Quality Control in Peptide Synthesis: Ensuring Purity in Retatrutide Research Flux
Unlock the secrets of Retatrutide research with our deep dive into the HPLC and MS quality standards required for precise metabolic studies and high-purity synthesis.
The Critical Role of Purity in Metabolic Research
The landscape of metabolic research has been fundamentally altered by the emergence of multi-receptor agonists. Among these, Retatrutide (LY3437943) stands as a pinnacle of molecular engineering, targeting the glucose-dependent insulinotropic polypeptide (GIP) receptor, the glucagon-like peptide-1 (GLP-1) receptor, and the glucagon receptor (GCGR). For researchers investigating insulin sensitivity, lipid metabolism, and thermogenesis, the integrity of the peptide sequence is paramount. Even minor deviations in purity can yield non-replicable data, activate off-target pathways, or induce confounding inflammatory responses in in vitro and in vivo models.
At Alpha Carbon Labs, we recognize that the validity of your research hinges on the chemical precision of your reagents. This guide delves into the rigorous standards of peptide synthesis, the interpretation of analytical documentation, and the specific challenges associated with synthesizing complex triple agonists like Retatrutide.
The Architecture of Retatrutide: A Synthetic Challenge
Retatrutide is a 39-amino acid backbone peptide, modified with a C18 fatty acid diacid moiety via a linker. This modification, intended to extend the half-life through albumin binding, significantly increases the complexity of the synthesis process. Unlike simpler peptides such as BPC-157, Retatrutide requires specialized Solid Phase Peptide Synthesis (SPPS) protocols to prevent "difficult sequences" where steric hindrance or aggregation occurs during chain elongation.
When sourcing research materials, understanding the difference between crude synthesis and pharmaceutical-grade purification is essential. High-grade Retatrutide must undergo multiple rounds of High-Performance Liquid Chromatography (HPLC) to isolate the target molecule from truncated sequences, diastereomers, and residual solvents.
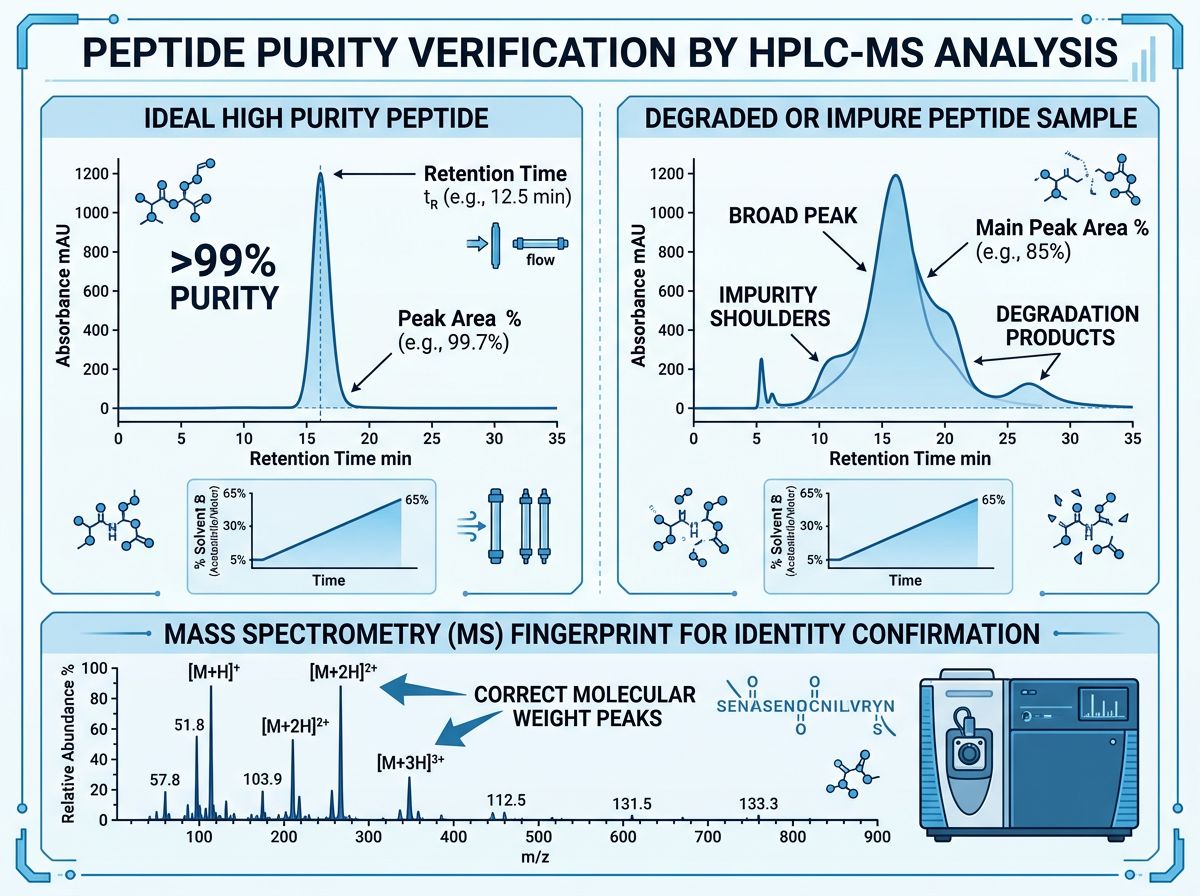
Advanced HPLC Analysis: Interpreting the Chromatogram
High-Performance Liquid Chromatography (HPLC) is the gold standard for determining the chemical purity of a peptide. For a researcher, the Certificate of Analysis (CoA) should provide an HPLC trace that demonstrates a single, sharp peak with minimal "noise" or secondary humps.
