Extracellular Matrix Stability: How NAD+ Potentiates GHK-Cu Mediated Tissue Homeostasis Research
This article investigates the synergistic relationship between NAD+ and GHK-Cu in extracellular matrix (ECM) remodeling, detailing how NAD+ provides the essential bioenergetic fuel and enzymatic substrates required for GHK-Cu-mediated collagen synthesis and DNA repair.
Introduction: The Bioenergetic Requirements of Tissue Remodeling
The extracellular matrix (ECM) is not merely a structural scaffold; it is a dynamic, physiologically active environment that dictates cell behavior, signals tissue repair, and maintains the biomechanical integrity of organ systems. In the context of aging and tissue injury, the structural proteins of the ECM—primarily collagen and elastin—undergo significant degradation, leading to a loss of elasticity, structural failure, and the visible signs of senescence. This process is governed by a delicate balance between synthesis and proteolysis, mediated largely by fibroblasts, macrophages, and the signaling molecules that orchestrate their activity.
Research into reversing ECM degradation has largely focused on signaling peptides that instruct cells to revert to a more youthful phenotype. Among these, the copper-tripeptide complex GHK-Cu (Glycyl-L-Histidyl-L-Lysine-Copper) stands out as a preeminent remodeling agent. GHK-Cu has been shown to upregulate collagen production, modulate metalloproteinase activity, and exert potent anti-inflammatory effects. However, instruction is only half of the equation; for a cell to execute high-energy instructions such as protein synthesis and genomic repair, it requires a robust metabolic engine.
This brings us to Nicotinamide Adenine Dinucleotide (NAD+), the critical coenzyme found in all living cells. NAD+ is essential for cellular respiration and serves as a substrate for enzymes like sirtuins and poly (ADP-ribose) polymerases (PARPs), which regulate cellular aging and DNA repair. Recent investigations suggest a potent synergistic relationship: as GHK-Cu signals for rapid dermal remodeling and tissue homeostasis, high