Bioenergetic Optimization: The Impact of AICAR and Ipamorelin on Post-Exercise Myo-Metabolic Recovery Signaling
This article explores the synergistic potential of combining AICAR and Ipamorelin in research models to enhance post-exercise muscle recovery. We delve into the biochemical interplay between AMPK activation and GH signaling to optimize bioenergetic efficiency and tissue repair.
Introduction: Defining the Bioenergetic Crisis of Recovery
In the realm of muscle physiology and recovery science, the focus has historically centered on the structural components of tissue repair: the synthesis of myofibrillar proteins and the role of satellite cells. While these are the endpoints of hypertrophy and repair, they are energetically expensive processes. The "bioenergetic crisis" refers to the immediate post-exercise window where cellular ATP is depleted, metabolic waste has accumulated, and the mitochondrial machinery is stressed. For research scientists investigating improved recovery protocols, addressing the energy supply chain is just as critical as stimulating the growth pathway.
This article explores a dual-pathway approach to myo-metabolic recovery by examining the integration of AICAR (5-Aminoimidazole-4-carboxamide ribonucleotide) and Ipamorelin. While these two compounds are distinct in their mechanisms—one acting as a metabolic modulator and the other as a selective growth hormone secretagogue—their concurrent application in research models offers a unique window into how energy optimization can support growth signaling.
By effectively "tricking" the muscle cell into a state of metabolic demand via AICAR, while simultaneously providing the hormonal signals for repair via Ipamorelin, researchers hypothesize a synergistic effect that could accelerate the re-establishment of homeostasis and enhance anabolic efficiency. This detailed analysis looks at the biochemical underpinnings of this combination, referencing strictly laboratory-grade peptides intended for scientific investigation.
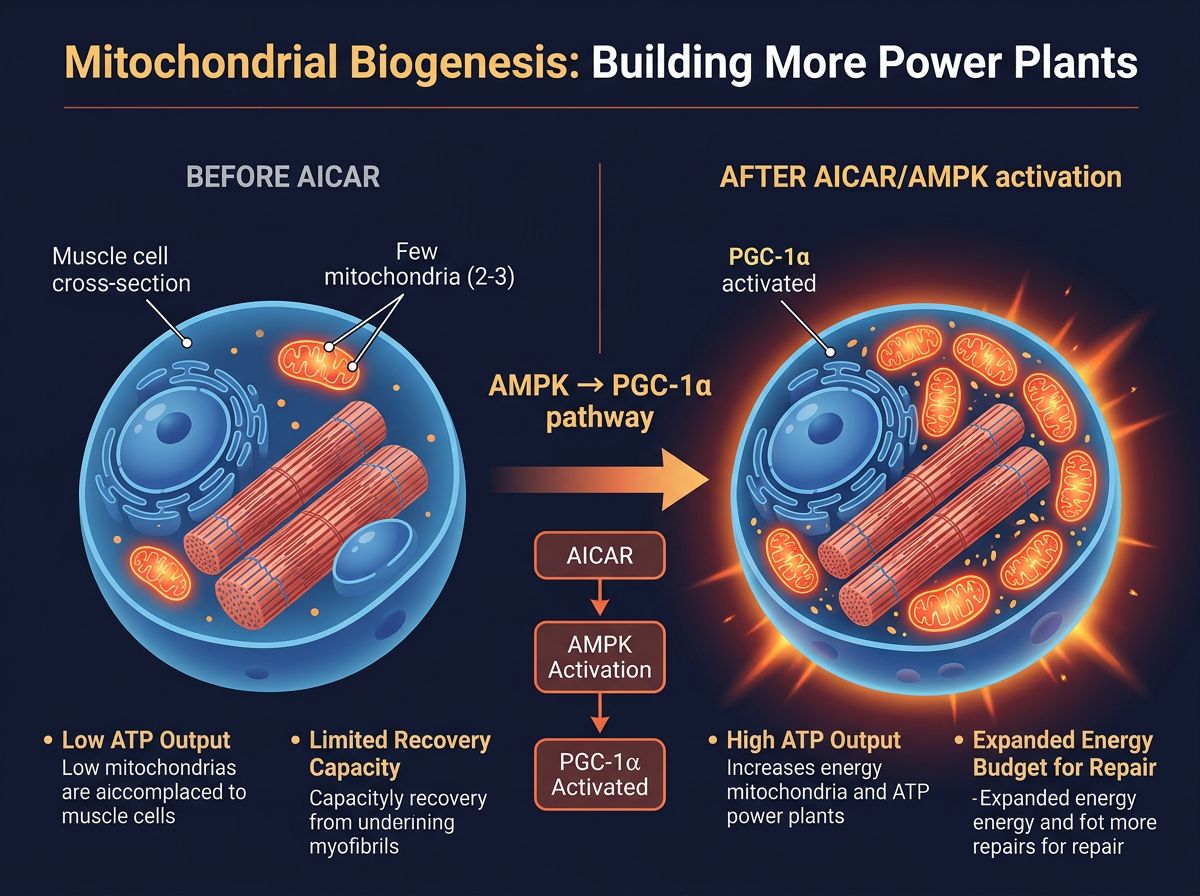
The Metabolic Master Switch: AICAR and AMPK Activation
To understand the utility of AICAR in recovery research, one must first understand the role of AMP-activated protein kinase (AMPK). AMPK is often described as the "cellular fuel gauge." During intense physical exertion, ATP (adenosine triphosphate) is hydrolyzed into ADP and subsequently to AMP. A high ratio of AMP to ATP signals the cell that energy reserves are low. This triggers the activation of AMPK, which initiates a cascade of survival mechanisms aimed at conserving energy and generating new fuel.