Key HPLC Parameters for Retatrutide
- Retention Time (Rt): The time it takes for the peptide to pass through the column. This must be consistent across batches to ensure batch-to-batch reproducibility.
- Peak Area Percentage: This represents the relative purity. For high-stakes metabolic studies, a purity of >99% is often required to ensure that observed physiological effects are solely attributable to the triple-agonist activity.
- Gradient Profile: The specific ratio of mobile phases (typically water and acetonitrile with TFA) used to elute the peptide.
In many instances, low-quality suppliers may present an HPLC graph with a "broad" peak. This often indicates the presence of closely related impurities, such as deamidated or oxidized forms of the peptide. In metabolic flux studies, these impurities may still bind to receptors but with different affinities (Ki), leading to skewed EC50 or IC50 values.
Mass Spectrometry (MS): Confirming Molecular Identity
While HPLC tells us how pure a substance is, Mass Spectrometry tells us exactly what that substance is. For Retatrutide, the theoretical molecular weight is approximately 5250.3 Da (depending on the specific salt form). MS analysis generates a "fingerprint" of the molecule based on its mass-to-charge (m/z) ratio.
Detecting Synthetic Errors
A high-resolution electrospray ionization (ESI-MS) spectrum should show clear peaks corresponding to the [M+H]+, [M+2H]2+, or [M+3H]3+ ions. Researchers should look for:
- Deletion Sequences: Peaks that are roughly 57-186 Da lighter than the target, suggesting an amino acid was missed during synthesis.
- Incomplete Deprotection: Peaks that are heavier than the target, indicating that protecting groups (like Fmoc or Boc) were not fully removed.
- Adducts: Sodium or potassium adducts that can interfere with solubility and binding.
At Alpha Carbon Labs, every batch of Retatrutide undergoes rigorous MS verification to ensure that the primary structure is identical to the sequence validated in clinical literature, such as those described in studies involving Tirzepatide and other incretin mimetics.
A Comparison of Metabolic Research Peptides
Researchers often compare Retatrutide with other agonists to map differential effects on glucagon signaling vs. insulinotropic pathways. The following table highlights the comparative focus areas and the necessity of purity for each:
| Peptide | Target Receptors | Primary Research Focus | Purity Requirement |
|---|---|---|---|
| Retatrutide | GIP, GLP-1, GCGR | Triple Agonism / Maximum Weight Loss / Hepatic Steatosis | >99% (Critical due to 3-way signaling) |
| Tirzepatide | GIP, GLP-1 | Dual Incretin Agonism / Glycemic Control | >98% |
| Semaglutide | GLP-1 | Mono-Agonism / Appetite Suppression | >98% |
| Cagrilintide | Amylin Receptor | Satiety / Combination Therapy with GLP-1 | >98% |
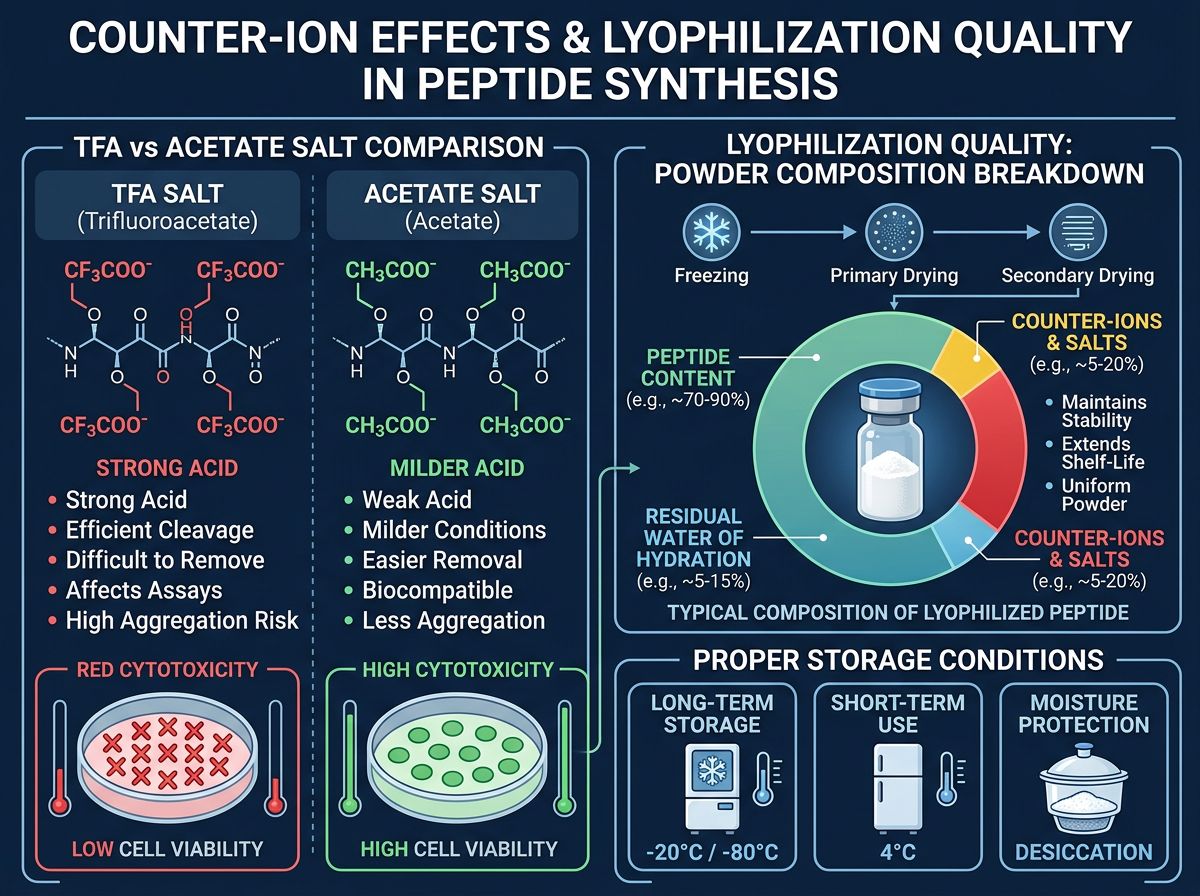
The Impact of Counter-Ions and Residuals
Beyond the peptide itself, the surrounding chemical environment plays a major role in research outcomes. During synthesis, peptides are often eluted using Trifluoroacetic Acid (TFA). While TFA is a standard reagent, high concentrations can be cytotoxic to certain cell cultures used in metabolic research.
Acetate vs. TFA Salt
