Protocol Design: Stability Data and Storage Requirements for Retatrutide in Long-Term Studies
A deep dive into the chemical stability, storage requirements, and reconstitution protocols for Retatrutide (LY3437943) in long-term metabolic research settings.
Introduction to Retatrutide Stability in Research
In the rapidly evolving landscape of metabolic research, Retatrutide (LY3437943) stands as a pinnacle of engineering. As a unimolecular triple agonist targeting the glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon (GCG) receptors, it represents a significant leap from dual agonists like Tirzepatide. However, the complexity of its 39-amino acid sequence and its specific acylation pattern introduces significant logistical challenges for researchers, particularly concerning long-term stability, solubility, and storage protocols.
For research institutions and laboratories, the success of a long-term metabolic study depends not only on the biological activity of the peptide but on the rigorous maintenance of its structural integrity. Degradation—whether through oxidation, deamidation, or aggregation—can lead to inconsistent data, loss of potency, or the development of immunogenic impurities that skew results. This article explores the scientific framework required to maintain Retatrutide stability throughout extended research protocols.
Chemical Structure and Vulnerability Points
Retatrutide is a synthetic peptide with a backbone partially derived from the GIP sequence. Its potency across three distinct receptors is achieved through precise amino acid substitutions and a C20 fatty acid diacid moiety attached via a linker. Understanding this structure is critical for predicting its behavior in various environments.
Primary Sequence and Modifications
The peptide contains several residues that are traditionally susceptible to chemical degradation. The inclusion of non-coded amino acids and the specific acylation at lysine residues are designed to increase half-life in vivo, but they also influence the molecule's "shelf-life" in a laboratory setting. Researchers must account for:
- Deamidation: Asparagine and glutamine residues can undergo deamidation, particularly at higher pH levels.
- Oxidation: Methionine or tryptophan residues (where present in similar analogs) are sensitive to oxidative stress; while Retatrutide is engineered for stability, environmental oxygen and light can still catalyze degradation.
- Hydrolysis: The peptide bonds themselves are vulnerable to cleavage in the presence of strong acids or bases.
