Pharmacological Exercise: Analyzing the ERR Alpha Agonism of SLU-PP-332 for Metabolic Optimization
SLU-PP-332 represents a breakthrough in metabolic research, functioning as a direct ERRα agonist that mimics the genetic and cellular effects of endurance exercise. This article analyzes its mechanism of action, compares it with older mimetics like AICAR, and explores its potential for fat loss and mitochondrial optimization.
Introduction: The Evolution of Exercise Mimetics
The quest to replicate the physiological benefits of physical exercise through pharmacological intervention has long been a "holy grail" in metabolic research. Exercise is a potent systemic stimulus that remodels skeletal muscle, enhances mitochondrial density, improves insulin sensitivity, and oxidizes adipose tissue. However, for populations suffering from sarcopenia, severe obesity, or heart failure, the physical capacity to exercise sufficiently to trigger these adaptations is often compromised. This clinical gap has driven the investigation into "exercise mimetics"—compounds that activate the genetic and metabolic pathways typically triggered by endurance training.
Historically, research has focused on the AMP-activated protein kinase (AMPK) pathway and the Peroxisome Proliferator-Activated Receptor delta (PPARδ) axis. Agents such as AICAR established the proof of concept that chemical signaling could induce fatigue resistance and fatty acid oxidation in sedentary models. Yet, the field is currently undergoing a paradigm shift with the emergence of direct Estrogen-Related Receptor alpha (ERRα) agonists.
Among the most promising of these novel agents is SLU-PP-332. Unlike its predecessors, SLU-PP-332 targets a nuclear receptor directly responsible for the transcriptional control of mitochondrial biogenesis and oxidative phosphorylation. This article provides a rigorous analysis of the pharmacological mechanisms of SLU-PP-332, comparing its efficacy and safety profile to established mimetics like AICAR, and exploring its potential role in the landscape of metabolic optimization and obesity research.
The Biological Target: Estrogen-Related Receptor Alpha (ERRα)
To understand the unique value proposition of SLU-PP-332, one must first appreciate the biological machinery it manipulates. The Estrogen-Related Receptor alpha (ERRα) is an orphan nuclear receptor—"orphan" because for decades, its endogenous ligand was unknown or considered non-existent. Despite its name, ERRα does not bind estrogen. Instead, it functions as a master regulator of cellular energy metabolism.
Transcriptional Co-Activation and PGC-1α
ERRα activity is tightly coupled with the Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha (PGC-1α). PGC-1α is often described as the master regulator of mitochondrial biogenesis. When physiological stress (like exercise) occurs, PGC-1α expression rises, binding to various transcription factors to turn on genes for energy production. ERRα is the primary DNA-binding partner for PGC-1α in the context of mitochondrial respiration.
Research has demonstrated that mice lacking ERRα cannot upregulate oxidative metabolism in response to cold or exercise, rendering them intolerant to physical stress. Conversely, activation of the ERRα/PGC-1α complex drives the expression of enzymes involved in the tricarboxylic acid (TCA) cycle, the electron transport chain, and fatty acid beta-oxidation.
The Druggability Challenge
Historically, targeting orphan nuclear receptors has been chemically challenging. Most nuclear receptors require a ligand binding pocket that accepts a specific hormone. Because ERRα is constitutively active (always "on" to some degree) and relies on co-activator presence rather than a binding hormone, designing a synthetic small molecule to further agonize it was difficult. SLU-PP-332 represents a breakthrough in medicinal chemistry as a potent, specific, synthetic agonist that stabilizes the active conformation of ERRα, recruiting co-activators more efficiently and driving gene expression supranormally.