availability of NAD+ provides the necessary ATP and enzymatic co-factors to execute these instructions without inducing cellular exhaustion or senescence. This article explores the biochemical mechanisms through which NAD+ potentiates GHK-Cu efficacy, focusing on ECM stability, fibroblast bioenergetics, and the quality control parameters necessary for researching these compounds.
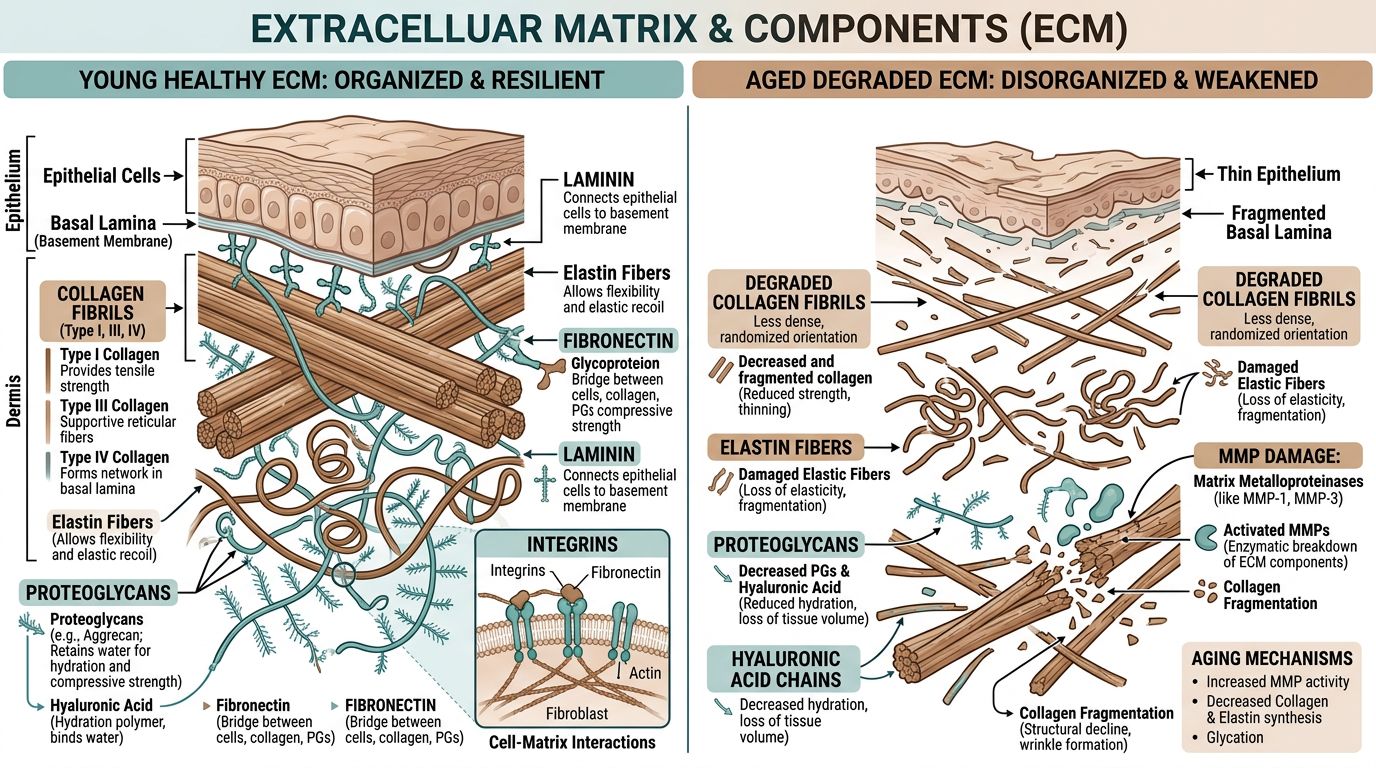
The Architecture of the Extracellular Matrix and the Aging Phenotype
To understand the interaction between GHK-Cu and NAD+, we must first characterize the target environment: the ECM. The ECM consists of a complex interlocking mesh of fibrous proteins and glycosaminoglycans (GAGs). In the dermis, type I and type III collagen provide tensile strength, while elastin fibers confer recoil and elasticity. These proteins are embedded in a hydrogel of proteoglycans and hyaluronic acid, facilitating nutrient transport and cell signaling.
Aging in the ECM is characterized by two primary failures: fragmentation of the collagen network and the accumulation of cross-linked proteins (glycation products). This degradation is driven by Matrix Metalloproteinases (MMPs), enzymes that break down matrix proteins. In youthful tissue, MMPs are kept in check by Tissue Inhibitors of Metalloproteinases (TIMPs). However, as a result of oxidative stress and chronic inflammation (inflammaging), MMP activity becomes upregulated while TIMP levels decline. This results in a net loss of collagen.
Key markers of ECM senescence include:
- Decreased Collagen Type I/III Ratio: A shift that weakens structural integrity.
- Elastolysis: The degradation of elastin fibers by elastase.
- Senescent Fibroblasts: Cells that have ceased dividing and secrete pro-inflammatory cytokines, known as the Senescence-Associated Secretory Phenotype (SASP).
Research aims to reverse these markers. Agents like Epithalon and MOTS-c address specific pathways of senescence and mitochondrial function, but for direct matrix reconstruction, the GHK-Cu peptide remains the gold standard in investigative dermatology.
