Reconstitution Science: Standardizing High-Concentration Semaglutide Solutions for Longitudinal In-Vitro Assays
Learn the scientifically rigorous methods for reconstituting and standardizing high-concentration semaglutide solutions to ensure maximum stability and reproducibility in long-term research assays.
Introduction: The Critical Role of Reconstitution in Semaglutide Research
In the rapidly evolving landscape of metabolic research, Semaglutide has emerged as a cornerstone peptide for investigating GLP-1 receptor agonism. However, the transition from a lyophilized powder to a bioactive solution is a phase where experimental integrity is most vulnerable. Variability in reconstitution techniques can lead to peptide aggregation, degradation, and concentration inaccuracies, ultimately compromising the reproducibility of longitudinal in-vitro assays.
For researchers utilizing high-purity peptides, such as those verified through our COA documents, standardizing the reconstitution protocol is not merely a logistical step; it is a fundamental requirement for scientific rigor. This guide explores the molecular characteristics of semaglutide and provides a standardized framework for preparing high-concentration solutions suitable for complex research environments.
Molecular Architecture of Semaglutide
To understand the requirements for reconstitution, one must first examine the structural modifications that distinguish semaglutide from native GLP-1. Semaglutide is a 31-amino acid peptide with two key modifications: the substitution of alanine with alpha-aminoisobutyric acid (Aib) at position 8, and the attachment of a C18 fatty diacid side chain via a hydrophilic spacer to the lysine at position 26.
These modifications are designed to enhance albumin binding and resist enzymatic degradation by dipeptidyl peptidase-4 (DPP-4). From a laboratory perspective, the hydrophobic nature of the fatty acid side chain introduces specific challenges regarding solubility and micelle formation in aqueous environments. High-concentration solutions must be managed carefully to avoid entrapment of the peptide in non-bioavailable aggregates.
Solubility and Isoelectric Point (pI)
