Verifying Chemical Integrity: A Researcher’s Guide to Sourcing Retatrutide for Sale
Discover the scientific framework for verifying Retatrutide's chemical integrity. This guide covers triple-agonist mechanisms, HPLC/MS verification, and the nuances of GIP, GLP-1, and Glucagon receptor binding.
Introduction: The Evolution of Incretin Research
The landscape of metabolic research has undergone a paradigm shift over the last decade. Originally focused on mono-agonist therapies like Semaglutide, which targets the Glucagon-Like Peptide-1 (GLP-1) receptor, the scientific community has moved rapidly toward multi-receptor agonism. This progression led to the development of dual-agonists like Tirzepatide, and more recently, the "triple-agonist" known as Retatrutide.
For researchers sourcing Retatrutide for sale in a laboratory context, understanding the chemical nuances of this molecule is paramount. Retatrutide (LY3437172) represents a sophisticated engineering feat—a single peptide chain capable of binding and activating three distinct G-protein coupled receptors (GPCRs): the GLP-1 receptor, the Glucose-dependent Insulinotropic Polypeptide (GIP) receptor, and the Glucagon receptor (GCGR). Verifying the chemical integrity of this compound is not merely a matter of checking a box; it is the foundation of reproducible data and valid scientific conclusions.
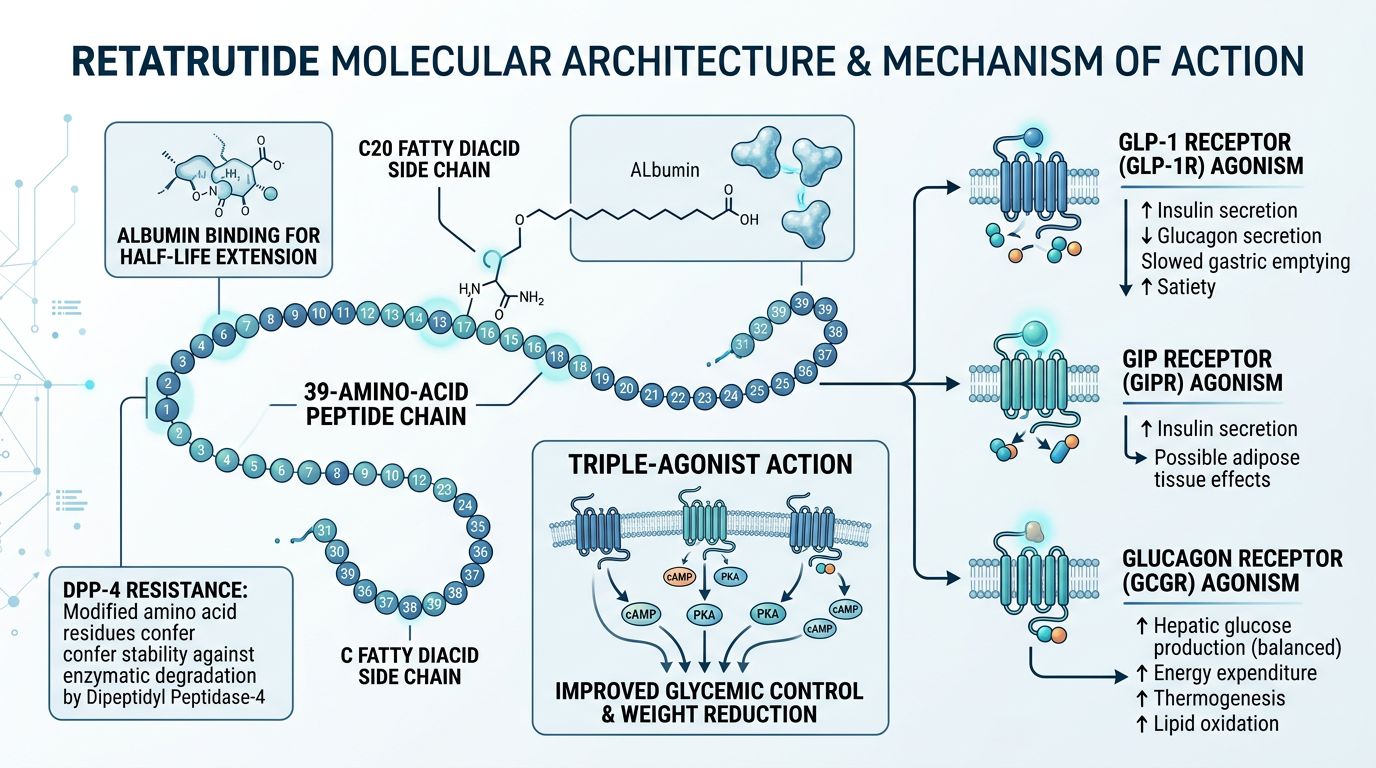
The Molecular Architecture of Retatrutide
Retatrutide is a synthetic 39-amino acid peptide. Its backbone is based on the GIP sequence but is heavily modified to provide the necessary affinity for GLP-1 and Glucagon receptors. These modifications are critical for its pharmacokinetic profile and its biological potency.