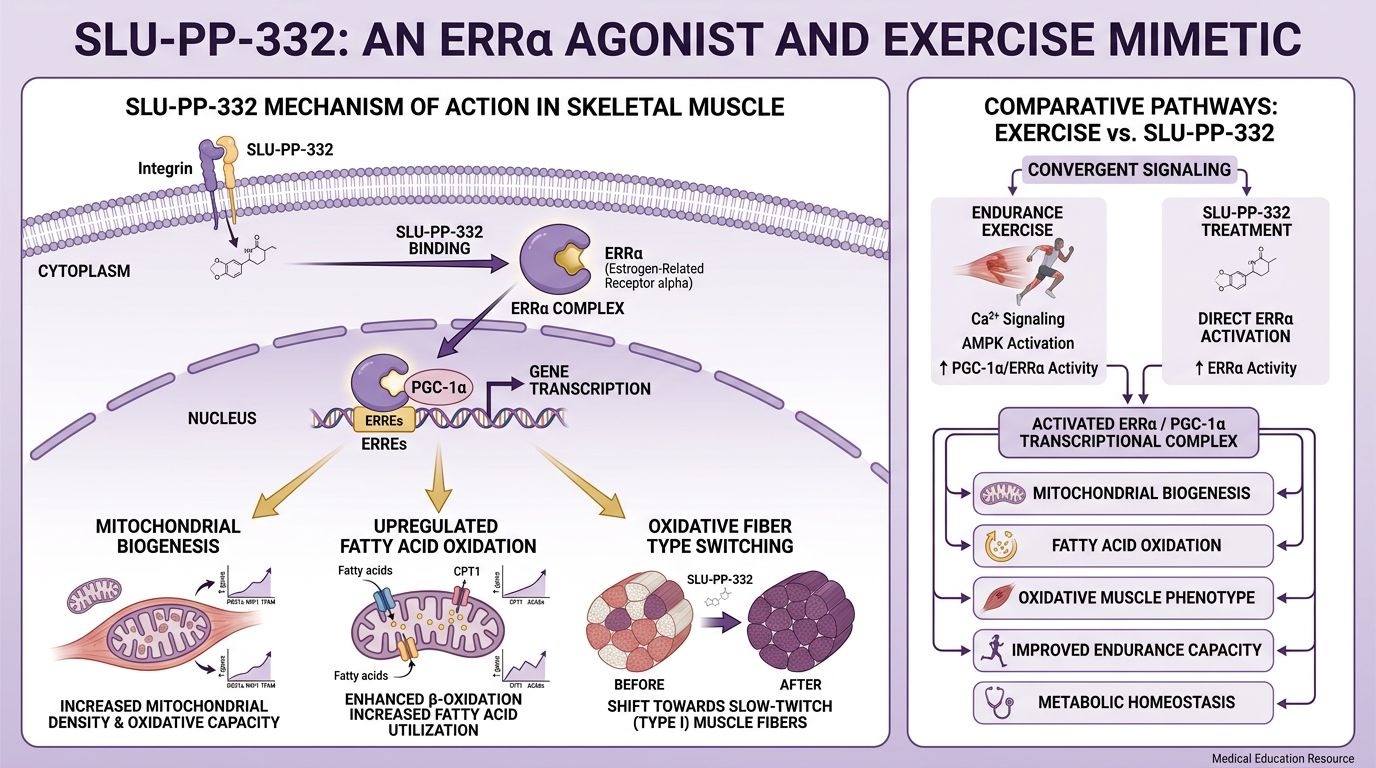
SLU-PP-332: Mechanism of Action and Pharmacodynamics
SLU-PP-332 is a novel chemical entity designed to enhance the transcriptional activity of ERRα. Its mechanism differs fundamentally from appetite suppressants or stimulants. It does not act on the central nervous system to reduce caloric intake; rather, it fundamentally alters the bioenergetics of skeletal muscle and adipose tissue.
Promotion of Oxidative Fiber Transformation
Skeletal muscle fibers typically exist on a spectrum from Type I (slow-twitch, oxidative, fatigue-resistant) to Type IIb (fast-twitch, glycolytic, fatigue-prone). Type IIa fibers are an intermediate, highly adaptable phenotype. Exercise training typically shifts muscle composition toward Type I and Type IIa, increasing mitochondrial volume.