Semaglutide exhibits a specific solubility profile influenced by its isoelectric point, which is approximately 4.5. Preparing solutions at a pH close to the pI can lead to precipitation. Therefore, most standardized protocols utilize buffered solutions that maintain a pH between 7.0 and 8.0 to ensure maximum stability and solubility during longitudinal studies.
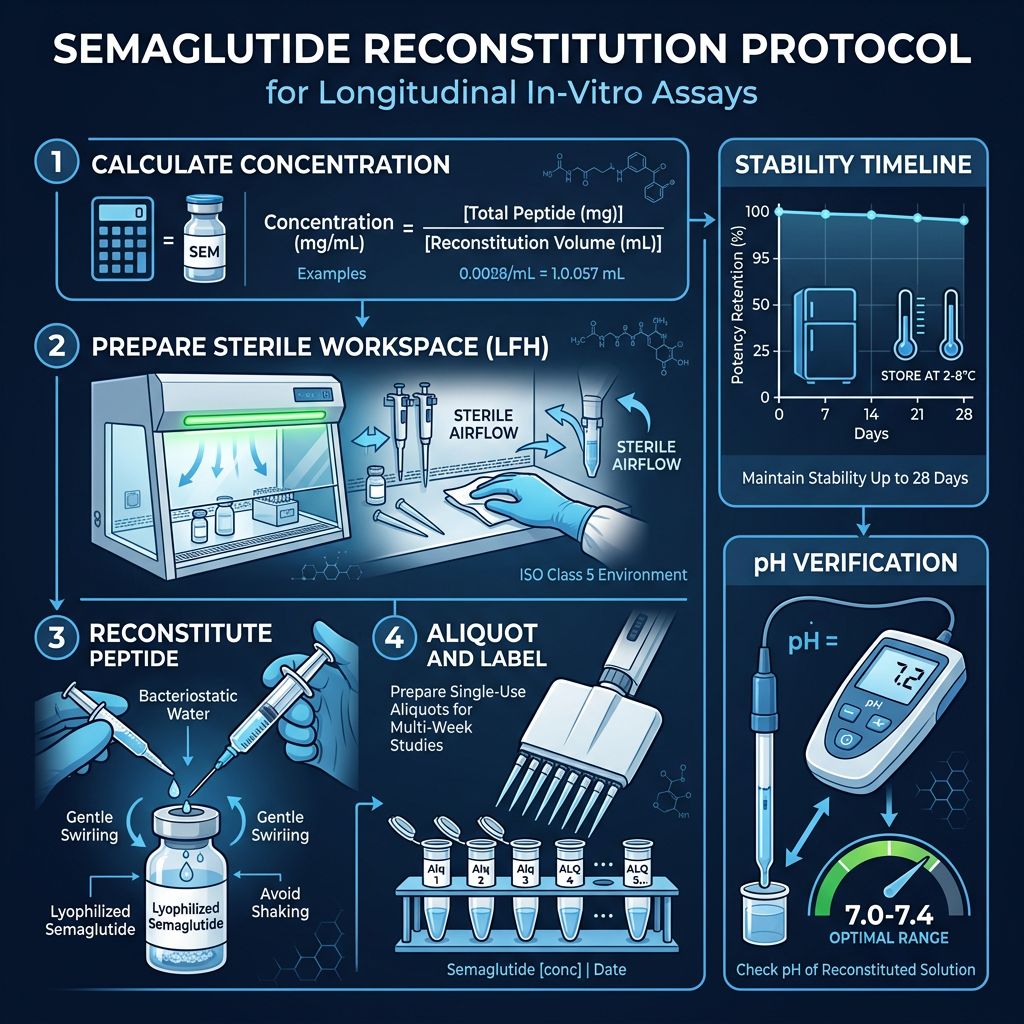
Advanced Reconstitution Protocols
Determining the Optimized Diluent
The choice of diluent is dictated by the intended application of the research. While Bacteriostatic Water is common for multi-use stability, sensitive cell culture assays often require sterile 0.9% Sodium Chloride (Normal Saline) or Phosphate-Buffered Saline (PBS). For high-concentration stocks, a specialized buffer may be necessary to mitigate the "salting out" effect observed with acylated peptides.
| Diluent Type | Primary Use Case | Pros | Cons |
|---|---|---|---|
| Bacteriostatic Water | Long-term repeated sampling | Inhibits microbial growth | Benzyl alcohol may affect cell viability in-vitro |
| 0.9% Normal Saline | Short-term in-vitro assays | High physiological compatibility | No antimicrobial properties |
| PBS (pH 7.4) | Stability-focused research | Excellent pH buffering | Can interact with certain metal ions |
Step-by-Step Standardization
- Temperature Equilibration: Remove the semaglutide vial from -20°C storage and allow it to reach room temperature (approx. 20-25°C) before opening. This prevents condensation from forming inside the vial, which can lead to premature peptide hydrolysis.
- Atmospheric Pressure Equalization: Using a sterile needle, equalize the pressure within the vacuum-sealed vial before introducing the diluent.
- Diluent Introduction: Using a precision pipette or syringe, slowly introduce the diluent along the inner wall of the vial. Direct impingement of the liquid onto the peptide cake can cause mechanical shearing and denaturation.
- Dissolution Strategy: Do NOT shake. Use a gentle swirling motion. For high concentrations (e.g., >5mg/mL), allow the vial to sit undisturbed for 10-15 minutes to ensure full hydration of the acylated side chains.
- Visual Inspection: The solution should be clear and free of particulate matter. Any turbidity indicates incomplete dissolution or aggregation.