Sequence Modifications and Chimerism
In peptide synthesis, the sequence of amino acids dictates the secondary and tertiary structures. Retatrutide incorporates specific non-natural amino acids derived from the Aib (alpha-aminoisobutyric acid) family. These substitutions serve two purposes: they enhance resistance to dipeptidyl peptidase-4 (DPP-4) enzymatic degradation and ensure the peptide adopts the correct alpha-helical conformation required for receptor binding.
Furthermore, the molecule features a C18 fatty acid diacid moiety attached via a linker at position 17 (Lysine). This lipid tail allows for albumin binding, which significantly extends the half-life of the peptide, a feature essential for longitudinal research studies. When a researcher searches for Retatrutide for sale, the presence and purity of this acylation must be confirmed via LC-MS (Liquid Chromatography-Mass Spectrometry).
Ligand-Receptor Binding: The Triple Agonist Mechanism
The primary value of Retatrutide in a research setting lies in its "nutrient-stimulated hormone" mimicry. By engaging three pathways simultaneously, it offers a synergistic approach to energy homeostasis that single agonists cannot match.
1. GIP Receptor Agonism
The GIP component of Retatrutide is often considered the "foundation" of the molecule. GIP receptors are found in both the pancreas and adipose tissue. In the presence of glucose, GIP enhances insulin secretion; however, its role in lipid metabolism and potentially buffering the nausea associated with GLP-1 agonism makes it a focal point of current obesity research.
2. GLP-1 Receptor Agonism
GLP-1 agonism is the most well-documented pathway in modern metabolic science. It stimulates glucose-dependent insulin secretion, inhibits glucagon secretion, and slows gastric emptying. In various preclinical trials, the GLP-1 component contributes heavily to the potent anorectic effects observed in animal models.
3. Glucagon Receptor (GCGR) Agonism
The inclusion of glucagon agonism is what separates Retatrutide from dual agonists like Mazdutide or Tirzepatide. Traditionally, glucagon was viewed as a glucose-elevating hormone. However, at the right ratios, GCGR activation increases energy expenditure and promotes thermogenesis in adipose tissue. This "triple-hit" allows for significant reductions in body mass without the metabolic slowdown often seen in calorie-restricted models.
Comparative Receptor Affinity
