The Triple Agonist Frontier: Pharmacological Dynamics of Retatrutide in Metabolic Research
Retatrutide is a groundbreaking triple-agonist peptide targeting GLP-1, GIP, and Glucagon receptors, offering unparalleled potential in metabolic research and weight loss modeling.
Introduction: The Shift from Mono to Triple Agonism
In the rapidly evolving landscape of metabolic research, the transition from single-receptor ligands to multi-receptor agonists represents a paradigm shift in pharmacological strategy. For years, the gold standard for incretin-based research was Glucagon-Like Peptide-1 (GLP-1) receptor agonism, exemplified by compounds like Semaglutide. However, the subsequent emergence of dual agonists, such as Tirzepatide (targeting GLP-1 and GIP), demonstrated that synergistic receptor activation could yield metabolic outcomes superior to those achievable by stimulating a single pathway.
Currently, the frontier of this research is defined by the "triple agonist" molecule: Retatrutide. This novel peptide, a unimolecular triple agonist, targets the GLP-1 receptor (GLP-1R), the Glucose-dependent Insulinotropic Polypeptide receptor (GIPR), and the Glucagon receptor (GCGR). By integrating glucagon agonism into the existing dual-incretin framework, Retatrutide offers a multi-modal approach to energy expenditure, lipid metabolism, and glycemic control that distinguishes it from its predecessors. For laboratories and researchers seeking Retatrutide for sale, understanding the nuanced molecular dynamics of this triple agonism is essential for contemporary metabolic modeling.
The Molecular Architecture of Retatrutide
Retatrutide (LY3437943) is an oxyntomodulin-based synthetic peptide consisting of 39 amino acids. Its sequence is meticulously engineered to balance the potencies across three distinct receptors. Unlike simple mixtures of separate peptides, Retatrutide is a single molecule designed to provide balanced co-activation, ensuring that all three metabolic pathways are engaged simultaneously at the target tissue site.
Key structural modifications include a C18 fatty diacid moiety, which facilitates reversible albumin binding. This is a critical feature for research applications, as it extends the half-life of the molecule to approximately six days in humans, allowing for a sustained pharmacological profile. Precision in peptide synthesis is paramount here; any variation in the sequence or the lipid side chain can drastically alter the affinity ratios between the GLP-1, GIP, and Glucagon receptors, potentially negating the synergistic benefits.
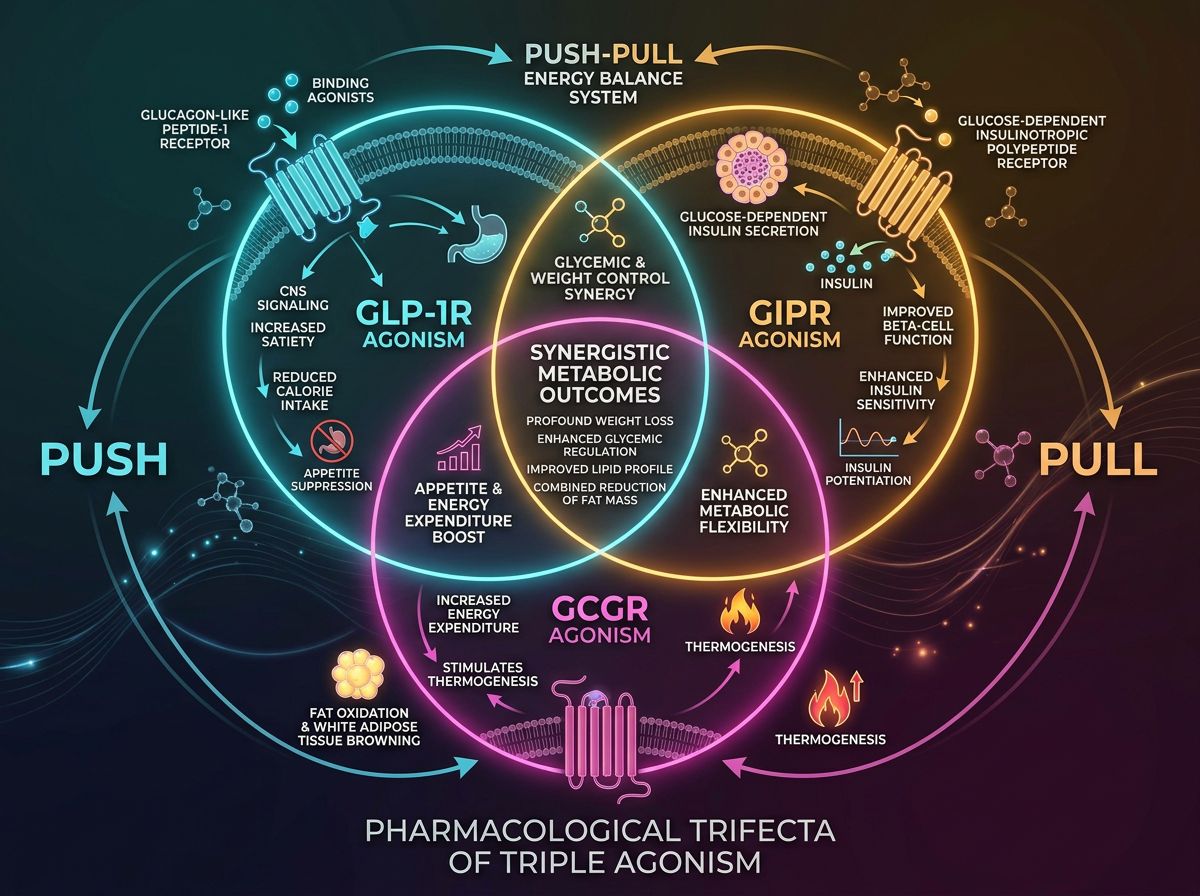
The Pharmacological Trifecta: Understanding the Roles
1. GLP-1 Receptor Agonism: The Foundation
