilizes the ghrelin receptor (GHS-R1a) primarily to amplify the signaling of the pituitary gland to release Growth Hormone (GH).
Key physiological outcomes of MK-677 administration in research subjects include:
- Pulsatile GH Release: It acts on the pituitary and hypothalamus to amplify the natural pulsatile secretion of GH without disturbing natural circadian rhythms.
- IGF-1 Elevation: The downstream effect of increased GH is the hepatic production of Insulin-Like Growth Factor 1 (IGF-1), a potent mediator of cellular proliferation, differentiation, and muscle protein synthesis (MPS).
- Nitrogen Retention: Classic studies have demonstrated MK-677's ability to reverse diet-induced nitrogen wasting, a marker of protein catabolism.
- Sustained Bioavailability: As an oral agent, it possesses high bioavailability and a long half-life, maintaining elevated IGF-1 levels over a 24-hour period.
Comparative Mechanisms Table
| Feature | Semaglutide | MK-677 (Ibutamoren) |
|---|---|---|
| Receptor Target | GLP-1 Receptor (Agonist) | GHS-R1a (Ghrelin Receptor Agonist) |
| Primary Metabolic Effect | Lipolysis via caloric deficit | Anabolism via GH/IGF-1 axis |
| Effect on Appetite | Significant suppression (Anorexigenic) | potential increase (Orexigenic) |
| Muscle Tissue | Potential indirect atrophy due to low energy | Promotes hypertrophy and retention |
| Insulin Sensitivity | Increases sensitivity | Transient decrease (competes with insulin) |
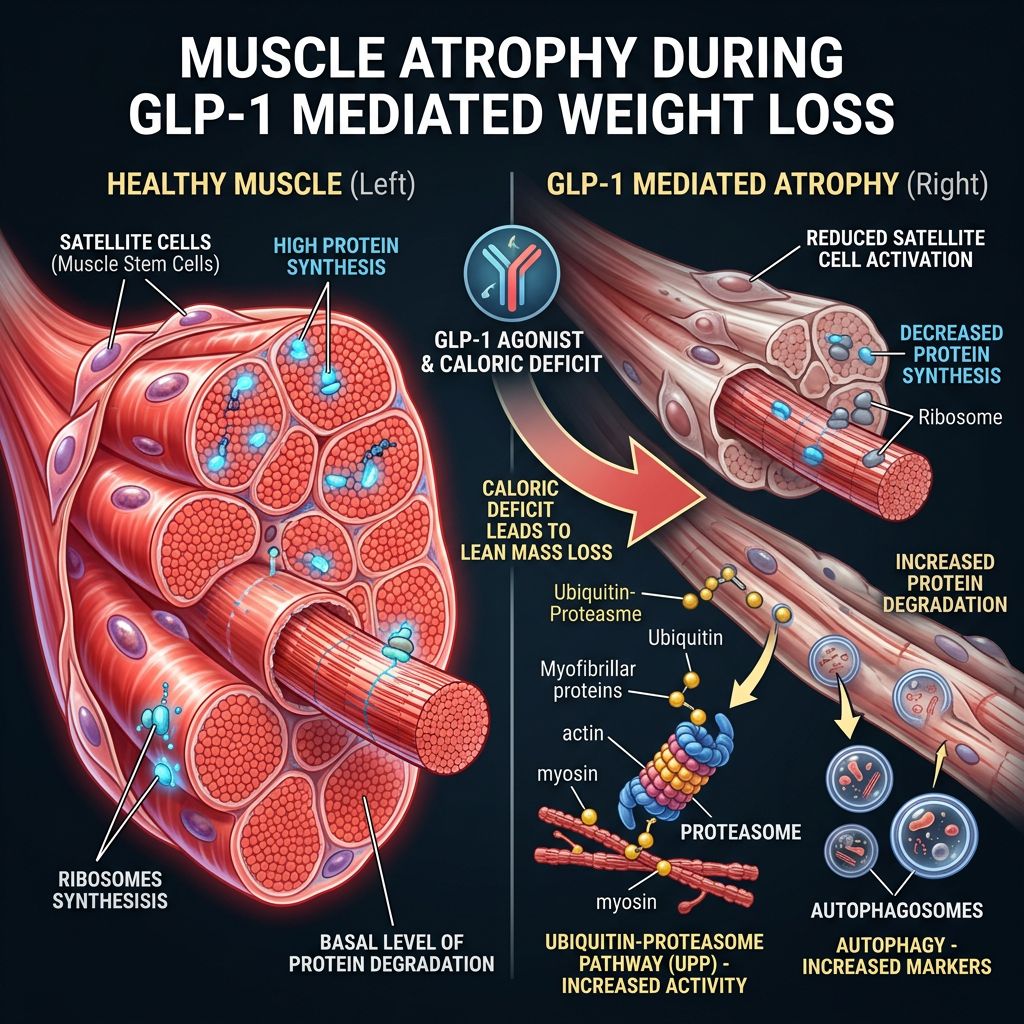
The Physiology of Muscle Atrophy During GLP-1 Mediated Weight Loss
A persistent concern in the deployment of potent GLP-1 agonists is the quality of weight lost. Clinical data from the STEP trials suggested that while Semaglutide results in massive weight reduction, approximately 20-40% of that weight can be attributed to lean body mass, including skeletal muscle and bone density.