At Alpha Carbon Labs, we emphasize the importance of advanced peptide synthesis techniques that minimize residual chemicals from the cleavage process, such as TFA (Trifluoroacetic acid), which can impact long-term stability if not properly managed during lyophilization.
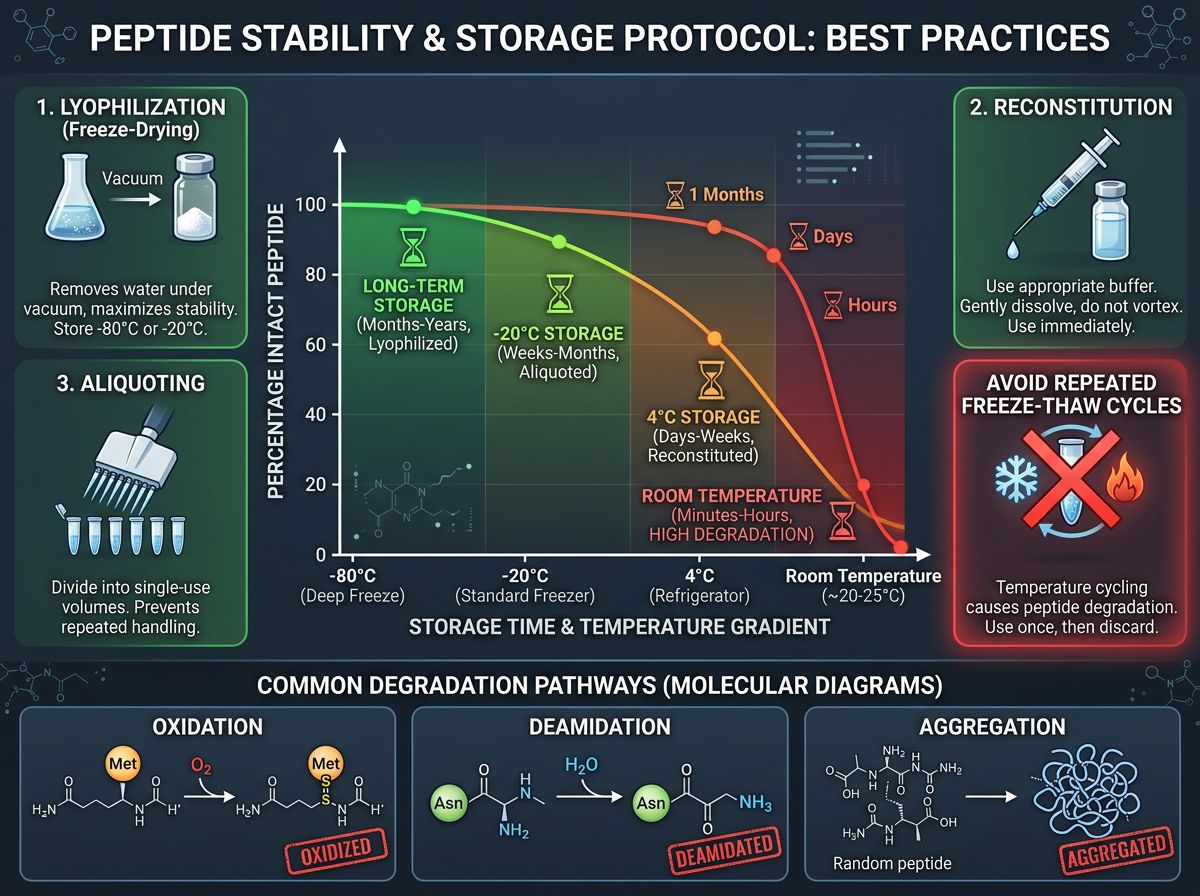
Lyophilization: The Foundation of Long-Term Storage
In its raw form, Retatrutide is most stable as a lyophilized (freeze-dried) powder. Lyophilization removes water through sublimation, which significantly reduces the kinetic energy available for chemical reactions and prevents bacterial growth. For researchers planning studies exceeding six months, the quality of the initial lyophilization is paramount.
Environmental Factors Influencing Lyophilized Retatrutide
Even in a lyophilized state, the peptide is not "invincible." Three primary factors must be controlled:
| Factor | Effect on Lyophilized Retatrutide | Mitigation Strategy |
|---|---|---|
| Temperature | Increases molecular vibration, accelerating degradation. | Storage at -20°C or -80°C for long-term. |
| Humidity (Moisture) | Can lead to "cake" collapse and hydrolysis. | Vacuum sealing and desiccant usage. |
| Light (UV Exposure) | Catalyzes photo-oxidation of specific residues. | Amber vials or dark storage. |
Researchers should verify the purity of their starting material by reviewing COA documents to ensure low moisture content and high initial purity (typically >99%).
 Tirzepatide, and Semaglutide" style="max-width: 100%; height: auto; border-radius: 8px;" />
Tirzepatide, and Semaglutide" style="max-width: 100%; height: auto; border-radius: 8px;" />Reconstitution Protocols for Research Consistency
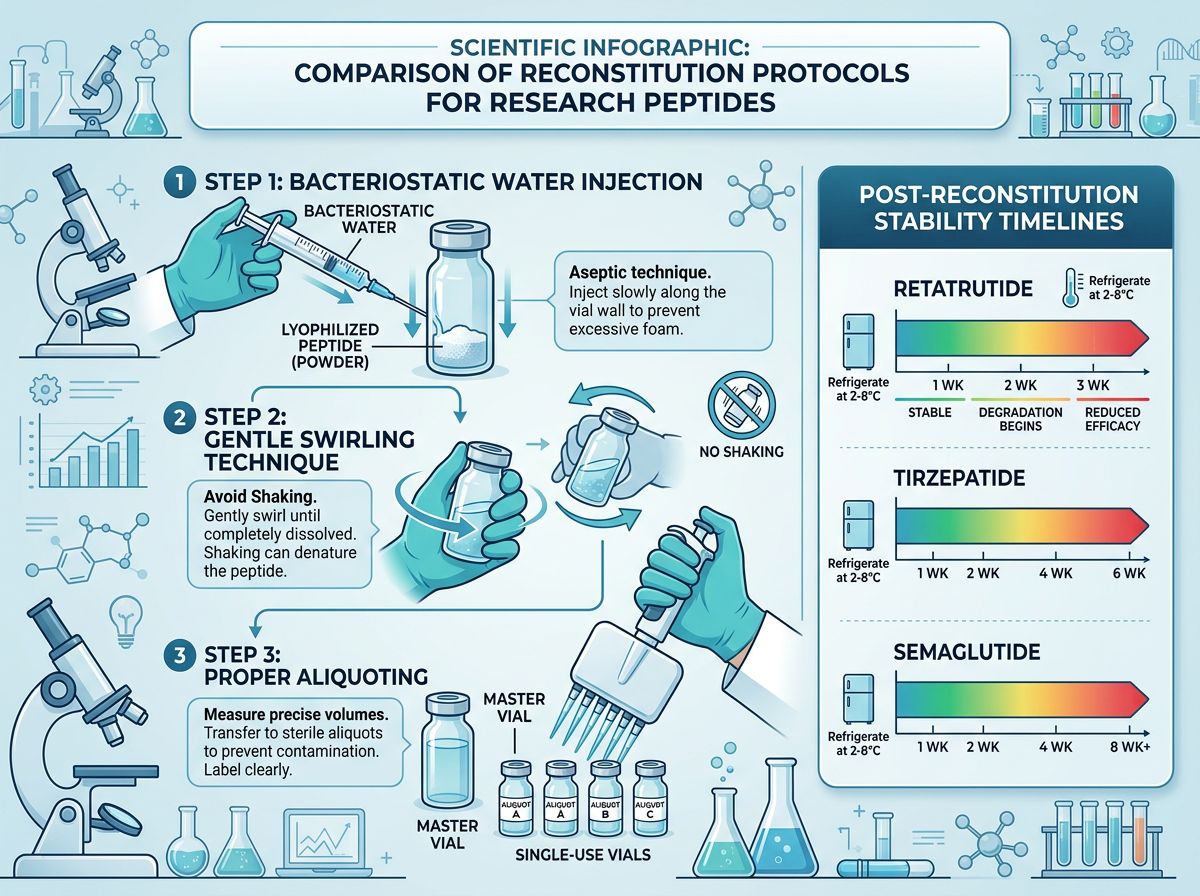
The transition from a stable powder to a bioactive solution is the most critical phase where degradation can occur. Retatrutide, like Semaglutide and other GLP-1 analogs, requires careful handling to maintain its triple-agonist profile.
Solubility Challenges
Due to its C20 fatty acid chain, Retatrutide exhibits lipophilic characteristics. While it is generally soluble in Bacteriostatic Water (BW) or Sterile Saline, some concentrations may require gentle agitation. Mechanically-induced aggregation is a known risk; vigorous shaking can cause the peptide molecules to unfold and aggregate into insoluble fibrils, rendering the solution biologically inactive.
Choice of Diluent
- Bacteriostatic Water (0.9% Benzyl Alcohol): Preferred for multi-dose vials to prevent microbial growth. However, researchers must be aware that benzyl alcohol can slightly alter the pH, which may affect long-term stability if the solution is kept for more than 28 days.