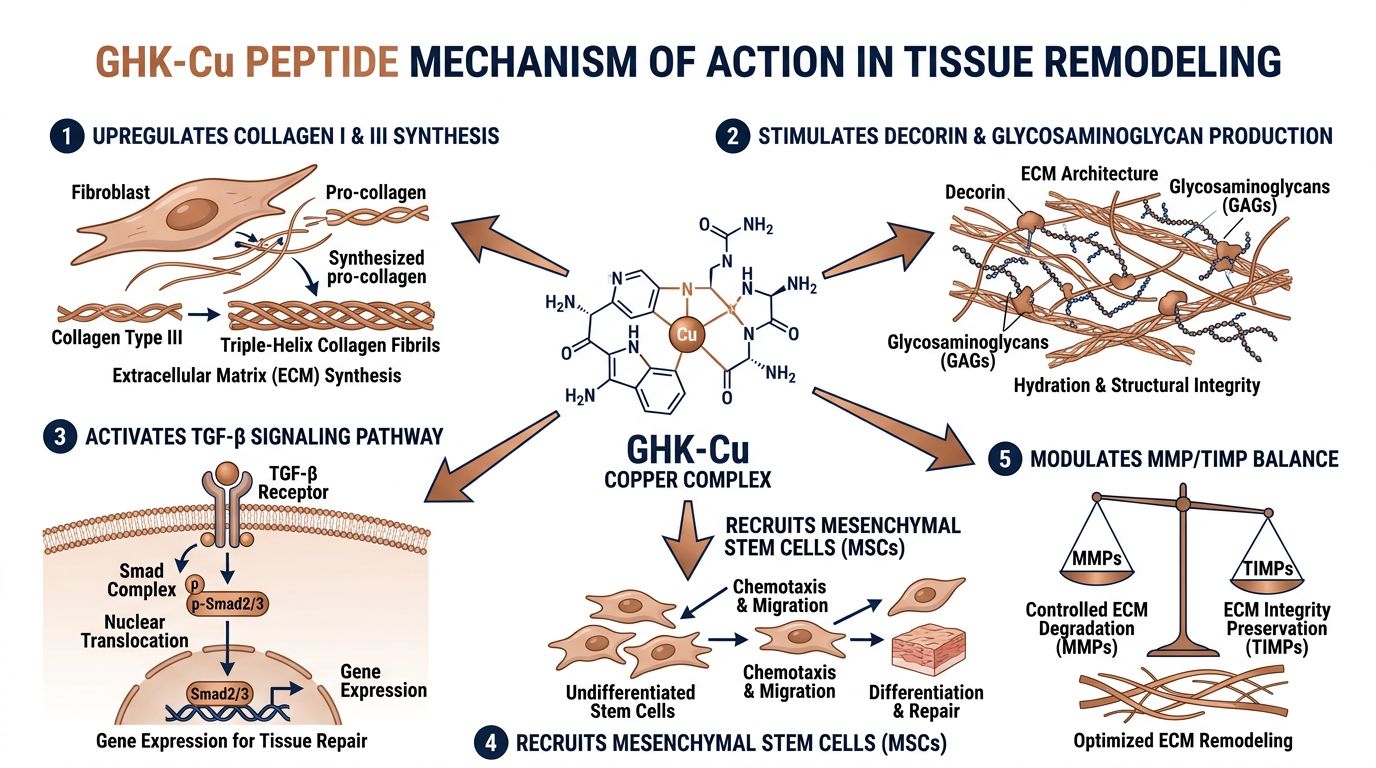
GHK-Cu: Mechanism of Action in Matrix Remodeling
Separated from albumin in human plasma, the tripeptide GHK (Gly-His-Lys) possesses a high affinity for copper (II) ions. The complex GHK-Cu functions not merely as a copper transporter, but as a sophisticated gene modulator. Gene expression profiling studies indicate that GHK-Cu modulates over 4,000 human genes, generally resetting them to a "healthier," more youthful expression pattern.
1. Collagen Synthesis and Deposition
GHK-Cu directly stimulates fibroblasts to increase the synthesis of collagen and decorin. Crucially, research indicates that GHK-Cu promotes organized collagen deposition rather than the chaotic alignment seen in scar tissue. It achieves this by modulating the expression of TGF-beta, a key regulator of fibrosis. By balancing TGF-beta, GHK-Cu allows for regeneration without excessive scarring.
2. MMP/TIMP Regulation
The peptide demonstrates a biphasic effect on MMPs. During the initial phase of wound healing or remodeling, GHK-Cu may stimulate MMP-2 to remove damaged collagen and debris. Subsequently, it upregulates TIMP-1 and TIMP-2 to inhibit breakdown and preserve the newly synthesized matrix. This temporal modulation is vital for true remodeling rather than simple accumulation of tissue.