The Catabolic Cascade
When an organism is placed in a deep caloric deficit—simulated by the effects of Semaglutide—the body shifts into a catabolic state. Evolutionarily, this is a survival mechanism: in the absence of exogenous amino acids (protein intake), the body catabolizes skeletal muscle to source gluconeogenic precursors (alanine and glutamine) to fuel the brain and essential organs.
Two primary pathways drive this myocellular atrophy:
- Ubiquitin-Proteasome Pathway: This is the principal mechanism for protein degradation in mammalian cells. During negative energy balance, specific E3 ubiquitin ligases (such as Atrogin-1 and MuRF1) tag myofibrillar proteins for destruction.
- Autophagy-Lysosome Pathway: While autophagy is essential for cellular health, excessive upregulation during starvation leads to the bulk degradation of cytosolic components.
Furthermore, hypophagia often leads to reduced insulin signaling in the muscle. Insulin is not only a glucose-regulating hormone but also a potent inhibitor of protein breakdown. Therefore, lower circulating insulin (or reduced signaling frequency due to fewer meals) removes a critical "brake" on muscle catabolism.
The Myostatin Factor
Research suggests that significant weight loss is also correlated with elevated levels of myostatin, a TGF-beta superfamily member that explicitly inhibits muscle growth. The combined environment of low energy availability, reduced mechanical loading (if activity decreases), and hormonal shifts creates a "perfect storm" for sarcopenia. This is where the introduction of a GH secretagogue becomes scientifically compelling.
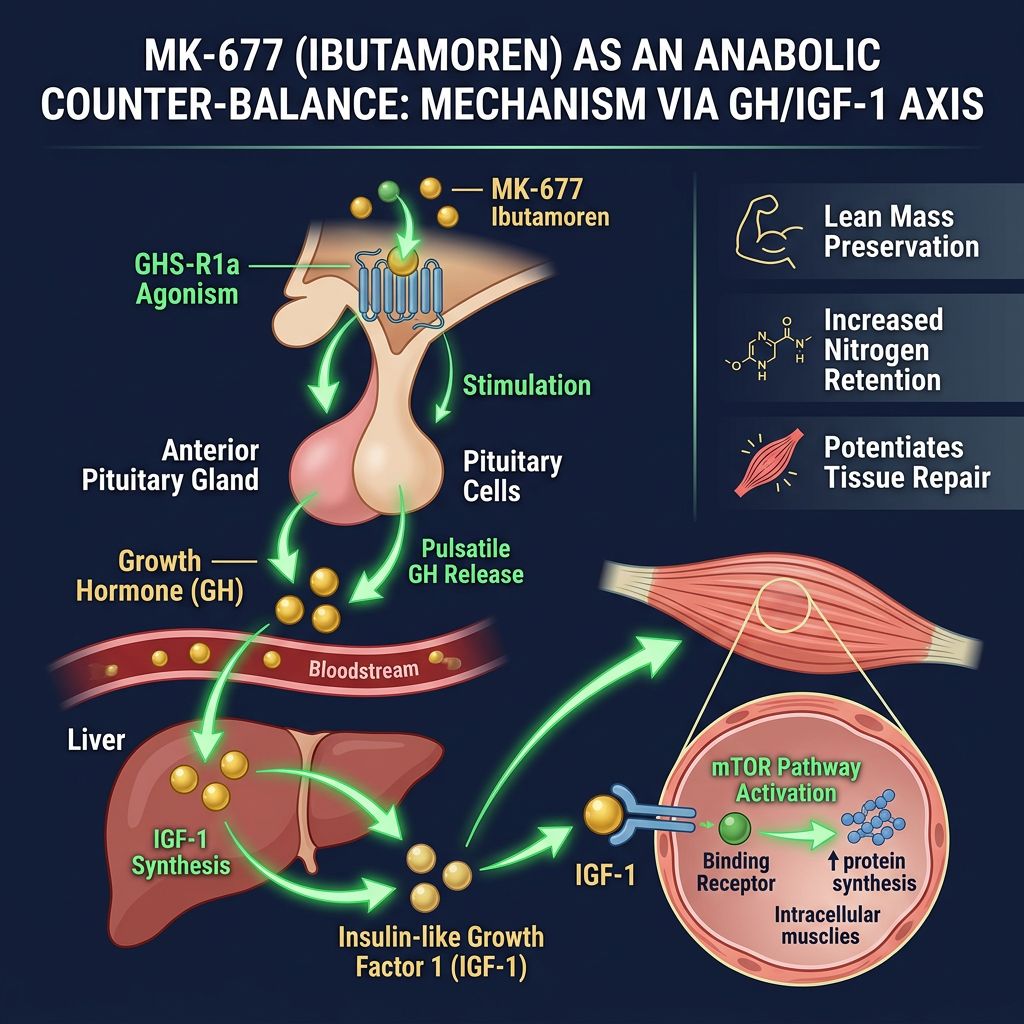
MK-677: The Anabolic Counter-Balance
The rationale for introducing MK-677 into a Semaglutide research protocol relies on its documented ability to uncouple weight loss from nitrogen wasting. Unlike exogenous recombinant HGH, which causes a supraphysiological non-pulsatile spike, MK-677 amplifies the endogenous pulsatile release. This is critical for maintaining receptor sensitivity and avoiding the tachyphylaxis often seen with other agents.
Preservation of Fat-Free Mass (FFM)