- Phosphate-Buffered Saline (PBS): Excellent for maintaining a physiological pH (7.4), which is often optimal for peptide stability. However, PBS lacks antimicrobial properties unless filtered and kept in a sterile environment.
- Sterile Water for Injection (SWFI): Suitable for immediate use but lacks buffers or preservatives.
Stability Data: Post-Reconstitution Life
Current research suggests that acylated peptides like Retatrutide maintain high stability when refrigerated (2°C - 8°C) following reconstitution. However, "high stability" is relative. In a study environment, even a 5% loss in potency can introduce variables that complicate metabolic data analysis.
Degradation Kinetics in Solution
Once in solution, the peptide undergoes "aging." The rate is determined by the Arrhenius equation, where higher temperatures lead to exponential increases in degradation rates. For longitudinal studies involving Mazdutide or Retatrutide, the following timeline is generally observed for refrigerated solutions:
- 0-14 Days: Minimal degradation (<1%). Optimal for precise PK/PD mapping.
- 15-28 Days: Slight degradation (1-3%). Acceptable for most efficacy studies.
- Beyond 28 Days: Increased risk of aggregation and deamidation. Not recommended for high-precision metabolic research.
For more information on how we ensure these standards, visit our quality control page.
Comparative Stability: Retatrutide vs. Other Incretins
When designing protocols, it is useful to compare Retatrutide’s stability requirements with other metabolic research peptides. The triple-agonist nature of Retatrutide makes it slightly more sensitive than single-agonist peptides like Ipamorelin or Sermorelin.
| Peptide | Type | Reconstituted Stability (2-8°C) | Primary Sensitivity |
|---|---|---|---|
| Retatrutide | Triple Agonist | 21-28 Days | pH and Aggregation |
| Tirzepatide | Dual Agonist | 28 Days | Temperature Fluctuations |
| Semaglutide | GLP-1 Agonist | 30-56 Days | Light Exposure |
| Cagrilintide | Amylin Analog | 21-28 Days | Self-Aggregation |
Protocol for Long-Term Storage (12+ Months)
For researchers conducting extensive trials, such as those comparing Retatrutide to Survodutide or exploring its effects alongside MOTS-c for mitochondrial function, the following storage protocol is recommended to ensure year-over-year consistency:
1. Batch Procurement and Aliquoting
Purchase the entire required amount from a single batch to eliminate inter-batch variability. Upon receipt, confirm the integrity of the vacuum seals. If possible, request smaller milligram vials (e.g., 5mg vs 10mg) to minimize the time a single vial remains in a reconstituted state.