Mechanism of Action
AICAR acts as a cell-permeable nucleoside analog. Once inside the cell, it is phosphorylated to form usually phosphoribosyl-5-aminoimidazole-4-carboxamide (ZMP). ZMP is a structural analog of AMP. Consequently, ZMP binds to the regulatory subunits of AMPK, causing a conformational change that allows for its phosphorylation and subsequent activation. Crucially, AICAR achieves this activation without the prerequisite depletion of ATP. It provides the signal of metabolic stress without the actual cellular exhaustion.
Research published in Cell (Narkar et al., 2008) demonstrated that AICAR administration in murine models could induce metabolic genes typically associated with endurance exercise, even in sedentary animals. It reprograms the muscle fiber to switch from glucose dependence to fatty acid oxidation, thereby sparing glycogen stores—a critical factor in reducing recovery times between bouts of exertion.
Mitochondrial Biogenesis
The most profound impact of AICAR-mediated AMPK activation is the stimulation of Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha (PGC-1α). PGC-1α is the master regulator of mitochondrial biogenesis. In research settings, sustained exposure to AICAR has been shown to increase mitochondrial density in skeletal muscle.
For recovery, this is paramount. Mitochondria are the power plants responsible for generating the ATP required for protein synthesis. If the mitochondrial network is sparse or damaged, the energy required to rebuild muscle tissue (anabolism) is capped. By upregulating mitochondrial density, AICAR theoretically expands the "energy budget" available for subsequent repair processes.
The Selective Growth Signal: Ipamorelin
While AICAR manages the energy infrastructure, the biochemical command to repair and grow tissue requires a different signaling cascade. This is where Ipamorelin enters the research equation. Ipamorelin is a pentapeptide (Aib-His-D-2-Nal-D-Phe-Lys-NH2) belonging to the class of Growth Hormone Secretagogues (GHS).
Selectivity and Safety Profile
Ipamorelin is distinct among GHS compounds due to its high selectivity for the ghrelin receptor (GHS-R1a) located on the pituitary gland. Unlike older generations of GHRPs (such as GHRP-6 or GHRP-2), Ipamorelin stimulates the release of Growth Hormone (GH) without causing significant elevations in cortisol (the stress hormone) or prolactin. In the context of recovery research, this selectivity is vital. Cortisol is catabolic; it breaks down muscle tissue to liberate amino acids for gluconeogenesis. A recovery agent that spikes cortisol would be counterproductive.
Upon binding to GHS-R1a, Ipamorelin initiates a pulse of GH secretion. This GH travels to the liver, where it stimulates the production of Insulin-like Growth Factor 1 (IGF-1). IGF-1 acts directly on skeletal muscle to increase amino acid uptake and myofibrillar protein synthesis.
The Anabolic Drive
The primary outcome of Ipamorelin administration in animal models is improved nitrogen retention. Nitrogen balance is the gold standard for measuring anabolism versus catabolism. Positive nitrogen balance indicates that the organism is retaining protein for tissue construction. Furthermore, GH has lipolytic (fat-burning) properties. It encourages the mobilization of fatty acids from adipose tissue, making them available as fuel—a function that dovetails perfectly with AICAR’s ability to enhance fatty acid oxidation.