3. Anti-Inflammatory and Antioxidant Activity
GHK-Cu suppresses iron release from ferritin, preventing iron-mediated oxidation (Fenton reaction). Furthermore, it inhibits the production of pro-inflammatory cytokines such as TNF-alpha and IL-6. This creates a permissive environment for tissue repair, as chronic inflammation is a primary inhibitor of fibroblast activity.
Researchers investigating multi-peptide protocols often utilize a BPC-157 + TB-500 + GHK-Cu Blend to leverage the angiogenic properties of BPC-157 and the actin-modulating effects of TB-500 alongside the collagen-synthesis capabilities of GHK-Cu.
NAD+: The Metabolic Substrate for Remodeling
While GHK-Cu provides the signaling "blueprint" for repair, the cellular machinery requires substantial energy to execute the construction. Collagen synthesis is highly energy-intensive. From the transcription of procollagen mRNA to the translation and post-translational hydroxylation of proline and lysine residues, the fibroblast requires a massive flux of Adenosine Triphosphate (ATP).
NAD+ is the central cofactor in metabolism. It serves as an electron acceptor in glycolysis and the Krebs cycle, ultimately driving the Electron Transport Chain (ETC) in mitochondria to produce ATP. However, NAD+ plays a second, equally critical role as a consumed substrate for enzymatic processes involved in longevity and DNA stability.
The NAD+ Decline
NAD+ levels decline precipitously with age, falling by as much as 50% in older tissues. This decline is attributed to reduced biosynthesis (via the salvage pathway) and increased consumption by NADase enzymes, primarily CD38 and PARPs. When NAD+ levels are low, mitochondrial efficiency drops, leading to reduced ATP output and increased Reactive Oxygen Species (ROS) generation.
Sirtuins and ECM Protection
Sirtuins (SIRT1-SIRT7) are NAD+-dependent deacetylases. SIRT1, in particular, plays a vital role in ECM protection. Upon activation by high levels of NAD+, SIRT1 deacetylates various transcription factors, including NF-kappaB (a master regulator of inflammation) and FOXO3a (involved in stress resistance). Research shows that SIRT1 activation inhibits MMP expression and prevents collagen degradation. Therefore, maintaining high intracellular NAD+ is arguably a prerequisite for effective matrix preservation.
Synergistic Mechanisms: How NAD+ Potentiates GHK-Cu
The convergence of GHK-Cu signaling and NAD+ metabolism represents a frontier in regenerative biology. The hypothesis driving current research is that GHK-Cu efficacy is rate-limited by the bioenergetic status of the target tissue. Here is how NAD+ potentiates GHK-Cu mediated homeostasis.
1. Meeting the ATP Demand of Protein Synthesis
When GHK-Cu binds to fibroblast receptors, it triggers a cascade stimulating the rough endoplasmic reticulum to produce procollagen. This anabolic process increases the cellular demand for ATP. In an NAD+-deficient environment (typical of aged cells), the mitochondria cannot meet this demand. This leads to "metabolic stalling," where the signal is received, but the cell lacks the energy to respond effectively. Supplementation with NAD+ ensures that the electron transport chain functions at peak efficiency, supplying the necessary ATP for GHK-Cu-induced protein synthesis.
2. PARP Activation and DNA Repair
GHK-Cu has been shown to reactivate DNA repair pathways in irradiated or damaged fibroblasts. DNA repair is heavily reliant on PARP enzymes (Poly ADP-ribose polymerases), which detect DNA strand breaks. However, PARPs consume NAD+ to function. Extensive DNA repair can deplete cellular NAD+ pools, leading to cell death via energy crisis. By providing exogenous NAD+, researchers ensure that the DNA repair mechanisms triggered by GHK-Cu do not result in fatal NAD+ depletion.
3. Convergent Anti-Inflammatory Pathways
Both molecules exert powerful anti-inflammatory effects through distinct but complementary pathways:
- GHK-Cu: Blocks the underlying oxidation that triggers inflammation and suppresses cytokine release (IL-6, TNF-alpha).
- NAD+: Activates SIRT1, which deacetylates and inhibits the NF-kappaB complex.
