The RUO Framework: Navigating the Legal Landscape of Domestic Peptide Research Compliance
A comprehensive guide to the regulatory distinctions between Research Use Only (RUO) labeling and FDA-approved pharmaceuticals. This article navigates the legal framework, quality standards, and compliance requirements for domestic peptide research.
Introduction: The Dual Nature of Bioactive Peptides
The landscape of modern biotechnology is defined by rapid innovation, particularly in the realm of amino acid sequencing and synthesis. Peptides—short chains of amino acids linked by peptide bonds—have emerged as a dominant class of molecules in both basic science and pharmaceutical development. However, this scientific prominence exists within a complex legal and regulatory framework that strictly delineates how these substances can be manufactured, sold, and utilized.
For independent researchers, academic institutions, and domestic peptide synthesis laboratories, understanding the distinction between "Research Use Only" (RUO) compounds and approved pharmaceutical drugs is not merely a matter of semantics; it is a critical compliance requirement enforced by federal agencies, most notably the U.S. Food and Drug Administration (FDA).
The "RUO Framework" refers to the set of regulations, chemical standards, and ethical guidelines that govern the existence of peptides intended solely for non-clinical investigation. While the internet has popularized terms like "research chemicals" in grey-market contexts, legitimate scientific inquiry relies on a precise adherence to these legal distinctions to ensure the integrity of data and the safety of the public. This guide aims to provide a comprehensive analysis of the legal landscape surrounding domestic peptide research, clarifying the boundaries between laboratory experimentation and clinical application.
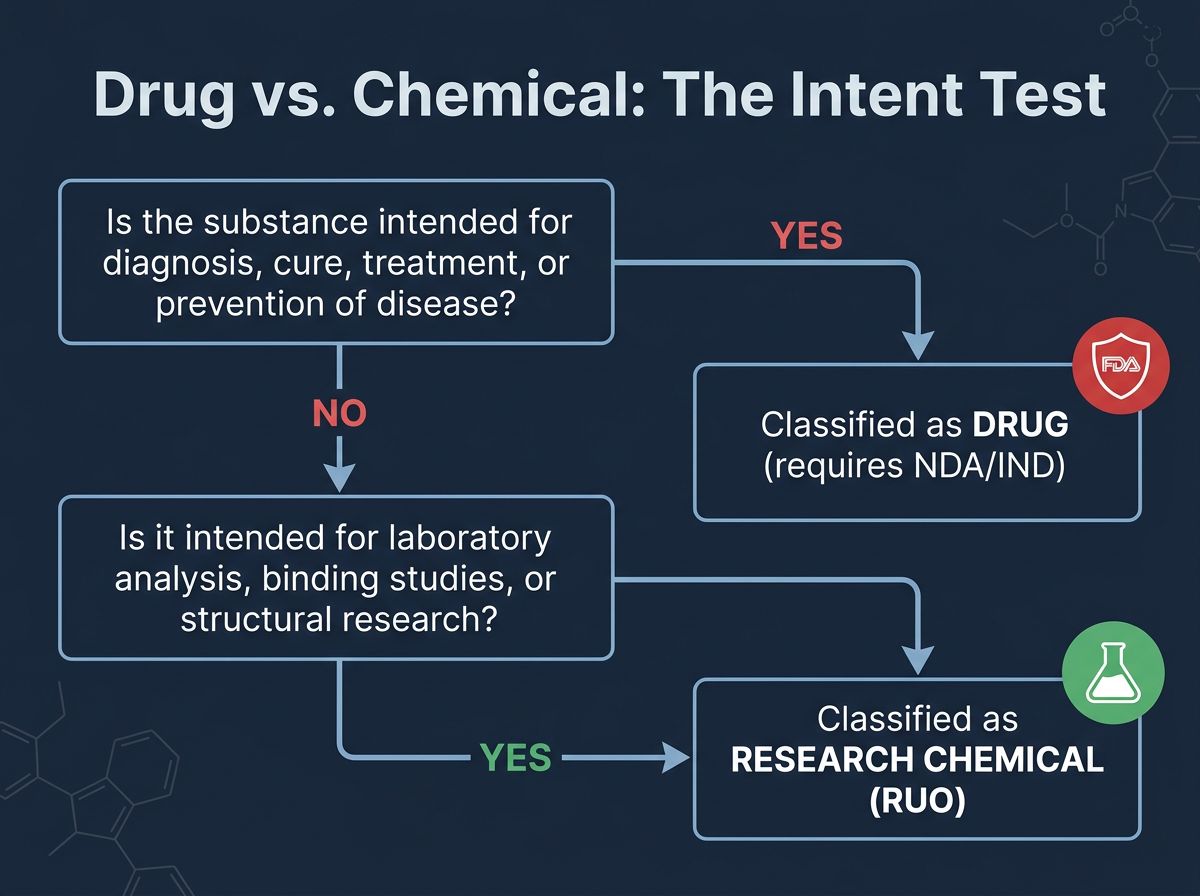
The Regulatory Bedrock: Defining "Drug" vs. "Chemical"
To navigate compliance, one must first understand how the federal government classifies substances. The primary statute governing this in the United States is the Federal Food, Drug, and Cosmetic Act (FD&C Act). Under Section 201(g)(1) of the FD&C Act, a "drug" is defined not by its chemical structure, but by its intended use.
The Intent Clause
A substance is classified as a drug if it meets any of the following criteria:
- It is recognized in the official United States Pharmacopoeia (USP) or Homoeopathic Pharmacopoeia.
- It is intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in man or other animals.
- It is intended to affect the structure or any function of the body of man or other animals.