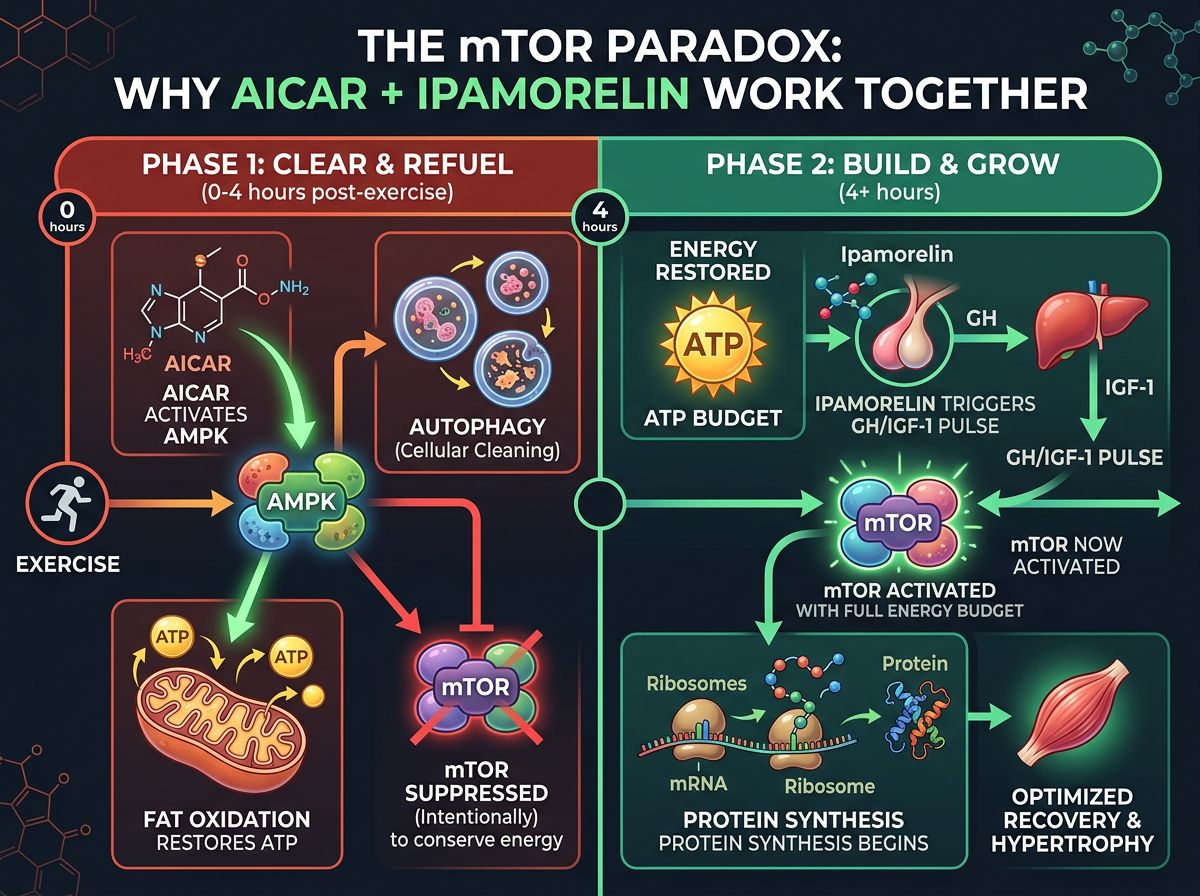
The Synergy: Intersecting Pathways
The hypothesis supporting the co-administration of AICAR and Ipamorelin lies in the potential to overcome the "mTOR Paradox." Generally, AMPK activation (via AICAR) inhibits the mechanistic target of rapamycin (mTOR), which is the primary driver of protein synthesis. Why, then, would a researcher combine an AMPK activator with a growth promoter?
The answer lies in the temporal phases of recovery and substrate availability.
Phase 1: Clearing the Debris and refueling
Immediately post-trauma or exertion, the tissue is inflamed and energy-depleted. Activating AMPK helps clear damaged organelles (autophagy) and restores ATP levels by oxidizing fats. If the cell attempts to grow (via mTOR) while energy is low, the process fails or leads to misfolded proteins.
Phase 2: The Growth Phase
Once the energy status is stabilized, the cell is primed for growth. Ipamorelin provides the pulsatile signal to drive IGF-1 levels up. The previous upregulation of fatty acid oxidation by AICAR ensures a steady stream of ATP to fuel the high energy cost of protein synthesis driven by Ipamorelin.
