Stability and Degradation: Critical Storage Parameters for Research-Grade Tirzepatide
Explore the critical storage parameters for Tirzepatide, including the effects of pH, temperature, and light on peptide stability to ensure data integrity in metabolic research.
The Biochemistry of Tirzepatide: Understanding Structural Vulnerability
In the landscape of clinical research, Tirzepatide represents a pinnacle of peptide engineering. As a dual Glucose-dependent Insulinotropic Polypeptide (GIP) and Glucagon-Like Peptide-1 (GLP-1) receptor agonist, its structural complexity is the source of its efficacy. However, this same complexity introduces significant challenges regarding chemical and physical stability. For researchers, maintaining the integrity of the peptide backbone and the specific acylation chain is paramount to achieving reproducible results in metabolic studies.
Tirzepatide is a 39-amino acid synthetic peptide. Its uniqueness lies in its structure: it is based on the GIP sequence but incorporates two aminoisobutyric acid (Aib) residues at positions 2 and 13 to enhance stability against dipeptidyl peptidase-4 (DPP-4) degradation. Crucially, it features a C20 fatty diacid moiety attached via a linker to the Lysine residue at position 20. This "fatty acid tail" facilitates albumin binding, extending the half-life but also introducing hydrophobic regions that can influence the peptide's behavior in aqueous solutions.
To ensure high-fidelity data, researchers must understand the degradation pathways—such as deamidation, oxidation, and aggregation—that can occur if storage parameters are ignored. At Alpha Carbon Labs, we emphasize that quality begins with synthesis but is preserved through rigorous quality control and proper handling.
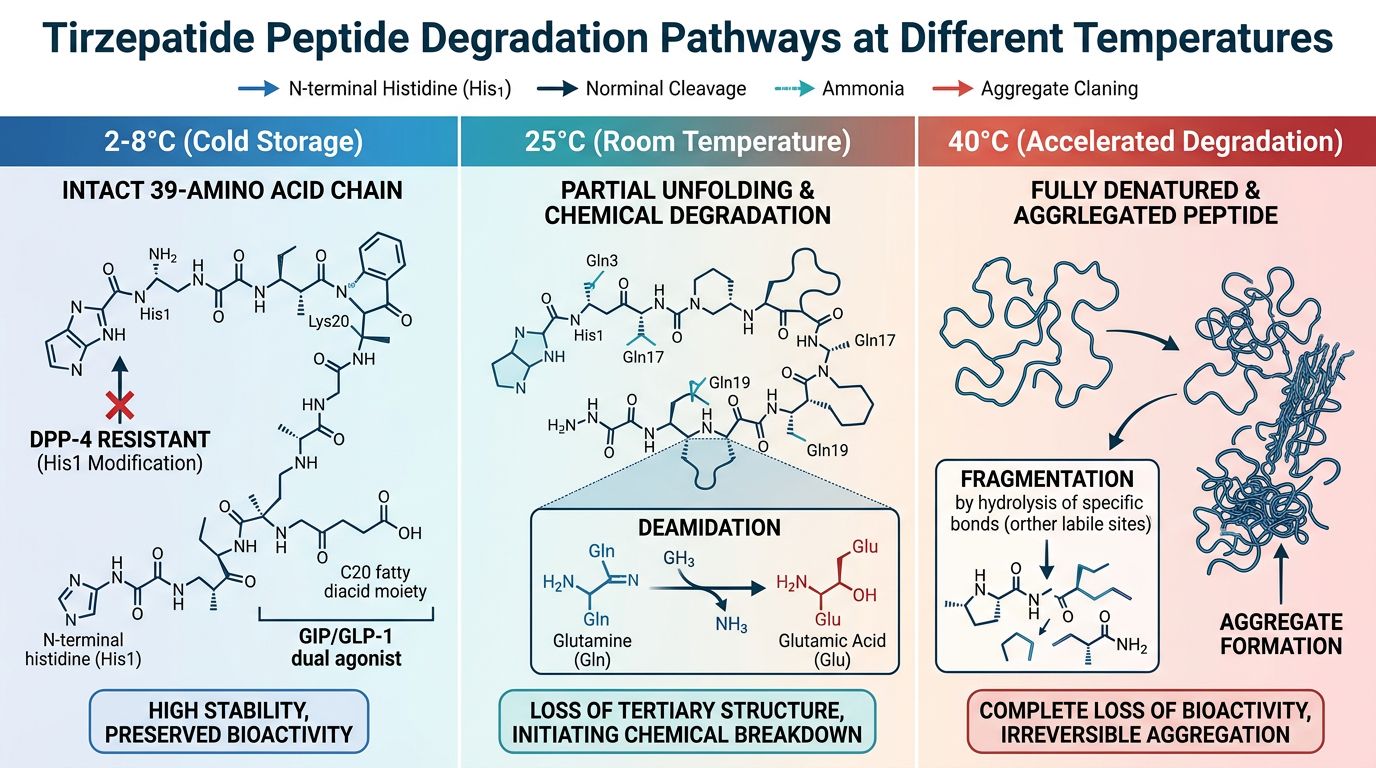
Critical Parameters: Temperature and Kinetic Degradation
Temperature is the primary driver of peptide degradation kinetics. For a complex molecule like Tirzepatide, elevated temperatures accelerate both chemical reactions and physical transitions (denaturation or aggregation).