This definition creates a legal pivot point based on "objective intent." If a supplier markets a peptide like BPC-157 with claims tackling gastric ulcers or tendon repair in humans, that chemical is legally essentially a new drug. Without an approved New Drug Application (NDA) or an Investigational New Drug (IND) exemption, selling it for that purpose is illegal.
Conversely, if that same chemical sequence is synthesized, labeled, and sold strictly for laboratory analysis—such as investigating its stability in varying pH levels, its binding affinity to cellular receptors in a petri dish, or its structural properties via mass spectrometry—it remains a "research chemical." The RUO framework operates in this space: supplying tools for scientific discovery, not treatments for medical conditions.
Unpacking the "Research Use Only" (RUO) Designation
The label "Research Use Only" is a functional regulatory category primarily derived from regulations concerning in vitro diagnostic products (21 CFR 809.10), but the principle extends broadly across the chemical supply industry. An RUO product is one in the laboratory phase of development. It is not intended for clinical use, meaning it should not be administered to human subjects for diagnostic or therapeutic purposes.
What RUO Compliance Entails
For a peptide to be genuinely RUO, the entire lifecycle of the product must reflect this limitation:
- Marketing and Labeling: The product cannot be advertised with health claims, dosing protocols for humans, or testimonials regarding physical benefits. The labeling must arguably state "For Research Use Only. Not for Human Consumption."
- Sales Channels: Vendors operating compliantly often restrict sales to qualified researchers or screen orders to ensure they are not supplying individuals seeking medical treatment outside the healthcare system.
- Technical Support: Support provided by the manufacturer is limited to chemical characteristics (solubility, molecular weight, sequence storage) rather than biological advice (how much to inject, expected physiological effects).
For example, researchers studying Semaglutide in a preclinical setting might be investigating its mechanism of action on Glucagon-Like Peptide-1 (GLP-1) receptors in isolated tissue samples. As long as the investigation remains preclinical (in vitro or animal models), the sourcing of RUO-grade materials is standard practice. However, once the intent shifts to treating a diabetic patient, the researcher must utilize an FDA-approved formulation obtained through a licensed pharmacy, not a chemical supplier.