The GLP-1 component remains the cornerstone of Retatrutide’s mechanism. Acting primarily in the pancreas and the central nervous system (CNS), GLP-1 stimulation promotes glucose-dependent insulin secretion, inhibits inappropriate glucagon release, and slows gastric emptying. Most significantly for weight-related research, GLP-1R activation in the hypothalamus promotes satiety. Researchers often compare Retatrutide to Semaglutide to isolate the specific contributions of the additional receptors.
2. GIP Receptor Agonism: The Metabolic Buffer
Initially thought to be counterproductive in metabolic research due to its glucagonotropic properties, GIPR agonism has proven to be a potent synergist. In Retatrutide, GIP agonism functions to enhance insulin sensitivity and potentially mitigate some of the gastrointestinal side effects associated with pure GLP-1 activation. This receptor also plays a role in adipose tissue metabolism, facilitating "healthy" fat storage and increasing the threshold for lipid deposition in ectopic sites like the liver.
3. Glucagon Receptor Agonism: The Thermogenic Driver
The inclusion of the Glucagon receptor (GCGR) is what separates Retatrutide from Tirzepatide. In traditional endocrinology, glucagon is viewed as the antagonist to insulin. However, at pharmacological levels, glucagon agonism increases energy expenditure through thermogenesis and directly stimulates hepatic lipid oxidation. By combining GCGR agonism with GLP-1 and GIP (which counteract glucagon’s hyperglycemic effects), Retatrutide achieves a state of "metabolic revving" without the glycemic instability typically associated with glucagon.
Comparative Analysis: Retatrutide vs. Dual Agonists
To contextualize the research potential of Retatrutide, one must look at how it compares to established dual agonists and other emerging metabolic molecules. The following table highlights the primary mechanical differences:
| Feature | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Receptor Targets | GLP-1R | GLP-1R + GIPR | GLP-1R + GIPR + GCGR |
| Mechanism Summary | Satiety & Insulin stimulation | CNS Satiety + Adipose insulin sensitivity | Satiety + Adipose health + Hepatic lipolysis + Thermogenesis |
| Weight Loss Potential (Experimental) | Moderate (approx. 15%) | High (approx. 20-22%) | Very High (extrapolated 24%+) |
| Primary Metabolic Focus | Appetite & Glucose | Appetite & Metabolic Efficiency | Total Energy Expenditure & Liver Health |
Synergy Over Addition
The primary discovery in recent trials, such as the Phase 2 data published in the New England Journal of Medicine, is that the effect of Retatrutide is not merely the sum of its parts. The triple-agonist approach appears to bypass the physiological plateaus often seen with single-agonist molecules. By increasing energy expenditure (via Glucagon) while simultaneously reducing caloric intake (via GLP-1 and GIP), Retatrutide creates a wider caloric deficit than can be achieved by appetite suppression alone.