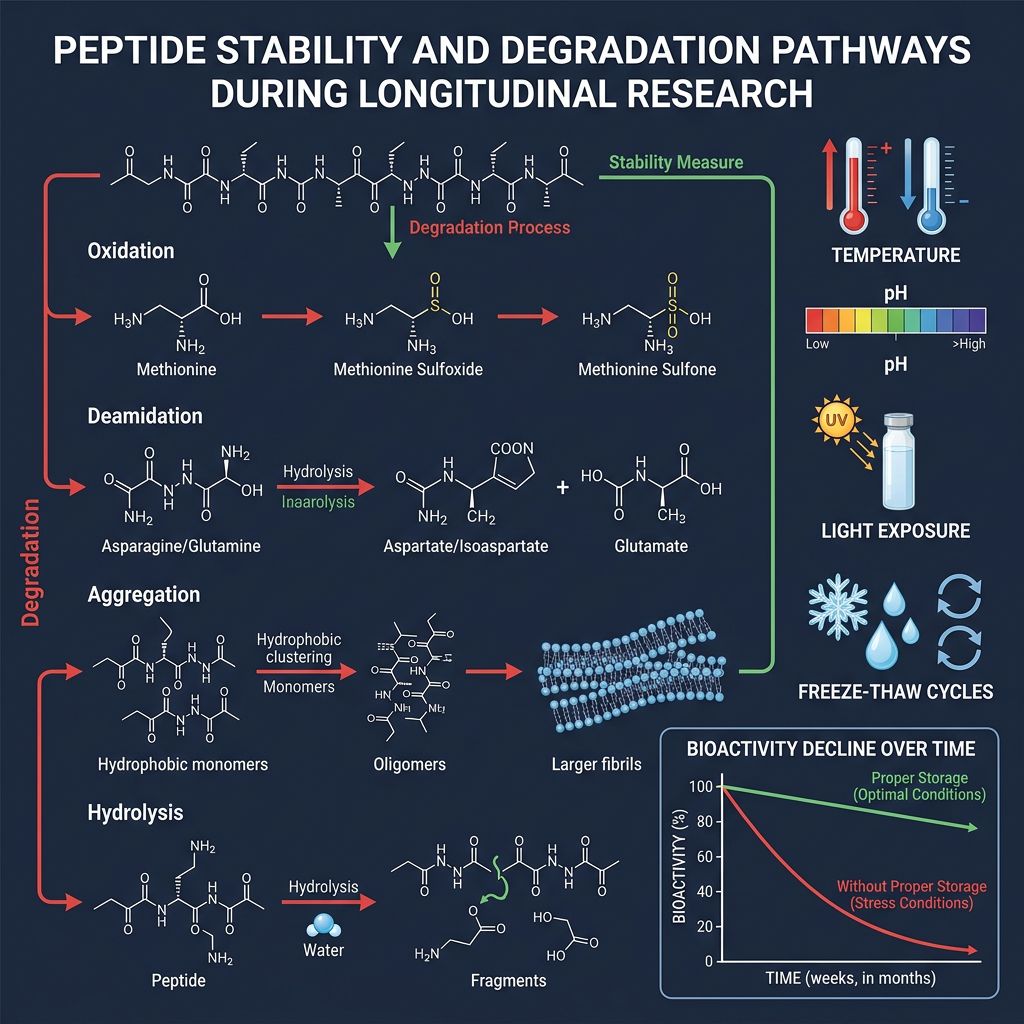
Ensuring Stability in Longitudinal Assays
Longitudinal research requires that the concentration of semaglutide remains constant over days or weeks of exposure to the experimental system. For researchers investigating comparative effects with other metabolic agents like Tirzepatide or Mazdutide, stability parity is essential.
Degradation Pathways
Peptides in solution are susceptible to several degradation pathways:
- Deamidation: Particularly at asparagine and glutamine residues.
- Oxidation: Often occurring at methionine or cysteine residues (though semaglutide lacks these, the C18 chain remains a pivot for oxidative stress in certain environments).
- Surface Adsorption: Peptides have a high affinity for plastic and glass surfaces. In low-concentration solutions, a significant percentage of the peptide can be "lost" to the walls of the container. Using Low Protein Binding (LPB) tubes is recommended for dilutions below 100µg/mL.
The Role of Cold Chain Integrity
At Alpha Carbon Labs, our peptide synthesis process includes rigorous lyophilization to ensure maximum stability. Once reconstituted, the "biological clock" begins. Research indicates that reconstituted semaglutide maintains >98% purity for up to 28 days when stored at 2-8°C, provided the diluent possesses antimicrobial properties.