Pivotal studies involving MK-677 have focused on catabolic states. In a randomized, controlled trial involving healthy young men subjected to severe caloric restriction, MK-677 reversed diet-induced nitrogen wasting. The subjects receiving the secretagogue maintained restricted caloric intake but ceased to lose lean tissue, indicating a metabolic shift toward utilizing adipose stores exclusively for energy deficits.
Furthermore, in older adults (who naturally exhibit reduced GH secretion, termed "somatopause"), MK-677 treatment over 12 months resulted in significant increases in FFM compared to placebo. The proposed mechanism is direct IGF-1 receptor stimulation on myocytes, which activates the PI3K/Akt/mTOR pathway—the central regulator of protein synthesis.
By activating mTOR, IGF-1 (elevated by MK-677) can directly oppose the action of myostatin and inhibit the transcription of the E3 ubiquitin ligases mentioned earlier. This suggests that MK-677 acts as a molecular "shield," preventing the catabolic machinery from dismantling muscle fibers even when energy availability is low.
Synergistic Research Applications: The "Stack"
The combination of Semaglutide and MK-677 represents a sophisticated approach to body recomposition research. This dual-vector strategy attempts to solve the "Sarcopenic Obesity" dilemma, where subjects have high adiposity but low muscle mass. Treating this phenotype with Semaglutide alone risks exacerbating sarcopenia; treating it with MK-677 alone might not yield sufficient lipolysis given the increased appetite often associated with ghrelin mimetics.
Balancing Appetite Signaling
One of the most intriguing interactions between these two peptides is their effect on appetite.
- Semaglutide: Strongly suppresses appetite.
- MK-677: Can stimulate appetite (ghrelin mimicry), though this effect is often transient and reported to subside after initial weeks of administration.
In a combined protocol, researchers hypothesize that Semaglutide's potent anorexigenic signaling at the CNS level may overpower the orexigenic signal from MK-677. This would theoretically allow for the beneficial GH/IGF-1 elevation of MK-677 without the counterproductive increase in caloric intake. Essentially, the subject remains in a caloric deficit (driven by Semaglutide) but retains the hormonal profile of a well-fed state (driven by MK-677).