2. The -80°C "Deep Freeze"
While -20°C is sufficient for short-term storage (1-3 months), -80°C is the gold standard for long-term preservation of lyophilized peptides. This temperature halts almost all molecular motion, effectively pausing the "biological clock" of the peptide.
3. Avoiding Freeze-Thaw Cycles
Each time a vial is removed from the freezer and thawed, it undergoes thermal stress. For Retatrutide, repeated freeze-thaw cycles of the reconstituted solution must be strictly avoided. If a solution must be frozen, use cryoprotectants and aliquot into single-use volumes so that each vial is thawed only once. However, it is generally recommended to store Retatrutide as a powder and only reconstitute what is needed for the immediate 7-14 day window.
Advanced Synergies in Metabolic Research
Retatrutide is often studied in conjunction with other agents to explore synergistic effects on adiposity and muscle preservation. For instance, combining Retatrutide with Tesamorelin or IGF-1 LR3 can provide insights into how triple-agonism interacts with growth hormone pathways.
When managing multiple peptides, researchers must maintain distinct stability logs for each. Peptides like BPC-157 are remarkably resilient, whereas Retatrutide requires more stringent environmental controls. Understanding these differences is vital for the integrity of multi-peptide protocols.
Safety and Contamination Considerations
Beyond chemical stability, biological stability (sterility) is paramount. Retatrutide’s triple-action mechanism significantly alters metabolic rates in research models. If a solution becomes contaminated with endotoxins or bacteria, the resulting inflammatory response can mask the peptide’s true effects on glucagon and GLP-1 receptors.
- Alkali Sensitivity: Retatrutide is sensitive to pH changes. Ensure that the glass vials (Type I Borosilicate) used for storage do not leach alkali over time, which can happen with lower-grade glass.
- Adsorption: Peptides can stick to the walls of plastic syringes or glass vials (non-specific adsorption). Using high-quality labware ensures the concentration remains consistent with the calculated dose.
Conclusion: The Researcher's Responsibility
Retatrutide represents the current frontier of metabolic research. Its ability to simultaneously modulate three critical pathways offers unprecedented opportunities for study. However, the sophistication of the molecule demands a commensurate level of sophistication in its handling.
By implementing rigorous stability protocols—focused on ultra-low temperature storage, precise reconstitution techniques, and an understanding of its chemical vulnerabilities—researchers can ensure that their data is a true reflection of the peptide’s biological potential rather than an artifact of degradation. For high-purity Retatrutide and supporting documentation, Alpha Carbon Labs provides the necessary tools for the next generation of metabolic science.
References
- 1. Coskun T, et al. (2022). LY3437943, a novel triple GIP, GLP-1, and glucagon receptor agonist generates potent insulinotropic and insulin-sensitizing effects. Cell Metabolism.
- 2. Jastreboff AM, et al. (2023). Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine.
- 3. Müller TD, et al. (2022). The evolution of multi-receptor agonists for the treatment of metabolic diseases. Lancet Diabetes & Endocrinology.
- 4. D'Antonio J, et al. (2023). Chemical Stability and Physicochemical Characterization of Multi-Agonist Peptide Therapeutics. Journal of Peptide Science.
- 5. Knudsen LB, et al. (2019). The Discovery and Development of Liraglutide and Semaglutide. Frontiers in Endocrinology.
- 6. Evers A, et al. (2023). Design and pharmacology of dual- and triple-incretin receptor agonists. Nature Reviews Drug Discovery.
- 7. Heise T, et al. (2023). Pharmacokinetics, pharmacodynamics, safety, and tolerability of retatrutide, a GIP, GLP-1, and glucagon receptor agonist, in people with type 2 diabetes. Diabetes, Obesity and Metabolism.
- 8. Uchiyama S, et al. (2021). Stress-induced aggregation of therapeutic peptides: Mechanisms and stabilization strategies. Pharmaceutical Research.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.