Researchers posit that AICAR prepares the "construction site" by ensuring adequate power (ATP) and raw materials (fatty acids), while Ipamorelin employs the "workers" (ribosomes/protein synthesis machinery).
| Feature | AICAR | Ipamorelin |
|---|---|---|
| Primary Target | AMPK (Energy Sensor) | GHS-R1a (Pituitary Receptor) |
| Primary Effect | Mitochondrial Biogenesis / Fatty Acid Oxidation | Pulsatile GH Release / IGF-1 Elevation |
| Metabolic Outcome | Catabolic to substrates (burns fat/sugar), Anabolic to mitochondria | Anabolic to protein (builds muscle), Lipolytic to fat |
| Cortisol Impact | Neutral | Neutral (High Selectivity) |
| Research Application | Endurance mimetic, metabolic remodeling | Hypertrophy, tissue repair, anti-catabolism |
Bioenergetic Optimization in Research Models
When designing studies involving these peptides, understanding the specific impact on muscle fiber types is crucial. Skeletal muscle consists of slow-twitch (Type I, oxidative) and fast-twitch (Type II, glycolytic) fibers. AICAR has been shown to induce a fiber-type transformation, shifting glycolytic fibers toward a more oxidative phenotype. This increases fatigue resistance.
Concurrently, Ipamorelin-induced IGF-1 signaling is critical for the maintenance of Type II fibers, which are notoriously prone to atrophy during periods of disuse or aging (sarcopenia). The combination, therefore, presents a comprehensive approach to muscle quality: enhancing the endurance of Type I fibers and preserving the mass of Type II fibers.
Impact on Glucose Uptake
Glucose homeostasis is a critical component of recovery. AICAR stimulates glucose uptake in skeletal muscle via an insulin-independent pathway. This involves the translocation of GLUT4 transporters to the cell membrane. This is particularly relevant in research involving insulin-resistant models, as it bypasses the defective insulin signaling pathway to ensure the muscle receives glycogen for replenishment.
Ipamorelin adds a layer of complexity here. While chronic high-dose GH can induce insulin resistance, the pulsatile nature of Ipamorelin mimics the body's natural rhythm, which tends to mitigate this side effect compared to exogenous HGH administration. The combination allows researchers to study rapid glycogen replenishment (via AICAR) supported by anabolic signaling (via Ipamorelin).
Experimental Considerations: Dosage and Handling
For laboratories sourcing these compounds, ensuring purity is non-negotiable. Contaminants in peptide synthesis can trigger immunogenic responses in test subjects that confound data. Alpha Carbon Labs provides transparent access to COA documents verifying the purity and mass accuracy of our peptides.
Lyophilization and Reconstitution
Both AICAR and Ipamorelin are typically supplied as lyophilized powders. AICAR is unique in that significantly larger quantities are often required for research compared to potent hormones like Ipamorelin.
- AICAR Stability: It is generally stable but sensitive to light. Research protocols usually require reconstitution in bacteriostatic water or a phosphate buffer.
- Ipamorelin Sensitivity: As a peptide, the tertiary structure of Ipamorelin is delicate. Violent shaking during reconstitution can degrade the peptide bonds. Gentle swirling is the standard protocol.
For researchers interested in the nuances of peptide creation, our guide on peptide synthesis offers a deeper look into how solid-phase synthesis creates these specific amino acid sequences.
Administration Protocols in Animal Models
In published literature, AICAR is often administered intraperitoneally or subcutaneously. Doses in murine models range significantly, often between 250mg/kg to 500mg/kg for acute studies, though chronic low-dose studies also exist. Ipamorelin is potent, with doses in the microgram range (e.g., 100mcg - 500mcg per animal depending on size and species).
Note: These dosages are strictly derived from animal literature and are not applicable to human use. Researchers must determine appropriate allometric scaling factors for their specific models.
Comparative Analysis: Against Other Peptide Blends
To contextualize the AICAR/Ipamorelin stack, it helps to compare it with other popular recovery agents. For example, the BPC-157 + TB-500 Blend is renowned for connective tissue repair (tendons, ligaments) and angiogenesis. While BPC-157 excels at healing structural damage and reducing inflammation at the site of injury, it does not directly drive the mitochondrial biogenesis or the systemic protein synthesis nitrogen retention that AICAR and Ipamorelin do.