Mitigating Metabolic Adaptation
Rapid weight loss invariably leads to "metabolic adaptation" or adaptive thermogenesis—a disproportionate reduction in Resting Energy Expenditure (REE) that fights against further weight loss. This is partly due to the loss of metabolically active tissue (muscle) and partly due to downregulation of thyroid hormone and leptin.
By preserving Lean Body Mass via the varying IGF-1 levels, MK-677 may help maintain REE. Muscle tissue is metabolically expensive; preserving it keeps the metabolic fire burning, preventing the plateau often observed in long-term Semaglutide monotherapy studies.
Hypothetical Study Outcomes: Monotherapy vs. Combination
| Parameter | Semaglutide Monotherapy | MK-677 Monotherapy | Semaglutide + MK-677 Combination |
|---|---|---|---|
| Total Weight Change | Significant Decrease | Neutral / Slight Increase (Water/Muscle) | Moderate-Significant Decrease |
| Fat Mass | Significant Decrease | Slight Decrease | Significant Decrease |
| Lean Body Mass (LBM) | Moderate Decrease | Increase | Preserved or Increased |
| Bone Mineral Density | Potential Decrease | Increase | Maintained / Increased |
| IGF-1 Levels | Unchanged / Bio-unavailable | Significantly Elevated | Elevated |
Comparative Analysis with Other Agents
While the focus here is MK-677, researchers often consider other peptides for muscle preservation. Assessing why MK-677 is frequently chosen over alternatives provides insight into experimental design optimization.
MK-677 vs. GHRPs (Ipamorelin, GHRP-6)
Ipamorelin and GHRP-6 are peptide-based secretagogues requiring subcutaneous injection. They have short half-lives (minutes to hours).
- Convenience in Research: MK-677 is orally active with a 24-hour half-life, simplifying administration protocols in animal models compared to the multiple daily injections required for GHRPs.
- IGF-1 Sustainability: MK-677 produces a sustained elevation in IGF-1, whereas short-acting GHRPs produce spikes. For constant anti-catabolic protection during 24-hour fasting windows, the sustained profile of MK-677 is theoretically superior.
Semaglutide vs. Tirzepatide/Retatrutide
Newer agents like Tirzepatide (dual GLP-1/GIP agonist) and Retatrutide (triple agonist) promote even greater weight loss. As the potency of the weight-loss agent increases, the risk of muscle loss potentially exacerbates. Therefore, the necessity of an agent like MK-677 becomes even more pronounced when researching these multi-agonist peptides.
Safety Profile and Experimental Considerations
Researching the combination of a GLP-1 agonist and a GH secretagogue requires careful attention to safety parameters, particularly glucose homeostasis.
Glucose Homeostasis and Insulin Sensitivity
This is the most complex biological interaction in the stack:
- The Semaglutide Effect: Semaglutide lowers blood glucose and improves insulin sensitivity. It is an anti-diabetic agent.
- The MK-677 Effect: Increased GH secretion can induce transient peripheral insulin resistance. Physiological GH counteracts insulin action to release glucose and fatty acids into the bloodstream.
Investigative query: Does Semaglutide negate the insulin resistance caused by MK-677? Early hypotheses suggest a favorable interaction where Semaglutide improves insulin sensitivity enough to offset the mild resistance induced by GH elevation. However, this necessitates rigorous monitoring of fasting blood glucose and HbA1c in longitudinal studies.
Fluid Retention
MK-677 is known to cause water retention via stimulation of the aldosterone pathway or direct mineralocorticoid receptor cross-talk. Semaglutide, conversely, often exerts a diuretic, natriuretic effect early in treatment. The combination might balance fluid dynamics, but researchers must differentiate between "lean mass gain" (which can be water) and true myofibrillar accretion / nitrogen retention.
Quality of Research Materials
The integrity of data derived from these experiments depends entirely on the purity of the peptides employed. Impurities in peptide synthesis can lead to immunogenic reactions or skewed data due to inconsistent dosing. Researchers are advised to utilize suppliers who provide transparent validation, such as COA Documents demonstrating HPLC and Mass Spectrometry analysis. For a deeper understanding of stringent manufacturing standards, review our Quality Control protocols.