Administration of SLU-PP-332 in murine models has been shown to mimic this shift. By activating ERRα, the compound upregulates genes characteristic of oxidative fibers (such as Myh7 and Mb for myoglobin) even in the absence of physical training. This results in a muscle phenotype that preferentially burns fatty acids for fuel rather than glycogen, preserving glucose for vital organs and delaying the onset of metabolic acidosis (the "burn") during exertion.
Fatty Acid Oxidation Over Glucose Utilization
A distinctive feature of SLU-PP-332 is its bias toward fatty acid oxidation (FAO). In sedentary obese mice, SLU-PP-332 treatment increases whole-body energy expenditure. Crucially, respiratory exchange ratio (RER) data indicates a shift toward lipid utilization. This is significant for metabolic syndrome research, where "metabolic inflexibility"—the inability to switch from glucose to fat burning—is a hallmark of pathology. By forcing the cellular machinery to process lipids, SLU-PP-332 directly combats lipid toxicity in muscle and liver tissue.
The "Silent" Weight Loss Effect
Perhaps the most compelling data regarding SLU-PP-332 surrounds its effect on body weight composition. In studies published in journals such as the Journal of Pharmacology and Experimental Therapeutics, mice treated with the agonist lost fat mass while maintaining lean muscle mass, despite having identical food intake to the control group. This suggests that the weight loss is purely a function of increased metabolic rate and futile cycling of energy substrates, rather than anorectic effects common to GLP-1 agonists like Semaglutide or Tirzepatide.
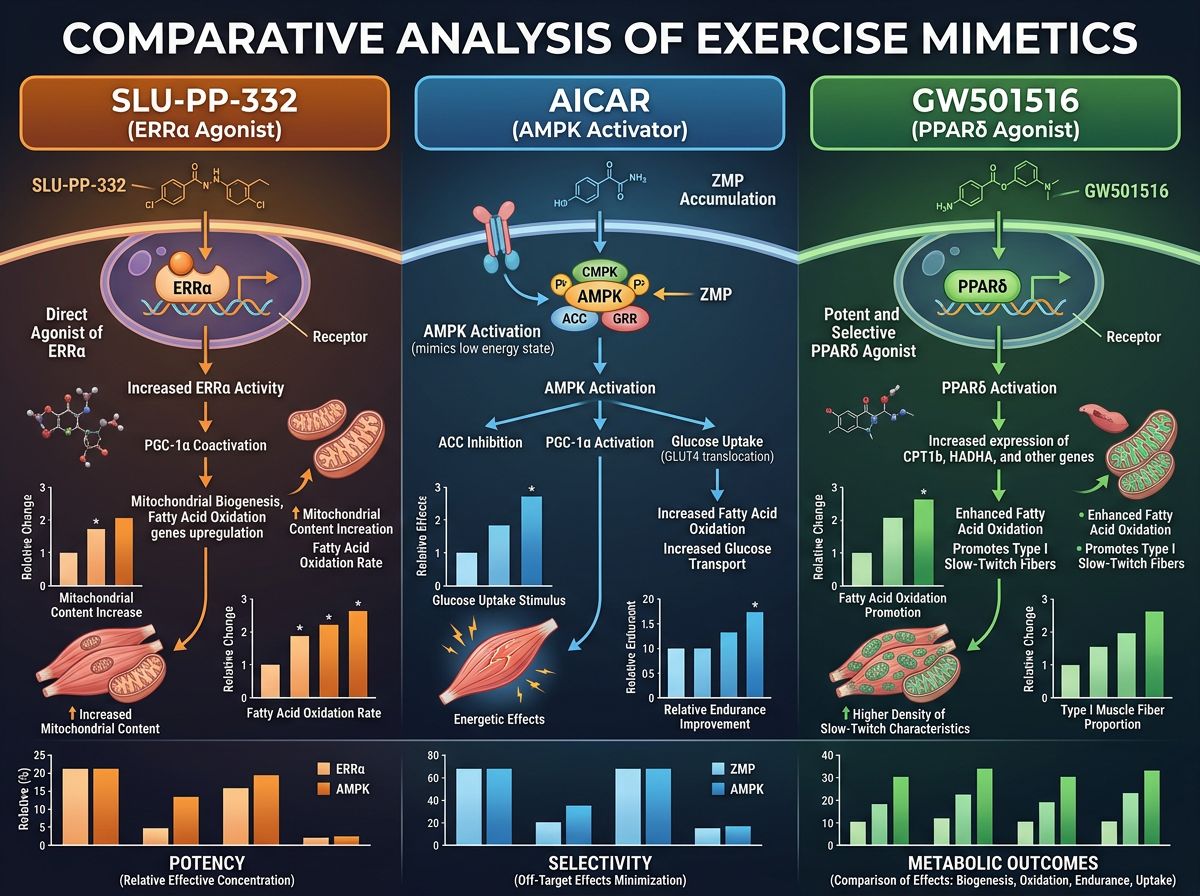
Comparative Analysis: SLU-PP-332 vs. AICAR
To contextualize the advanced mechanism of SLU-PP-332, it is necessary to compare it with the "first generation" exercise mimetic, AICAR (5-Aminoimidazole-4-carboxamide ribonucleotide). Both compounds aim to replicate the metabolic milieu of exercise, but they act at different leverage points within the cell.
AICAR: The AMPK Activator