Good Manufacturing Practice (GMP) vs. Research Grade
A major technical and legal distinction in the peptide world lies in the manufacturing standards. This is often the dividing line between a legally compliant research tool and a compliant pharmaceutical ingredient.
Non-GMP (Research Grade)
Most RUO peptides are synthesized under non-GMP conditions. This does not mean they are low quality; rather, it means they are not produced with the exhaustive documentation and environmental controls required for human drugs. Research grade synthesis focuses on chemical purity (often >99%) and sequence accuracy.
Common features of Research Grade production:
- Synthesis Method: Solid Phase Peptide Synthesis (SPPS).
- Validation: Verified by HPLC (High-Performance Liquid Chromatography) and MS (Mass Spectrometry).
- Traceability: Batch-level tracking, but less granular than GMP.
- Cost: Significantly lower, allowing for high-throughput screening and experimentation.
cGMP (Current Good Manufacturing Practice)
cGMP is a regulatory standard enforced by the FDA (21 CFR Parts 210 and 211) that ensures drugs are consistently produced and controlled. It covers every aspect of production, from air quality in the facility to the training of the janitorial staff.
| Feature | Research Grade (RUO) | cGMP (Clinical/Commercial) |
|---|---|---|
| Primary Use | In vitro studies, animal models, material science | Human clinical trials (Phase I-III), Commercial Drugs |
| Regulatory Oversight | General chemical regulations | Strict FDA enforcement & regular audits |
| Documentation | Certificate of Analysis (CoA) | Extensive batch records, validation reports, stability data |
| Environmental Control | Standard laboratory environment | Clean rooms (ISO 5-8), strict biocontamination controls |
Researchers must understand that RUO peptides, regardless of their purity, cannot be used in human trials because they lack the cGMP "pedigree" required to ensure patient safety.
The Critical Role of Quality Control in Compliance
Legitimate research requires reproducible data. While RUO products are not for humans, they must still meet rigorous chemical standards to be useful to scientists. A peptide labeled RUO that is impure or misidentified renders any resulting data invalid.
Domestic suppliers like Alpha Carbon Labs emphasize quality control not just for customer satisfaction, but as a pillar of scientific integrity. Adherence to strict testing protocols is what differentiates a professional laboratory supplier from grey-market vendors.
Certificate of Analysis (CoA) as a Legal & Scientific Document
The CoA (Certificate of Analysis) is the primary document attesting to the quality of a chemical batch. In the RUO framework, a credible CoA must include:
- Chromatogram (HPLC): Visual proof of purity, showing the main peak of the peptide versus impurities (truncated sequences, protection groups).
- Mass Spectrum (MS): Confirmation of molecular weight to identify the correct sequence.
- Solubility and Appearance: Physical characteristics.
- Date of Analysis: Ensuring the data is current.
For researchers, purchasing peptides without verifiable CoAs introduces variables that can ruin experiments. If a researcher is testing the efficacy of Tirzepatide in a rodent obesity model, and the supplied peptide is only 80% purity with 20% unknown trifluoroacetic acid (TFA) salts or deletion errors, the metabolic data gathered will be flawed. This highlights why high-purity RUO peptides utilize comparable synthesis technologies to pharma production, even if the regulatory paperwork differs.
Navigating the "Research Use" Boundary: Practical Scenarios
To further clarify the RUO framework, we can examine specific scenarios where legal boundaries are either maintained or crossed.
Scenario A: The University Laboratory (Compliant)
A PhD candidate is studying the effects of GHK-Cu on collagen synthesis in aged dermal fibroblasts.
Action: The lab purchases lyophilized GHK-Cu from a domestic chemical supplier labeled "Research Use Only."
Usage: The peptide is dissolved in media and applied to cell cultures. The results are published in a peer-reviewed journal.
Verdict: Fully compliant. The substance was used for in vitro investigation to generate knowledge.