The following table illustrates the relative potency of Retatrutide across its target receptors compared to native ligands, based on current pharmacological data:
| Target Receptor | Primary Physiological Effect | Potency Relative to Native Ligand |
|---|---|---|
| GIPR | Lipid metabolism, Insulin secretion | High Agonist Activity |
| GLP-1R | Appetite suppression, Insulinogenesis | Moderate-to-High Activity |
| GCGR | Increased energy expenditure, Thermogenesis | Low-to-Moderate Activity |
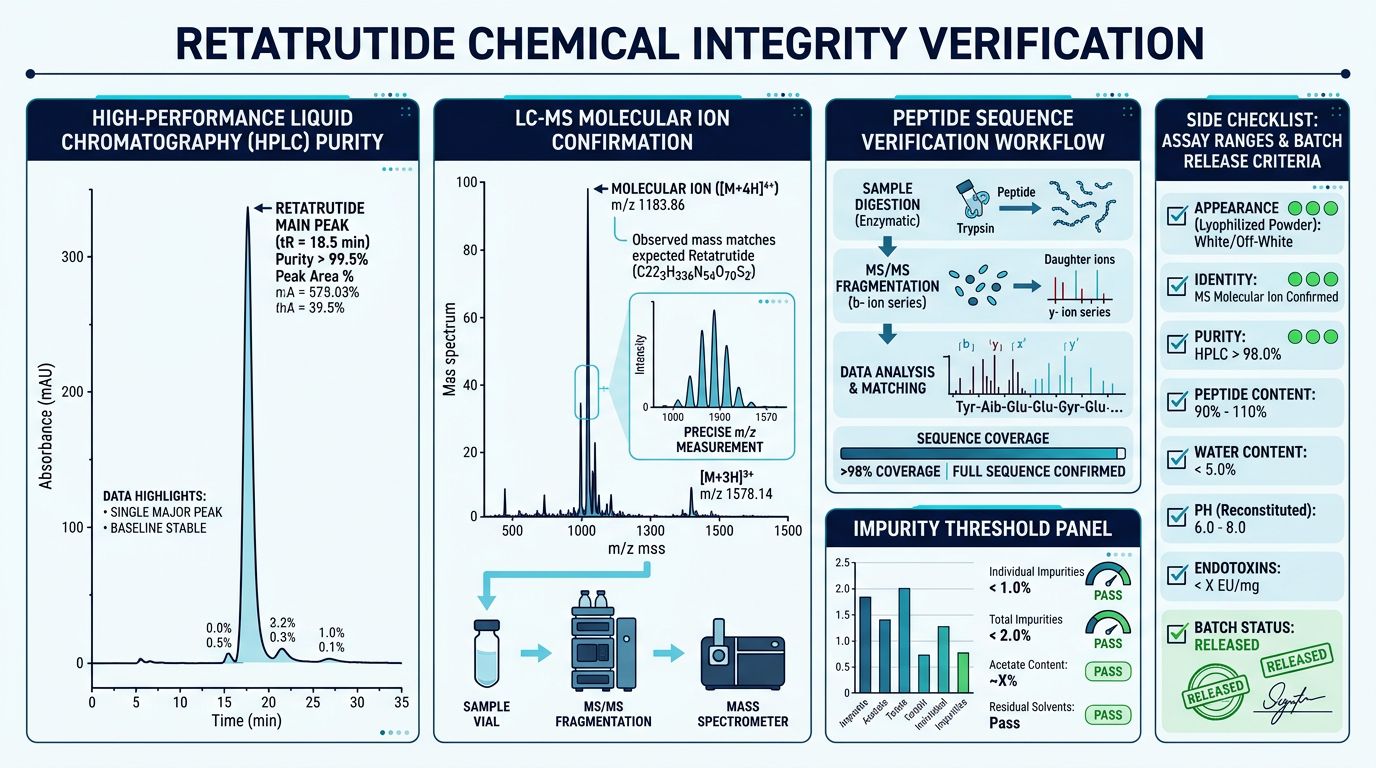
Verifying Chemical Integrity: The Analytical Benchmarks
When obtaining peptides from a peptide synthesis facility, researchers must demand high-resolution analytical data. At Alpha Carbon Labs, we emphasize that "purity" is a multi-faceted metric. For a complex acylated peptide like Retatrutide, three specific tests are non-negotiable.
High-Performance Liquid Chromatography (HPLC)
HPLC is used to determine the purity of the peptide. In this process, the sample is passed through a column under high pressure. Different molecules travel at different speeds based on their hydrophobicity. A high-quality Retatrutide sample should show a single, sharp peak on the chromatogram, typically exceeding 98% purity. Contaminants or "truncated sequences" (shorter versions of the peptide that failed during synthesis) appear as smaller "shoulder" peaks.
Mass Spectrometry (MS)
While HPLC tells you how pure the sample is, MS tells you what the sample is. By measuring the mass-to-charge ratio of the ions, researchers can confirm the molecular weight (MW) of the peptide. For Retatrutide, the expected MW is approximately 4731.33 Da. If the MS result deviates from the theoretical mass, the chemical integrity of the ligand is compromised, rendering research data invalid.
Visual and Solvent Analysis
Beyond the primary assays, checking for residual solvents and ensuring the lyophilization (freeze-drying) process was complete is vital. A crisp, white "cake" or powder indicates proper lyophilization, which is critical for the long-term stability of the peptide during storage. Researchers can verify these details through our COA (Certificate of Analysis) documents.
The Research Utility of Retatrutide vs. Other Multigonists
Researchers often choose between various agonists depending on the specific metabolic pathway they wish to isolate or emphasize. While Semaglutide remains the gold standard for GLP-1 isolation, and Tirzepatide serves as the benchmark for dual-agonism, Retatrutide is the primary tool for studying the "triple-agonist" effect.