Future Directions in Peptide Research
The horizon of metabolic research suggests a move toward "poly-pharmacy" within single formulations. We are already seeing the mixing of BPC-157 and TB-500 for tissue repair (e.g., BPC-157 + TB-500 Blend). It is plausible that future investigations will explore fixed-dose combinations of GLP-1 analogs and non-peptide secretagogues to create a "Body Recomposition Pill" that strips fat while building muscle simultaneously.
Researchers are also investigating the utility of other mitochondrial peptides like MOTS-c or SS-31 alongside this stack to enhance fatty acid oxidation efficiency or the addition of 5-amino-1mq to target NNMT pathways for further metabolic optimization.
Potential Clinical Implications (Theoretical)
If validated in human trials, the Semaglutide + MK-677 protocol could revolutionize the treatment of:
- Sarcopenic Obesity in the Elderly: Allowing safe weight loss without frailty.
- Post-Bariatric Recovery: Preventing the "skinny fat" phenotype post-surgery.
- Cachexia: In chronic illness where muscle wasting is fatal, but metabolic regulation is also compromised.
Conclusion
The intersection of Glucagon-Like Peptide-1 agonists and Growth Hormone Secretagogues represents one of the most exciting frontiers in metabolic research. By combining the potent, targeted lipolysis of Semaglutide with the nitrogen-retaining, anabolic prowess of MK-677, researchers aim to decouple weight loss from muscle atrophy.
This synergistic approach addresses the primary limitation of current anti-obesity pharmacotherapy—the degradation of lean tissue. Through careful modulation of the GLP-1 and GH/IGF-1 axes, it may be possible to achieve profound shifts in body composition that favor long-term metabolic health and functional longevity. As investigation continues, the importance of using high-purity research compounds, such as those verified by Alpha Carbon Labs' peptide synthesis standards, cannot be overstated in generating reproducible and valid scientific data.
References
- 1. Murphy, K. G., & Bloom, S. R. (2006). Gut hormones and the regulation of energy homeostasis. Nature, 444(7121), 854-859.
- 2. Wilding, J. P. H., et al. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity. The New England Journal of Medicine, 384, 989-1002.
- 3. Svensson, J., et al. (1998). Two-month treatment of obese subjects with the oral growth hormone (GH) secretagogue MK-677 increases GH secretion, fat-free mass, and energy expenditure. The Journal of Clinical Endocrinology & Metabolism, 83(2), 362-369.
- 4. Nass, R., et al. (2008). Effects of an oral ghrelin mimetic on body composition and clinical outcomes in healthy older adults: a randomized, controlled trial. Annals of Internal Medicine, 149(9), 601-611.
- 5. Blundell, J., et al. (2017). Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity. Diabetes, Obesity and Metabolism, 19(9), 1242-1251.
- 6. Copinschi, G., et al. (1996). Effects of a 7-day treatment with a novel, orally active, growth hormone (GH) secretagogue, MK-677, on 24-hour GH profiles, insulin-like growth factor I, and adrenocortical function in normal young men. The Journal of Clinical Endocrinology & Metabolism, 81(7), 2376-2381.
- 7. Sardh, E., et al. (2010). Safety, pharmacokinetics and pharmacodynamics of the oral growth hormone secretagogue MK-677 in healthy older adults. Growth Hormone & IGF Research, 20(2), 160-165.
- 8. Caron, A., et al. (2018). GLP-1 receptor agonists for the treatment of obesity: Role of central and peripheral mechanisms. Diabetes & Metabolism, 44(2), 111-122.
- 9. Clemmensen, C., et al. (2019). Emerging hormonal-based combination pharmacotherapies for the treatment of metabolic diseases. Nature Reviews Endocrinology, 15(2), 90-104.
- 10. Chapman, I. M., et al. (1996). Stimulation of the growth hormone (GH)-insulin-like growth factor I axis by daily oral administration of a GH secretagogue (MK-677) in healthy elderly subjects. The Journal of Clinical Endocrinology & Metabolism, 81(12), 4249-4257.
- 11. Papadopoulou-Marketou, N., et al. (2020). Ghrelin and the Growth Hormone Secretagogue Receptor 1a (GHS-R1a) in the Central Regulation of Energy Balance. Current Medicinal Chemistry, 27(34), 5707-5722.
- 12. McCarthy, D., et al. (2023). Muscle loss with glucagon-like peptide-1 receptor agonists: mechanisms and implications for older adults. Journal of Cachexia, Sarcopenia and Muscle, 14(6), 2820-2826.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.