Long-Term Storage (Lyophilized State)
In its lyophilized (freeze-dried) state, Tirzepatide is at its most stable. The removal of water significantly reduces the rate of hydrolysis and deamidation. Nevertheless, the Arrhenius equation dictates that even in a solid state, molecular motion and degradation occur. For long-term preservation, researchers should adhere to the following temperature guidelines:
- -20°C to -80°C: Ideal for storage exceeding six months. At these temperatures, chemical degradation is virtually halted.
- 4°C (Refrigeration): Acceptable for short-term storage (1-3 months), provided the vial remains vacuum-sealed and protected from light.
- Room Temperature (25°C): While Tirzepatide exhibits moderate robustness during shipping, prolonged exposure to room temperature in a research setting can lead to a measurable loss of purity over weeks.
Post-Reconstitution Stability
Once reconstituted, the "stability clock" accelerates. In solution, the peptide is susceptible to hydrolysis. Data suggests that reconstituted Tirzepatide should be used within 7 to 14 days when stored at 4°C. Repeated freeze-thaw cycles must be avoided, as the mechanical stress of ice crystal formation can shear the peptide chain or cause the fatty acid linker to dissociate/aggregate.
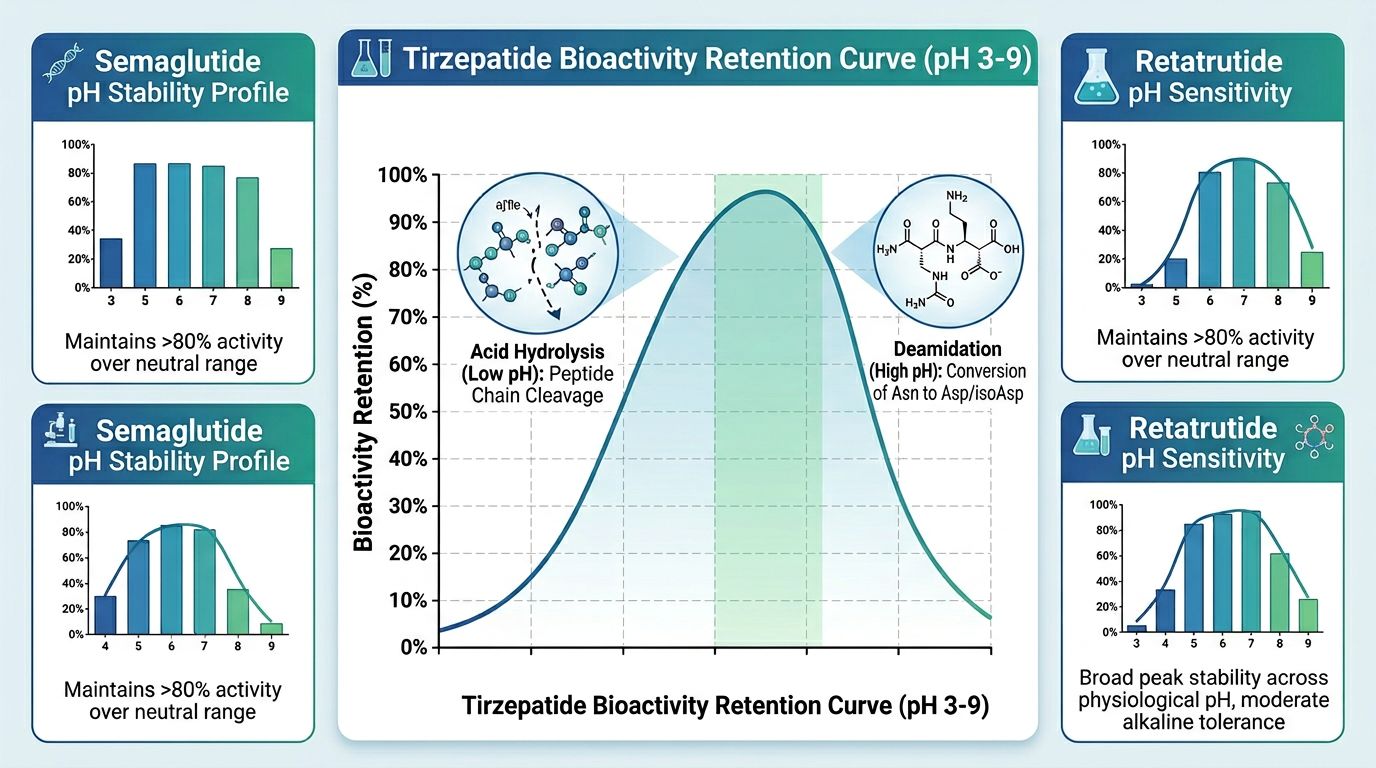
The Impact of pH on Chemical Integrity
The pH of the reconstitution buffer is perhaps the most overlooked factor in research-grade peptide stability. Tirzepatide contains several ionizable side chains, and its stability profile is highly pH-dependent.
Alkaline vs. Acidic Environments
In highly alkaline environments (pH > 8.0), the risk of deamidation (particularly at Asparagine or Glutamine residues) increases significantly. Conversely, in highly acidic environments (pH < 3.0), the peptide backbone may undergo hydrolysis. The C20 fatty acid moiety also exhibits different solubility and aggregation behaviors depending on the charge state of the molecule.