AICAR works by entering the cell and being converted to ZMP, an analog of AMP (adenosine monophosphate). This surge in intracellular ZMP fools the cell into believing its energy levels are depleted (a high AMP:ATP ratio). In response, the cell activates AMPK, the master energy sensor.
- Mechanism: Phosphorylation of downstream targets to inhibit anabolism (building) and stimulate catabolism (breakdown for energy).
- Research Utility: Proven to increase endurance by roughly 44% in sedentary mice after 4 weeks of treatment.
- Limitations: AMPK is ubiquitously expressed. Systemic activation can have off-target effects, including cardiac hypertrophy and accumulation of lactic acid, as AMPK shuts down gluconeogenesis in the liver.
SLU-PP-332: The Transcriptional Regulator
SLU-PP-332 bypasses the energy sensing step and goes directly to the gene expression machinery.
- Mechanism: Binding to ERRα to drive transcription of mitochondrial genes.
- Research Utility: Shown in initial studies to increase endurance capacity by up to 70% and recover mitochondrial function in heart failure models.
- Safety Advantage: ERRα expression is high in tissues with high energy demands (heart, skeletal muscle, kidney) but lower elsewhere. This tissue specificity may offer a cleaner safety profile compared to systemic AMPK activation.
Table 1: Comparative Pharmacodynamics
| Feature | AICAR | SLU-PP-332 |
|---|---|---|
| Primary Target | AMPK Agonist (Allosteric) | ERRα Agonist (Nuclear Receptor) |
| Pathway Level | Cytosolic Signaling (Enzyme Activation) | Nuclear Transcription (Gene Expression) |
| Muscle Fiber Effect | Increases oxidative enzymes via PGC-1α recruitment (indirect) | Directly transforms fibers to Type IIa/I oxidative phenotype |
| Fat Loss Mechanism | Increased mitochondrial biogenesis; AMPK-mediated lipolysis | Upregulation of beta-oxidation enzymes; Increased basal metabolic rate |
| Renal/Cardiac Implications | Potential for hypertrophy; lactic acidosis risk | Potential protective effects against heart failure and kidney fibrosis |
Metabolic Implications in Research
The introduction of SLU-PP-332 into the research supply chain opens several avenues for investigation beyond simple weight loss.