Most research-grade peptides are provided as TFA salts. However, for sensitive in vitro assays monitoring mitochondrial function—similar to studies performed with SS-31 or MOTS-c—researchers may require an acetate exchange. This process replaces the TFA ions with acetate, which is generally better tolerated by cells but increases the cost of production due to the additional purification steps required. Alpha Carbon Labs maintains strict quality control to ensure residual TFA levels are minimized and documented.
Water Content and Lyophilization Excellence
The physical state of the peptide, typically a white lyophilized powder, hides a key variable: water and solvent content. A peptide "purity" of 99% refers to the peptide fraction. However, the total "peptide content" within the vial might be 80-85%, with the remainder being water of hydration and counter-ions. In quantitative flux studies, failing to account for net peptide content can lead to a 15-20% error in calculated concentrations.
Researchers should always calculate their molarity based on the net peptide content, not the gross weight of the powder. This level of precision is what separates high-impact research from preliminary pilot data.
Storage and Stability in Flux
Retatrutide, like GHK-Cu and IGF-1LR3, is susceptible to degradation if not stored correctly. Oxidation of methionine or tryptophan residues (if present in the sequence) and deamidation of asparagine or glutamine can occur if the peptide is exposed to moisture or repeated freeze-thaw cycles. We recommend aliquoting reconstituted Retatrutide into single-use vials and storing lyophilized stock at -20°C or -80°C.
Conclusion: The Alpha Carbon Labs Commitment
In the rapidly evolving field of metabolic research, the quality of your Retatrutide is the foundation of your discoveries. Whether you are investigating its effects in conjunction with 5-amino-1mq for adipocyte modulation or exploring its neuroprotective potential alongside Cerebrolysin, the purity of the molecule is non-negotiable.
Alpha Carbon Labs provides the research community with top-tier analytical data, ensuring that every vial of Retatrutide meets the stringent requirements of modern science. By emphasizing HPLC/MS transparency and rigorous synthesis standards, we empower researchers to push the boundaries of what is possible in metabolic health and beyond.
Explore our full range of research tools, from growth factor mimetics like CJC-1295 to metabolic enhancers like AICAR, and ensure your lab is equipped with the purest compounds available.
References
- 1. Frias JP, et al. (2022). Retatrutide, a GIP, GLP-1, and Glucagon Receptor Agonist, for Glycemic Control and Weight Reduction. New England Journal of Medicine.
- 2. Mumenthaler SM, et al. (2021). Importance of Peptide Purity in Preclinical Research. Journal of Pharmaceutical Sciences.
- 3. D'Hondt M, et al. (2014). Related impurities in peptide medicines. Journal of Pharmaceutical and Biomedical Analysis.
- 4. Andersson L, et al. (2000). Large-scale synthesis of peptides. Biopolymers (Peptide Science).
- 5. Erak M, et al. (2018). Peptide chemistry and the next wave of peptide drugs. Expert Opinion on Drug Discovery.
- 6. Coskun T, et al. (2022). LY3437943 (Retatrutide), a novel triple GIP, GLP-1, and glucagon receptor agonist. Cell Metabolism.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.