Titration and Concentration Calculation
In high-concentration solutions, the volumetric displacement of the peptide powder itself must be accounted for in precise calculations. For a 5mg vial of Semaglutide, the powder volume is typically negligible below 2mg/mL, but in "high-concentration" protocols (up to 10mg/mL), the total volume of the solution may be slightly higher than the volume of diluent added.
Molar Concentration vs. Mass Concentration
Researchers often prefer working in Micromolar (µM) or Nanomolar (nM) concentrations for cell-based assays. The molecular weight of Semaglutide is approximately 4113.58 g/mol. A 1mg/mL solution is therefore approximately 243 µM. This conversion is crucial when comparing Semaglutide to dual or triple agonists such as Retatrutide or Cagrilintide.
Advanced Synergies in Metabolic Research
Increasingly, research is moving toward combinatorial treatments. Standardizing the reconstitution of semaglutide allows for more accurate data when studied alongside other research molecules. For instance, the combination of GLP-1 agonists with mitochondrial modulators like MOTS-c or metabolic regulators like 5-amino-1mq requires a standardized baseline to determine if synergistic effects are additive or potentiating.
Standardizing Multi-Peptide Blends
In certain research designs, multiple peptides are utilized simultaneously. While we offer pre-formulated options like the BPC-157 + TB-500 + GHK-Cu Blend, semaglutide is typically studied in isolation before moving to co-administration models with agents like AOD9604 or IGF-1LR3. The reconstitution protocol for each constituent must be individually validated to prevent cross-peptide aggregation or precipitation.
Quality Control and Verification
To maintain high standards in longitudinal assays, researchers should periodically verify the purity of their reconstituted stocks. Our quality control department utilizes High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) to ensure that the source material meets the highest standards of purity before it reaches your lab.
Researchers can replicate this oversight by performing a simple UV-Vis spectrophotometry test to confirm concentration, using the extinction coefficient of semaglutide to ensure that the reconstitution process has achieved the intended molarity.
Safety and Handling in the Laboratory
Semaglutide is a highly potent peptide. Lab safety protocols must include:
- Use of a certified laminar flow hood for reconstitution to maintain sterility.
- Double-gloving during the handling of lyophilized powder to prevent accidental inhalation or dermal absorption.
- Proper disposal of used vials and needles in biohazard containers.
Conclusion
The success of in-vitro research involving Semaglutide depends heavily on the precision of the initial reconstitution. By standardizing the diluent choice, temperature management, and mixing mechanics, researchers can minimize variables that lead to experimental noise. As the field expands into more complex molecules like Survodutide and SLU-PP-332, these foundational principles of peptide science will remain the bedrock of reliable, peer-reviewed discovery.
For more technical data or to view our latest batch results, please visit our COA Documents page or contact our technical support team for detailed specification sheets.
References
- 1. Mahapatra MK, Karuppasamy M, Sahoo BM. Semaglutide, a once weekly human GLP-1 analogue, for type 2 diabetes mellitus. Curr Drug Metab. 2022;23(5):372-384.
- 2. Christie JT, et al. Peptide Aggregation and Stability: Challenges in Formulation. Journal of Pharmaceutical Sciences. 2021;110(4):1500-1515.
- 3. Knudsen LB, Lau J. The Discovery and Development of Liraglutide and Semaglutide. Front Endocrinol (Lausanne). 2019;10:155.
- 4. Gomez-Zepeda D, et al. Mass Spectrometry-Based Characterization of Synthetic Peptides for Research. Analytical Chemistry. 2020;92(14):9432-9440.
- 5. Bower KC, et al. Preservation of Peptide Bioactivity During Lyophilization and Reconstitution. International Journal of Pharmaceutics. 2018;548(1):340-349.
- 6. Lau J, et al. Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. J Med Chem. 2015;58(18):7370-80.
- 7. Werle M, Bernkop-Schnürch A. Strategies to improve plasma half-life time of peptide and protein drugs. Amino Acids. 2006;30(4):351-67.
- 8. Finocchario CV, et al. Effects of pH and Ionic Strength on the Stability of Acylated Glucagon-like Peptide-1 Analogs. Pharmaceutical Research. 2022;39:1125–1139.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.