1. Combatting Sarcopenic Obesity
One of the critical failures of caloric restriction is the concomitant loss of lean muscle mass. Catabolism often targets skeletal muscle proteins for gluconeogenesis during starvation states. Research utilizing SLU-PP-332 suggests a "muscle-sparing" effect. By shifting fuel preference to lipids very aggressively, the body may spare amino acid oxidation. This makes it a compound of high interest for sarcopenic obesity models, where the goal is to reduce fat mass while preserving or enhancing functional muscle tissue.
2. Heart Failure and Myocardial Efficiency
The heart is the most metabolically active organ in the body, relying heavily on fatty acids. In heart failure, the myocardium loses metabolic flexibility and mitochondrial density decreases. PGC-1α and ERRα levels typically drop in failing hearts. Studies indicate that restoring ERRα activity via SLU-PP-332 can preserve cardiac function in pressure-overload models (e.g., aortic banding in mice). This highlights the compound's potential as a therapeutic for bioenergetic failure in cardiac tissue.
3. Synergy with Mitochondrial Peptides
Researchers are increasingly exploring "stacks" or combinations of metabolic agents. While SLU-PP-332 drives the creation of new mitochondria (biogenesis), other peptides address mitochondrial function and quality. For instance, SS-31 (Elamipretide) stabilizes cardiolipin in the inner mitochondrial membrane, optimizing electron transport efficiency. Meanwhile, MOTS-c, a mitochondrial-derived peptide, acts to regulate insulin sensitivity and metabolic homeostasis. A theoretical research protocol combining an ERRα agonist (for quantity) with a cardiolipin stabilizer (for quality) represents the cutting edge of mitochondrial medicine.
Synergistic Potential and Advanced Protocol Design
For laboratory professionals and researchers designing metabolic studies, understanding how SLU-PP-332 interacts with other pathway modulators is essential. The principle of vertical inhibition versus horizontal synergy applies here.
Vertical Synergy: The PGC-1α Axis
Since SLU-PP-332 works downstream of PGC-1α, agents that upstream upregulate PGC-1α could theoretically potentiate its effects. Compounds that activate SIRT1 (Sirtuins), for example, deacetylate and activate PGC-1α. In theory, a SIRT1 activator combined with SLU-PP-332 creates a "push-pull" dynamic where PGC-1α is both activated and has its primary downstream partner (ERRα) hyper-agonized.
Horizontal Synergy: Complementary Pathways
Combining SLU-PP-332 with agents that work via non-mitochondrial pathways can broaden the metabolic impact.
- NNMT Inhibition: 5-amino-1mq inhibits the enzyme NNMT, which prevents the wastage of NAD+ in adipose tissue. Higher NAD+ levels fuel Sirtuins and mitochondrial enzymes. Combining the fat-mobilizing effects of the 5-amino-1mq with the oxidative capacity enhanced by SLU-PP-332 creates a comprehensive fat-burning environment in adipocytes.
- Lipolysis Induction: Peptides like AOD9604 or HGH Fragment 176-191 stimulate the release of fatty acids from adipose tissue. SLU-PP-332 provides the "furnace" to burn these released fatty acids. Without the oxidative capacity provided by ERRα agonism, liberated fatty acids might simply re-esterify (return to storage). This combination addresses both liberation and oxidation of lipids.
Synthesis, Purity, and Quality Control
The complexity of the SLU-PP-332 molecule presents specific challenges in synthesis compared to simple linear peptides. As a small molecule rather than a peptide chain, its synthesis requires rigorous organic chemistry techniques to ensure the correct enantiomer and structure are achieved. Purity is paramount, as impurities in small molecule synthesis can possess vastly different toxicological profiles than the target compound.
When sourcing SLU-PP-332 for research, verifying the identity and purity through High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) is non-negotiable. Investigating the quality control protocols of your supplier ensures that experimental data remains reproducible. At Alpha Carbon Labs, we provide transparency through our COA documents, ensuring that the SLU-PP-332 used in your research meets the stringent requirements of peptide synthesis and small molecule production standards.