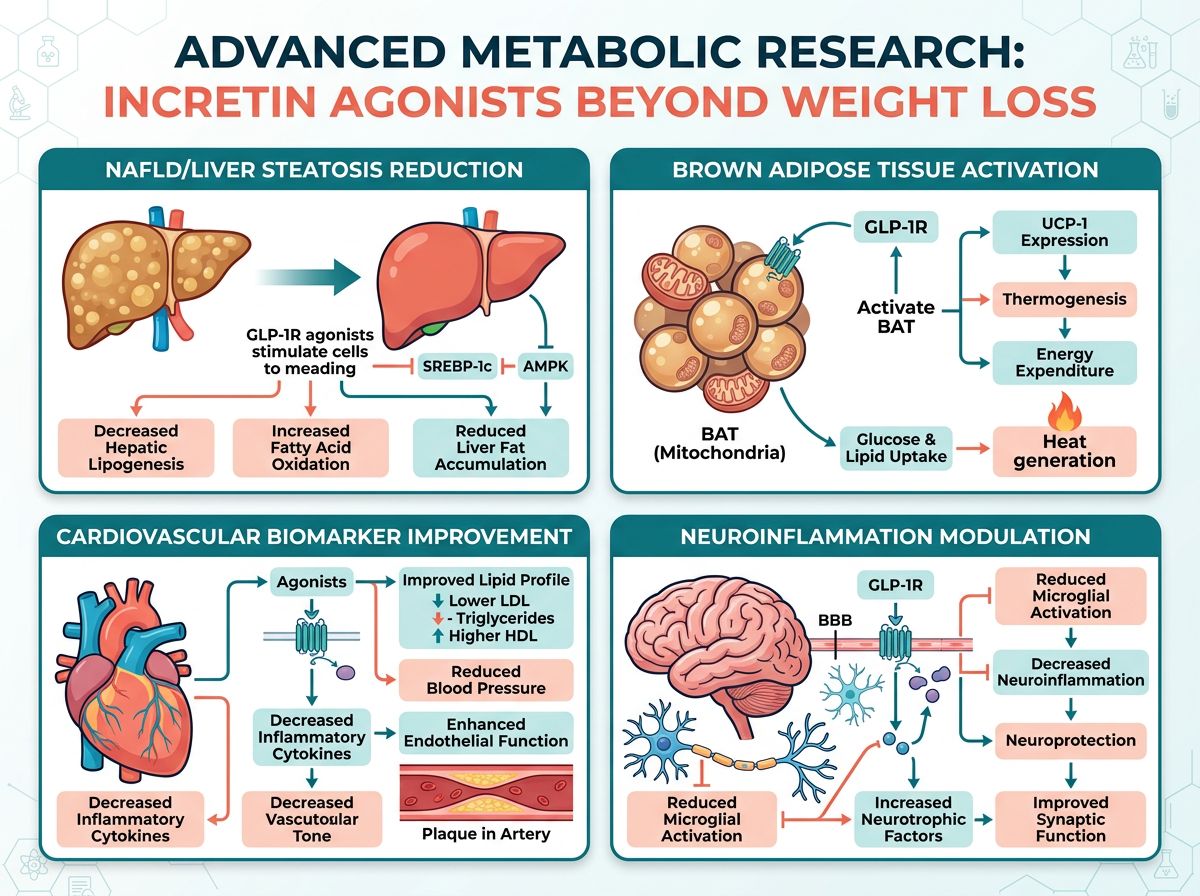
Advanced Metabolic Applications in Research
Hepatic Health and NAFLD/MASH
One of the most promising areas for Retatrutide research is in Non-Alcoholic Fatty Liver Disease (NAFLD), recently redefined as Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Because the glucagon receptor is highly expressed in the liver, Retatrutide has shown a profound ability to reduce intrahepatic fat content. In early studies, high doses of Retatrutide resulted in a >80% reduction in liver fat for a significant percentage of research cohorts. This makes Retatrutide an excellent candidate for studies involving mitochondrial health, often paired with mitochondrial-specific peptides like SS-31 or MOTS-c.
Cardiovascular Dynamics
Researchers investigating Retatrutide must monitor heart rate variations. Due to the GCGR agonism, Retatrutide tends to increase resting heart rate more than pure GLP-1 agonists. This SNS (Sympathetic Nervous System) activation is part of the thermogenic mechanism but remains a key area of study for long-term cardiovascular safety. Contrasting this with the cardioprotective effects of BPC-157 or the vascular support of ARA-290 provides a broad field for multi-peptide experimental designs.