Scenario B: The Compounding Inquiry (Non-Compliant)
A wellness clinic wants to offer Thymosin Alpha-1 injections to patients for immune support.
Action: They attempt to purchase RUO-labeled vials from a chemical website to administer to patients.
Verdict: Non-compliant and illegal. Buying RUO chemicals to compound into drugs for humans violates the FD&C Act. The clinic must source FDA-approved or 503B compounding pharmacy-sourced medication.
Scenario C: The Independent Researcher (Compliant)
A private citizen is conducting recognized observational research on peptide stability.
Action: They purchase Epithalon to test its degradation rates when exposed to UV light using their own HPLC equipment.
Verdict: Compliant. As long as the intent is material analysis and not consumption, this falls under lawful chemical research.
The Spectrum of Peptide Research Targets
The RUO framework enables the exploration of vast libraries of peptides before they ever reach clinical trials. It is the engine of discovery. Several categories of peptides are currently subjects of intense scrutiny in laboratory settings.
Metabolic Research
With the rise of GLP-1 agonists, metabolic research has exploded. Peptides like Semaglutide, Tirzepatide, and Retatrutide are heavily researched in animal models to understand their effects on insulin secretion, gastric emptying, and adipose tissue browning. In the RUO sphere, these compounds allow scientists to map the precise signaling pathways of the GIP and GLP-1 receptors without the restrictions of branded pharmaceutical purchasing.
Regenerative / Growth Factor Research
Peptides derived from growth factors, such as IGF-1LR3 and MGF (Mechano Growth Factor), are staples in muscle physiology research. Scientists use these to understand satellite cell activation and muscle hypertrophy signaling. Similarly, TB-500 (Thymosin Beta-4 fragment) is widely studied for its potential in actin sequestration and cell migration, fundamental processes in tissue repair.
Nootropic and Neuropeptide Research
Compounds like Semax and Selank, originally developed in Russia, are studied in the West primarily for their neuroprotective properties and influence on BDNF (Brain-Derived Neurotrophic Factor). RUO access allows neurology labs to investigate these peptides' potential roles in mitigating oxidative stress in neurons.
Safety Considerations in Peptide Handling
Even though RUO peptides are not for human consumption, safety in the laboratory is paramount. Comprehensive knowledge of the Safety Data Sheet (SDS) for each compound is required.
Lyophilization and Reconstitution
Most research peptides arrive as lyophilized (freeze-dried) powders. This state renders them stable for shipping but requires careful handling.
Inhalation Risk: Generally low for peptides, but standard PPE (gloves, goggles, lab coat) is required to prevent contamination of the sample and exposure to the researcher.
Solvents: Reconstitution often involves bacteriostatic water or dilute acetic acid. Researchers must be aware of the chemical properties of the solvent and the peptide’s isoelectric point to prevent precipitation.
Storage Protocols
Maintaining the integrity of the research material is part of the compliance mindset—degraded materials yield bad data.
Short Term: Lyophilized peptides are generally stable at room temperature for weeks, but refrigeration (4°C) is preferred.
Long Term: Storage at -20°C or -80°C is standard to prevent hydrolysis or oxidation.
Freeze-Thaw Cycles: Repeated freezing and thawing damages the peptide structure. Aliquoting—splitting the reconstituted peptide into smaller single-use vials—is the standard best practice.
The Future of Domestic Peptide Regulation
The regulatory environment is dynamic. In recent years, the FDA has taken a more aggressive stance against vendors who blur the lines between RUO and unapproved drugs. This crackdown benefits the scientific community by removing bad actors who sell low-quality, impure products disguised as research materials.
For domestic suppliers, the path forward involves increased transparency. This includes:
- Third-Party Testing: Independent verification of purity becomes a baseline requirement, not a luxury.
- KYC (Know Your Customer): Suppliers implementing screening to ensure they are selling to legitimate researchers rather than consumers.