Solubility and Handling
Unlike many water-soluble peptides, SLU-PP-332 is hydrophobic. In research settings, it often requires reconstitution in solvents like DMSO or ethanol before being diluted into an aqueous buffer for administration. Proper handling is critical to ensure accurate dosing; precipitation in the syringe or vehicle can lead to significant experimental error.
Future Directions and Clinical Relevance
The current landscape of metabolic research is dominated by GLP-1 receptor agonists. While effective, these drugs primarily function by reducing caloric intake—a method that hits a "floor" when patients cannot restrict calories further without malnutrition or when muscle loss becomes excessive. Exercise mimetics like SLU-PP-332 represent the next frontier: increasing the "calories out" side of the equation at a cellular level.
Future research will likely focus on:
- Oral Bioavailability: Optimizing the structure of SLU-PP-332 for better oral absorption in human models.
- Combination Therapies: Using GLP-1s to control intake while using ERRα agonists to maintain metabolic rate, preventing the metabolic slowdown often seen with rapid weight loss.
- Geroprotection: Investigating if the improvements in mitochondrial function translate to increased healthspan and delay of age-related frailty.
Conclusion
SLU-PP-332 represents a sophisticated tool in the arsenal of metabolic research. By selectively targeting the ERRα nuclear receptor, it offers a distinct mechanism from previous exercise mimetics like AICAR or current clinical standards like Semaglutide. Its ability to mimic the transcriptional signature of endurance exercise—enhancing mitochondrial biogenesis, oxidative capacity, and fatty acid oxidation—without the need for mechanical stress makes it uniquely valuable for studying metabolic disorders, heart failure, and sarcopenia.
For researchers, the data is clear: ERRα agonism is a potent lever for metabolic optimization. As we continue to unravel the complexities of mitochondrial signaling, compounds like SLU-PP-332 will serve as critical probes in understanding how we can decouple the benefits of exercise from the physical act itself, offering hope for conditions where exercise is not a viable therapeutic option.
References
- 1. Billon, C., et al. (2023). A Synthetic ERRα Agonist Alleviates Metabolic Syndrome in Mice. Journal of Pharmacology and Experimental Therapeutics, 385(1), 1-13.
- 2. Narkar, V. A., et al. (2008). AMPK and PPARdelta agonists are exercise mimetics. Cell, 134(3), 405-415.
- 3. Fan, W., & Evans, R. M. (2017). Exercise Mimetics: Impact on Health and Performance. Cell Metabolism, 25(2), 242-247.
- 4. Schreiber, S. N., et al. (2004). The estrogen-related receptor alpha (ERRalpha) functions in PGC-1alpha-induced control of mitochondrial biogenesis. Proceedings of the National Academy of Sciences, 101(17), 6472-6477.
- 5. Huss, J. M., & Kelly, D. P. (2005). Mitochondrial energy metabolism in heart failure: a question of balance. Journal of Clinical Investigation, 115(3), 547-555.
- 6. Sopariwala, D. H., et al. (2021). Estrogen-related receptor alpha is essential for maintaining mitochondrial integrity in skeletal muscle. Skeletal Muscle, 11(1), 12.
- 7. Hardie, D. G. (2011). AMPK: a sensor of cellular energy status. Physiology, 26(1), 18-24.
- 8. Giguère, V. (2008). Transcriptional control of energy homeostasis by the estrogen-related receptors. Endocrine Reviews, 29(6), 677-696.
- 9. Whitfield, J., et al. (2013). Adenosine Monophosphate Protein Kinase (AMPK) as a Potential Target for the Treatment of Type 2 Diabetes. Current Drug Targets, 14(6), 633-646.
- 10. Ljubicic, V., et al. (2010). The Role of PGC-1α in Muscle Fiber Plasticity and Mitochondrial Biogenesis. Journal of Applied Physiology, 109(5), 1530-1533.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.