- For Thermogenesis Studies: Retatrutide is superior due to its Glucagon receptor activity.
- For Adipose Tissue Research: The GIP component makes Retatrutide or SLU-PP-332 highly relevant.
- For Cardiovascular Research: Investigating the impact of triple-pathway activation on heart rate and blood pressure is a burgeoning field of study.
Safety and Handling in the Laboratory
Retatrutide is a highly potent biological agent. In laboratory settings, it is typically reconstituted in Bacteriostatic Water or sterile saline. Because it is a peptide, it is susceptible to mechanical degradation (shearing) and temperature fluctuations.
Reconstitution Protocol
- Allow the vial to reach room temperature before reconstitution to prevent condensation.
- Gently introduce the diluent down the side of the glass wall.
- Do not shake the vial; instead, gently swirl until the powder is fully dissolved.
- Store reconstituted Retatrutide at 2°C to 8°C (36°F to 46°F) to maintain stability.
For more information on laboratory standards, visit our quality control page.
Interdisciplinary Applications: Retatrutide and Synergistic Peptides
Advanced metabolic research often involves the co-administration of Retatrutide with other peptides to observe multi-system interactions. For instance, some researchers look at the combination of metabolic agonists with mitochondrial enhancers like MOTS-c or SS-31 to study cellular energy production under conditions of rapid fat oxidation.
Others may investigate the relationship between intensive weight loss modeling and tissue repair, utilizing BPC-157 or TB-500 to monitor how rapid physiological changes affect connective tissue integrity in animal models.
Key Indicators of a Reliable Supplier
When searching for Retatrutide for sale, the following criteria should be used to evaluate a research supplier:
- Transparency: Does the supplier provide batch-specific HPLC and MS data?
- Expertise: Is the supplier focused on research-grade chemicals with a deep understanding of peptide chemistry?
- Consistency: Are there internal controls to ensure that every vial contains the stated milligram amount (quantitative analysis)?
Conclusion
Retatrutide represents the current pinnacle of incretin-based peptide engineering. Its ability to concurrently activate GIP, GLP-1, and Glucagon receptors provides researchers with an unprecedented tool to investigate the complexities of human metabolism. However, the complexity of its 39-amino acid structure and fatty acid acylation means that chemical integrity cannot be taken for granted.
Ensuring that your laboratory is equipped with high-purity, verified Retatrutide is the first step in successful metabolic research. By prioritizing analytical verification through HPLC and MS, researchers can focus on the data that matters: uncovering the next generation of metabolic insights.
To explore our full catalog of research peptides, including Cagrilintide, Survodutide, and more, please visit our product pages.
References
- 1. Coskun, T., et al. (2022). "LY3437172, a novel glucagon receptor, glucose-dependent insulinotropic polypeptide receptor, and glucagon-like peptide-1 receptor triagonist." Cell Metabolism.
- 2. Urva, S., et al. (2023). "Pharmacokinetics and Pharmacodynamics of Retatrutide, a GIP, GLP-1, and Glucagon Receptor Triagonist, in Patients with Type 2 Diabetes." Diabetes Care.
- 3. Jall, S., et al. (2017). "Monomeric GLP-1/GIP/glucagon triagonism corrects obesity, hepatosteatosis, and dyslipidemia in female mice." Molecular Metabolism.
- 4. Heise, T., et al. (2023). "Tirzepatide vs insulin degludec as add-on to metformin in patients with type 2 diabetes: SURPASS-3." The Lancet. (Contextual for Multi-agonist benchmarking).
- 5. Müller, T. D., et al. (2022). "The history and future of targeting the glucagon and GLP-1 systems for the treatment of metabolic diseases." Diabetologia.
- 6. Luttrell, L. M. (2006). "Reviews in Molecular Biology and Biotechnology: Transmembrane Signaling by G Protein-Coupled Receptors." Molecular Biotechnology.
- 7. Knudsen, L. B., & Lau, J. (2019). "The Discovery and Development of Liraglutide and Semaglutide." Frontiers in Endocrinology.
- 8. Thomas, M. K., et al. (2023). "Retatrutide Phase 2 Data: Triple Agonist for Obesity and Type 2 Diabetes." New England Journal of Medicine.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.