- Supply Chain Auditability: Being able to trace the raw amino acids used in synthesis back to their source.
As the scientific potential of peptides like MOTS-c (a mitochondrial-derived peptide) becomes more apparent, the demand for high-quality RUO materials will grow. The distinction between "research" and "therapy" will remain the central legal pillar supporting this industry.
Conclusion
The "Research Use Only" framework is a vital component of the scientific ecosystem. It allows for the rapid, cost-effective exploration of biological mechanisms that eventually lead to medical breakthroughs. For researchers, understanding the legal boundaries of RUO products is essential for maintaining ethical standards and ensuring regulatory compliance.
By respecting the distinction between chemical supply and pharmaceutical intervention, and by prioritizing purity through rigorous quality control, the scientific community can continue to unlock the potential of peptides safely and legally. Whether investigating the tissue-repair properties of BPC-157 or the metabolic pathways of Semaglutide, the integrity of the research depends on the integrity of the source and the strict adherence to the intended use.
References
- Federal Food, Drug, and Cosmetic Act, 21 U.S.C. Section 201(g)(1). Link
- United States Food and Drug Administration. "Distribution of In Vitro Diagnostic Products Labeled for Research Use Only or Investigational Use Only: Guidance for Industry and Food and Drug Administration Staff." FDA.gov. Link
- Hagenstein, J., & Sewald, N. "Chemical Synthesis of Proteins and Peptides." In: Bioorganic Chemistry, 2013. PubMed
- D'Souza, P., & Shah, S. "Peptide therapeutics: current status and future directions." Drug Discovery Today, vol. 20, no. 1, 2015, pp. 122-128. DOI
- Code of Federal Regulations Title 21, Part 312. "Investigational New Drug Application." Link
- Isidro-Llobet, A., et al. "Amino Acid-Protecting Groups." Chemical Reviews, vol. 109, no. 6, 2009. DOI
- Ambrogelly, A., et al. "Analytical challenges in the development of peptide pharmaceuticals." Journal of Pharmaceutical and Biomedical Analysis, vol. 191, 2020. DOI
- Fosgerau, K., & Hoffmann, T. "Peptide therapeutics: current status and future directions." Drug Discovery Today, vol. 20, no. 1, 2015. PubMed
- Goodman, M., et al. "Synthesis of Peptides and Peptidomimetics." Houben-Weyl Methods of Organic Chemistry, Vol E22. Link
References
- 1. United States Food and Drug Administration. "Distribution of In Vitro Diagnostic Products Labeled for Research Use Only or Investigational Use Only: Guidance for Industry and Food and Drug Administration Staff." FDA.gov.
- 2. Hagenstein, J., & Sewald, N. "Chemical Synthesis of Proteins and Peptides." In: Bioorganic Chemistry, 2013.
- 3. Isidro-Llobet, A., et al. "Amino Acid-Protecting Groups." Chemical Reviews, vol. 109, no. 6, 2009, pp. 2455-2504.
- 4. D'Souza, P., & Shah, S. "Peptide therapeutics: current status and future directions." Drug Discovery Today, vol. 20, no. 1, 2015, pp. 122-128.
- 5. Federal Food, Drug, and Cosmetic Act, 21 U.S.C. Section 201(g)(1).
- 6. Code of Federal Regulations Title 21, Part 312. "Investigational New Drug Application."
- 7. Ambrogelly, A., et al. "Analytical challenges in the development of peptide pharmaceuticals." Journal of Pharmaceutical and Biomedical Analysis, vol. 191, 2020.
- 8. Fosgerau, K., & Hoffmann, T. "Peptide therapeutics: current status and future directions." Drug Discovery Today, vol. 20, no. 1, 2015.
- 9. Goodman, M., et al. "Synthesis of Peptides and Peptidomimetics." Houben-Weyl Methods of Organic Chemistry, Vol E22.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.