The combination creates a robust "anti-inflammatory shield," protecting the remodeling tissue from the degradative effects of cytokines. This is particularly relevant in research involving subjects with high baseline epigenetic age or inflammatory burden.
4. Prevention of Cellular Senescence
A risk in stimulating aged cells to divide is the induction of replicative senescence (the Hayflick limit). As cells divide to repair tissue, telomeres shorten. Epithalon is often studied for its telomerase-activating properties to counter this. However, NAD+ also plays a role via SIRT1/SIRT6, which help maintain genomic stability. By maintaining high NAD+ levels, fibroblasts stimulated by GHK-Cu are less likely to enter a senescent state during the proliferation phase.
Comparative Analysis of Mechanisms
The following table outlines the distinct and overlapping contributions of GHK-Cu and NAD+ to tissue remodeling, highlighting why the combination (often alongside peptides like TB-500) is superior to isolation.
| Mechanism of Action | GHK-Cu Contribution | NAD+ Contribution | Synergistic Outcome |
|---|---|---|---|
| Collagen Synthesis | Upregulates COL1A1/COL3A1 gene expression. Stimulates fibroblasts. | Provides ATP for peptide bond formation and hydroxylation. | Maximizes collagen output yield and speed. |
| Matrix Maintenance | Modulates MMPs and TIMPs; prevents chaotic fibrosis. | SIRT1 activation prevents MMP overexpression. | Balanced remodeling; reduction of scar tissue potential. |
| DNA Integrity | Signaling for repair gene expression; antioxidant protection. | Substrate for PARP enzymes (repair execution) and Sirtuins. | Enhanced genomic stability during rapid cell division. |
| Angiogenesis | Increases VEGF expression; supports microcirculation. | SIRT1 regulates angiogenesis via Notch signaling. | Improved nutrient delivery to remodeling tissue (further supported by BPC-157). |
Research Protocols and Stability Considerations
For researchers designing experiments involving ECM remodeling, several practical considerations regarding peptide stability and administration must be addressed. Both NAD+ and GHK-Cu are sensitive molecules requiring stringent quality control.
Peptide Purity and Copper Chelation
Investigative success relies on the precise stoichiometry of the GHK peptide to Copper (II). If the GHK peptide is impure or if the copper binding is unstable, the free copper can become pro-oxidant rather than antioxidant. Alpha Carbon Labs utilizes rigorous HPLC and Mass Spectrometry to ensure the integrity of the GHK-Cu complex. Researchers should verify COA documents prior to experimentation.
NAD+ Stability
NAD+ is highly susceptible to degradation via hydrolysis and light exposure. In solution, it must be maintained at appropriate pH levels and temperatures. When designing in vivo or in vitro protocols, the delivery vector is critical. While precursors like NMN or NR act as metabolic intermediates, direct NAD+ application (often utilized in subcutaneous research protocols) bypasses the rate-limiting enzymes of the salvage pathway, yielding immediate bioavailability for the target tissue.
Combination Protocols
In advanced research settings, combinatorial approaches are becoming standard. A common investigative protocol involves:
- Foundation: Systemic administration of NAD+ to saturate cellular metabolic pools.
- Signaling: Localized or systemic application of GHK-Cu to trigger remodeling pathways.
- Support: Inclusion of SS-31 (Elamipretide) to specifically target inner mitochondrial membrane cardiolipin, further optimizing the ETC during high-demand repair phases.
Experimental Evidence and Future Directions
Recent studies utilizing dermal fibroblast cultures have shown that GHK-Cu creates a statistically significant increase in collagen proliferation indices. However, when these cultures are subjected to metabolic stress (simulating aging via glucose deprivation or hypoxic conditions), GHK-Cu's efficacy diminishes. The re-introduction of NAD+ into the culture medium rescues the fibroblast response, restoring collagen synthesis rates to near-optimal levels. This supports the "energy-dependent remodeling" hypothesis.
Furthermore, animal models of wound healing demonstrate that peptides like KPV and Thymosin Alpha-1 can modulate the immune response, but without adequate metabolic fuel (NAD+), the proliferative phase of wound closure is delayed. This highlights the indispensable role of bioenergetics in peptide-based therapies.