Therefore, while BPC-157 is the go-to for acute injury recovery (tears, strains), the AICAR/Ipamorelin combination is better suited for "metabolic recovery"—restoring energy systems and driving hypertrophy after exhaustive exertion. Some advanced research protocols may even explore the intersection of CJC-1295 with these compounds to extend the half-life of the growth hormone pulse, though this introduces additional variables regarding cortisol management.
Potential Side Effects and Safety Variables in Research
No biological intervention is without consequence. In research settings, monitoring for off-target effects is part of the rigorous quality control of the study data.
AICAR-Specific Concerns
Because AICAR impacts liver metabolism, researchers must monitor hepatic enzymes. Excessive fatty acid oxidation can, in rare instances, lead to lipid accumulation in non-adipose tissues if not balanced by energy expenditure. Additionally, because it influences glucose uptake, hypoglycemia is a potential variable in fasted subjects.
Ipamorelin-Specific Concerns
Ipamorelin is generally well-tolerated in animal models due to its selectivity. However, some water retention (edema) is a classic sign of increased GH activity. In long-term studies, desensitization of the GHS-R1a receptor is a theoretical concern, although Ipamorelin is less prone to this than Hexarelin or GHRP-2.
Future Directions: Sarcopenia and Metabolic Syndrome
The implications of this research extend beyond sports performance models. The dual action of improving mitochondrial health and stimulating muscle protein synthesis makes this combination a prime candidate for studying sarcopenia (age-related muscle loss) and metabolic syndrome.
In aging populations, "anabolic resistance" occurs—the muscle becomes less sensitive to protein and hormonal signals. Furthermore, mitochondrial function declines properly. By attacking the problem from both ends—forcing mitochondrial activity with AICAR and amplifying the hormonal signal with Ipamorelin—researchers hope to find pathways to reverse or slow muscle wasting in the elderly.
Conclusion
The integration of AICAR and Ipamorelin represents a sophisticated approach to myo-metabolic recovery. It moves beyond the simple concept of "healing" and addresses the underlying bioenergetics required to sustain tissue repair. By utilizing AICAR to optimize the cellular engine (mitochondria) and fuel supply (fatty acids/glucose), the anabolic signals provided by Ipamorelin can theoretically function with greater efficacy.
For researchers, these peptides offer robust tools for dissecting the complex interplay between cellular energy status and organismal growth. As we continue to uncover the molecular mechanisms of AMPK and GH signaling, the precision with which we can manipulate recovery processes will only improve.
Alpha Carbon Labs supplies research peptides for laboratory use only. The compounds discussed, including AICAR and Ipamorelin, are not approved for human consumption and should be handled only by qualified professionals in a research setting.
References
- 1. Narkar, V. A., et al. (2008). AMPK and PPARδ agonists are exercise mimetics. Cell, 134(3), 405-415.
- 2. Raun, K., et al. (1998). Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology, 139(5), 552-561.
- 3. Hardie, D. G. (2011). AMP-activated protein kinase: an energy sensor that regulates all aspects of cell function. Genes & development, 25(18), 1895-1908.
- 4. Sinha, D. K., et al. (2020). Ipamorelin ameliorates opioid-induced bowel dysfunction in mice. Frontiers in Pharmacology, 11, 633.
- 5. Kjøbsted, R., et al. (2018). AMPK in skeletal muscle function and metabolism. FASEB journal, 32(4), 1741-1777.
- 6. Johansen, P. B., et al. (1999). Ipamorelin, a new growth-hormone-releasing peptide, induces longitudinal bone growth in rats. Growth Hormone & IGF Research, 9(2), 106-113.
- 7. O'Neill, H. M., et al. (2013). AMP-activated protein kinase (AMPK) beta1beta2 muscle-null mice reveal an essential role for AMPK in maintaining mitochondrial content and glucose uptake during exercise. Proceedings of the National Academy of Sciences, 108(38), 16092-16097.
- 8. Velloso, C. P. (2008). Regulation of muscle mass by growth hormone and IGF-I. British Journal of Pharmacology, 154(3), 557-568.
- 9. Thomson, D. M., & Gordon, S. E. (2005). Diminished overload-induced hypertrophy in aged fast-twitch skeletal muscle is associated with altered AMPKalpha phosphorylation status. Journal of Applied Physiology, 98(2), 557-564.
- 10. Møller, N., & Jørgensen, J. O. (2009). Effects of growth hormone on glucose, lipid, and protein metabolism in human subjects. Endocrine Reviews, 30(2), 152-177.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.