| pH Range | Effect on Tirzepatide | Suitability for Research |
|---|---|---|
| 2.0 - 4.5 | Increased risk of peptide bond hydrolysis; poor solubility. | Low |
| 5.0 - 7.5 | Optimal stability; minimal deamidation and oxidation. | High |
| 8.0 - 10.0 | Rapid deamidation and potential disulfide scrambling/aggregation. | Low |
For most metabolic research, Bacteriostatic Water or sterile 0.9% Saline is used. However, for specialized kinetic studies, a buffered solution such as Phosphate Buffered Saline (PBS) at pH 7.4 is recommended to maintain the peptide in its most stable, physiologically relevant state.
Advanced Storage Considerations: Light and Oxidation
Beyond temperature and pH, researchers must account for photo-oxidation and oxidative stress. Certain amino acids within Tirzepatide, such as Methionine or Tryptophan (depending on the specific analog or impurities), are sensitive to reactive oxygen species (ROS) and ultraviolet (UV) light.
Photostability
Exposure to light can catalyze the formation of free radicals, leading to the cleavage of the peptide backbone or the modification of the C20 linker. It is imperative to store Tirzepatide in amber vials or light-shielded containers. This is particularly important when comparing Tirzepatide to other incretin mimetics like Semaglutide or newer triple agonists like Retatrutide, as differences in side-chain composition change their photostability profiles.
Oxidative Degradation
The headspace in a vial contains oxygen, which can induce oxidation over time. For high-fidelity research, particularly when evaluating long-term metabolic impacts in vivo, purging the vial with an inert gas like Nitrogen or Argon prior to sealing can dramatically enhance shelf life. This level of care is consistent with the standards found in our peptide synthesis facility.