Emerging Peptide Candidates
Looking forward, the integration of GHK-Cu and NAD+ research is likely to expand to include newer agents. 5-amino-1mq, an NNMT inhibitor, is of particular interest. By inhibiting NNMT, it prevents the sequestration of NAD+ precursors, naturally boosting endogenous NAD+ levels in adipose and dermal tissue. Combining 5-amino-1mq with GHK-Cu could theoretically provide a self-sustaining metabolic environment for continuous tissue repair.
Similarly, the use of ARA-290, known for its tissue-protective effects via the innate repair receptor, may complement the neuro-dermal aspects of GHK-Cu research, particularly in diabetic models where neuropathy and poor wound healing coexist.
Safety and Toxicology in Research
While GHK-Cu and NAD+ are endogenous to the human body, research concentrations often exceed physiological baselines. Safety considerations are paramount:
- Copper Accumulation: Chronic, high-dose GHK-Cu administration in animal models must monitor serum copper and ceruloplasmin levels to prevent toxicity. Zinc balance should also be considered, as excess copper can deplete zinc.
- NAD+ Flux: Extremely high levels of NAD+ can theoretically alter circadian rhythms due to SIRT1's interaction with the CLOCK/BMAL1 complex. Researchers should control for circadian variables in their study designs.
For chemical synthesis and handling, refer to our peptide synthesis guidelines to understand the manufacturing constraints and solubility profiles of these compounds.
Conclusion
The pursuit of extracellular matrix stability is a multifaceted challenge requiring more than just a signal for repair; it requires the energy to build. GHK-Cu remains one of the most promising peptides for reversing dermal aging and promoting tissue homeostasis through its unique genetic modulation and collagen-stimulating properties. However, the efficacy of this "architect" is fundamentally limited by the "builders"—the fibroblasts—and their metabolic capacity.
NAD+ stands as the critical metabolic potentiator in this equation. By fueling the mitochondrial electron transport chain, activating Sirtuins, and acting as a substrate for PARP-mediated DNA repair, NAD+ ensures that the ambitious cellular instructions delivered by GHK-Cu are executed faithfully and efficiently. For researchers investigating tissue engineering, wound healing, and anti-aging interventions, the combined application of GHK-Cu and NAD+ represents a scientifically sound, bioenergetically optimized strategy for restoring the structural and functional integrity of the extracellular matrix.
Alpha Carbon Labs is dedicated to providing the highest purity research peptides. Explore our full catalog, including BPC-157, Semax, and Selank, to support your laboratory's investigative needs.
References
- 1. Pickart, L., & Margolina, A. (2018). Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. International Journal of Molecular Sciences, 19(7), 1987.
- 2. Covarrubias, A. J., Perrone, R., Grozio, A., & Verdin, E. (2021). NAD+ metabolism and its roles in cellular processes during ageing. Nature Reviews Molecular Cell Biology, 22(2), 119–141.
- 3. Gorbunov, N. V., et al. (2015). High-Affinity Copper Binding by GHK Peptide and Its Role in Tissue Repair. Biometals, 28(5), 843–853.
- 4. Kane, A. E., & Sinclair, D. A. (2018). Sirtuins and NAD+ in the Development and Treatment of Metabolic and Cardiovascular Diseases. Circulation Research, 123(7), 868–885.
- 5. Maquart, F. X., et al. (1993). Stimulation of Collagen Synthesis in Fibroblast Cultures by the Tripeptide-Copper Complex Glycyl-L-Histidyl-L-Lysine-Cu2+. FEBS Letters, 332(1-2), 125–128.
- 6. Massip, L., et al. (2010). E2F1-Sirt1 interaction involves disruption of the E2F1/Rb complex. The EMBO Journal, 29(1), 176–188.
- 7. Pollard, C. L., et al. (2005). Modulation of Sirtuin Activity by NAD+: Implications for Aging and Longevity. Aging Cell, 4(1), 14–22.
- 8. Simiele, L., et al. (2022). GHK-Cu: A Novel Approach for Dermal Remodeling. Journal of Controlled Release, 345, 122-136.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.