Weight Regain and Metabolic Set-Point
Fundamental to obesity research is the concept of the "metabolic set-point." Retatrutide research focuses on whether the triple-agonist approach can "reset" this point more effectively than dual agonists. By stimulating energy expenditure, there is a theoretical potential for better weight maintenance post-cessation compared to molecules that only address appetite. This can be further explored in conjunction with 5-amino-1mq to observe intracellular NAD+ levels and metabolic rate.
Stability, Reconstitution, and Quality Control
For research integrity, the quality of the peptide is non-negotiable. Retatrutide is a complex molecule; its 39-amino acid sequence requires sophisticated peptide synthesis techniques to prevent truncated sequences or isomeric impurities. At Alpha Carbon Labs, we emphasize the importance of COA documents and rigorous quality control.
Reconstitution Protocols
Retatrutide is typically provided in a lyophilized (freeze-dried) state to ensure stability during transport. For research purposes, it should be reconstituted with Bacteriostatic Water. Due to its long half-life, researchers must be precise in dosage calculations to avoid over-saturation of the GCGR, which could lead to transient hyperglycemia before the incretin components (GLP-1 and GIP) fully Compensate.
Potential Synergies in Experimental Modeling
While Retatrutide is potent as a standalone agent, contemporary research often explores it within a broader peptide stack:
- Retatrutide + IGF-1 LR3: To study the preservation of lean muscle mass during rapid adipose tissue loss. IGF-1LR3 provides the anabolic stimulus that may be dampened by significant caloric deficits.
- Retatrutide + GHK-Cu: Investigating skin elasticity and collagen synthesis during rapid weight loss to mitigate the "skin sag" often observed in clinical models. GHK-Cu is the gold standard for ECM remodeling.
- Retatrutide + MOTS-c: Exploring the combined impact of triple-agonism and mitochondrial-derived peptides on insulin sensitivity and exercise mimetic effects. MOTS-c complements the hepatic fat oxidation of Retatrutide.
Safety and Research Considerations
As with all high-potency research peptides, Retatrutide demands a strict adherence to safety protocols. The most common adverse effects observed in animal models and clinical trials are gastrointestinal in nature (nausea, vomiting, diarrhea), which are dose-dependent. Notably, the glucagon component introduces a skin-related sensitivity in some models, and the increased heart rate requires careful monitoring in subjects with underlying cardiovascular concerns.
Unlike AOD9604, which is a fragment of HGH designed specifically for lipolysis without glycemic impact, Retatrutide is a systemic metabolic regulator. Researchers should distinguish between the targeted lipolysis of fragments and the comprehensive metabolic overhaul offered by triple agonists.
Conclusion: The Future of Incretin Research
Retatrutide represents the current zenith of incretin-based pharmacology. By simultaneously targeting the GLP-1, GIP, and Glucagon receptors, it offers a level of metabolic control that was previously thought unattainable with a single molecule. For those in the research community, Retatrutide is not just another weight loss peptide; it is a tool for exploring the complex interplay of thermogenesis, satiety, and hepatic lipid management.
As research continues to unfold, particularly regarding Retatrutide's role in treating MASH and obstructive sleep apnea, the demand for high-purity, verified peptides will only increase. Alpha Carbon Labs remains committed to providing researchers with the highest quality Retatrutide for sale, backed by comprehensive quality control protocols to ensure that your data is as accurate as your hypotheses.
To explore our full range of metabolic research peptides, including Mazdutide, Cagrilintide, and Survodutide, please visit our catalog or view our latest COA documents for batch-specific purity data.
References
- 1. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. N Engl J Med. 2023.
- 2. Müller TD, et al. The Evolution of GLP-1-Based Drugs: From Single to Triple Agonists. Diabetologia. 2022.
- 3. Coskun T, et al. LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: From bench to clinical proof of concept. Cell Metab. 2022.
- 4. Finan B, et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nat Med. 2015.
- 5. Heise T, et al. Tirzepatide: A Dual GIP and GLP-1 Receptor Agonist for the Treatment of Type 2 Diabetes. Expert Opin Pharmacother. 2022.
- 6. Urva S, et al. Dose-dependent effects of the triple glucagon-like peptide-1, glucose-dependent insulinotropic polypeptide and glucagon receptor agonist retatrutide. Diabetes Obes Metab. 2023.
- 7. Knerr PJ, et al. Next generation multi-receptor agonists for the treatment of metabolic diseases. International Journal of Molecular Sciences. 2022.
- 8. Baggio LL, Drucker DJ. Glucagon-like peptide-1 receptor co-agonists for the treatment of metabolic disease. Endocr Rev. 2021.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.