Comparison of Metabolic Research Peptides
When designing a study, it is helpful to compare the stability requirements of Tirzepatide with other agents used in weight loss and metabolic research. While Tirzepatide is a dual agonist, other researchers might utilize Cagrilintide (an amylin analog) or Mazdutide. Each has unique solubility and stability hurdles.
| Peptide | Mechanism | Stability Profile |
|---|---|---|
| Tirzepatide | GLP-1/GIP Agonist | Moderate-High (requires pH 7.0-7.5) |
| Semaglutide | GLP-1 Agonist | High (highly resistant to DPP-4) |
| Retatrutide | GLP-1/GIP/GCG Agonist | Moderate (complex triple-chain interaction) |
| Survodutide | Glucagon/GLP-1 Agonist | Moderate (sensitive to oxidation) |
Protocol for Reconstitution and Handling
To ensure the highest quality results, researchers should follow a standardized protocol for handling Tirzepatide. This minimizes mechanical stress and contamination.
- Equilibration: Allow the vial to reach room temperature before opening. This prevents moisture from the air from condensing on the lyophilized powder, which could lead to immediate degradation.
- Solvent Selection: Use sterile, preservative-free water for injection or PBS. If the research requires long-term use of the same vial, Bacteriostatic Water (with 0.9% benzyl alcohol) may be used, though some researchers prefer avoiding benzyl alcohol due to its potential impact on cell culture assays.
- Gentle Mixing: Slowly introduce the diluent down the side of the vial. Do not shake. Gently swirl the vial until the powder is completely dissolved. Tirzepatide should form a clear, colorless solution.
- Aliquotting: If the total volume is not needed at once, aliquot the reconstituted solution into smaller, single-use sterile vials and store them at -20°C. This avoids the damage caused by frequent freeze-thaw cycles.
The Consequences of Degradation in Research
What happens when a researcher uses degraded Tirzepatide? The implications are significant for data integrity:
- Reduced Potency: Deamidation or hydrolysis reduces the concentration of active peptide, leading to underestimation of the compound's efficacy.
- Immunogenicity: Aggregated peptides can induce an immune response in animal models, leading to the formation of anti-drug antibodies (ADAs). This can neutralize the peptide and alter its pharmacokinetic profile.
- Off-Target Effects: Degradation products may interact with receptors in ways the parent molecule does not, introducing confounding variables into the study.
Researchers should always verify their peptide's purity through COA documents and consider periodic testing if a study spans several months.
Conclusion: Setting the Standard for Metabolic Research
Ensuring the stability of research-grade Tirzepatide is not merely a matter of convenience; it is a foundational requirement for scientific validity. By controlling temperature, managing pH, and protecting against light and oxidation, researchers can ensure that the biological responses they observe are truly the result of Tirzepatide’s dual agonism and not the result of degradation artifacts.
For those expanding their research into related pathways, Alpha Carbon Labs provides a comprehensive range of metabolic peptides, including AOD9604 for adipolytics, Ipamorelin for growth hormone modulation, and 5-amino-1mq for NNMT inhibition. Maintaining the same rigorous storage standards across all research materials ensures the reproducibility and excellence of your laboratory’s output.
References
- 1. Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
- 2. Silva FM, et al. Peptide Stability and Storage: Key Considerations for Research. Journal of Peptide Science. 2022;28(9):e3421.
- 3. Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
- 4. Manning MC, et al. Stability of Protein Pharmaceuticals: An Update. Pharmaceutical Research. 2010;27(4):544-575.
- 5. D'Antonio EL, et al. The effect of temperature and pH on peptide stability: A comprehensive review. Analytical Biochemistry. 2021;630:114321.
- 6. Finan B, et al. A strategically designed multi-agonist of GLP-1 and GIP receptors for treatment of obesity and diabetes. Science Translational Medicine. 2013;5(209):209ra151.
- 7. Ghasemi S, et al. Chemical and physical stability of therapeutic peptides. Journal of Pharmaceutical Analysis. 2020;10(2):113-121.
- 8. He P, et al. Deamidation of Asparagine and Glutamine Residues in Peptides and Proteins. Journal of Biological Chemistry. 2018;293(12):4321-4330.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.