Modulating the HPG Axis: The Impact of Intracellular NAD+ Flux on Gonadorelin and HCG Efficacy
This article explores the critical role of intracellular NAD+ flux in regulating the sensitivity of the Hypothalamic-Pituitary-Gonadal (HPG) axis, specifically influencing the efficacy of Gonadorelin and HCG in research settings.
Introduction: The Metabolic-Reproductive Crosstalk
In the field of regenerative research and endocrinology, the Hypothalamic-Pituitary-Gonadal (HPG) axis has traditionally been viewed through the lens of pure receptor-ligand kinetics. Researchers have largely focused on how Gonadotropin-Releasing Hormone (GnRH) pulses stimulate the pituitary to release Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH), which in turn drive gonadal steroidogenesis. However, emerging data suggests this view is incomplete without considering the metabolic environment in which these signaling events occur. Specifically, the intracellular flux of Nicotinamide Adenine Dinucleotide (NAD+) appears to act as a "gatekeeper" for HPG axis sensitivity and efficacy.
For researchers utilizing analogues like Gonadorelin and Human Chorionic Gonadotropin (HCG), understanding the bioenergetic status of the target tissues—hypothalamic neurons, pituitary gonadotrophs, and Leydig or granulosa cells—is crucial. The synthesis of sex steroids is one of the most energy-demanding processes in mammalian physiology, heavily reliant on mitochondrial function. As NAD+ levels decline with age or metabolic stress, the efficacy of exogenous peptides may be comprised not by receptor downregulation, but by a failure of the downstream metabolic machinery required to execute the hormonal signal.
This article explores the intricate molecular relationship between intracellular NAD+ levels and the pharmacodynamics of Gonadorelin and HCG. We will examine how NAD+-dependent enzymes, particularly Sirtuins (SIRT1), regulate the transcriptional machinery of steroidogenesis (such as the StAR protein) and how optimizing NAD+ flux may theoretically enhance the responsiveness of the HPG axis in research models.
The Molecular Machinery of Gonadorelin and HCG
Gonadorelin: The Pulsatile Initiator
Gonadorelin is a synthetic decapeptide identical to endogenous GnRH. In research settings, it is primarily used to assess the functional integrity of the pituitary gland or to stimulate the HPG axis in a pulsatile manner as seen in physiological conditions. Upon administration, Gonadorelin binds to the GnRH receptor (GnRHR), a G-protein coupled receptor (GPCR) predominantly located on the surface of gonadotroph cells in the anterior pituitary.
The binding initiates a cascade involving the molecular activation of phospholipase C (PLC), which hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers calcium release from intracellular stores, while DAG activates Protein Kinase C (PKC). This calcium influx is the immediate trigger for the exocytosis of LH and FSH vesicles.
However, the synthesis of new LH and FSH to replenish these stores requires significant energy transcription and translation, processes heavily dependent on ATP availability and, consequently, NAD+ redox states.
HCG: The Luteinizing Surrogate
While Gonadorelin acts centrally, HCG acts distally. Structurally similar to LH, HCG binds to the LH/choriogonadotropin receptor (LHCGR) on Leydig cells in the testes or theca cells in the ovaries. This binding activates adenylate cyclase, increasing intracellular cAMP levels and activating Protein Kinase A (PKA).
The critical downstream effect of PKA activation is the transport of cholesterol into the mitochondria, the rate-limiting step in steroidogenesis. This transport is facilitated by the Steroidogenic Acute Regulatory (StAR) protein. Once inside the mitochondria, cholesterol is converted to pregnenolone by the cytochrome P450scc enzyme (CYP11A1). This step is essentially the "ignition" of testosterone and estrogen synthesis.
Crucially, the cytochrome P450 enzymes involved in steroidogenesis consume electrons. These electrons are supplied via transporting chains that rely on NADPH, which is metabolically linked to the NAD+/NADH pool. Therefore, the efficacy of HCG is mostly limited by the mitochondrial capacity of the Leydig cell to process cholesterol—a capacity defined by mitochondrial health and NAD+ abundance.
NAD+: Beyond a Metabolic Cofactor
Nicotinamide Adenine Dinucleotide (NAD+) exists in two forms: oxidized (NAD+) and reduced (NADH). The ratio of NAD+/NADH is a primary indicator of a cell's metabolic state. However, beyond its role in the Electron Transport Chain (ETC) to generate ATP, NAD+ is a mandatory substrate for several critical enzyme families involved in cell signaling and survival:
- Sirtuins (SIRT1-7): A family of NAD+-dependent deacetylases that regulate gene expression, mitochondrial biogenesis, and inflammation. SIRT1, in particular, is highly relevant to the HPG axis.
- PARPs (Poly (ADP-ribose) polymerases): Enzymes involved in DNA repair that consume NAD+.
- CD38: A cyclic ADP-ribose synthase that modulates calcium signaling and consumes NAD+.
In high-energy tissues like the testes and ovaries, the "drain" on NAD+ is substantial. Research indicates that as organisms age, CD38 activity increases while NAD+ synthesis (via the salvage pathway) declines, leading to a systemic NAD+ deficit. This deficit impairs Sirtuin activity, leading to mitochondrial dysfunction and a reduced ability to respond to hormonal stimuli like HCG or Gonadorelin.
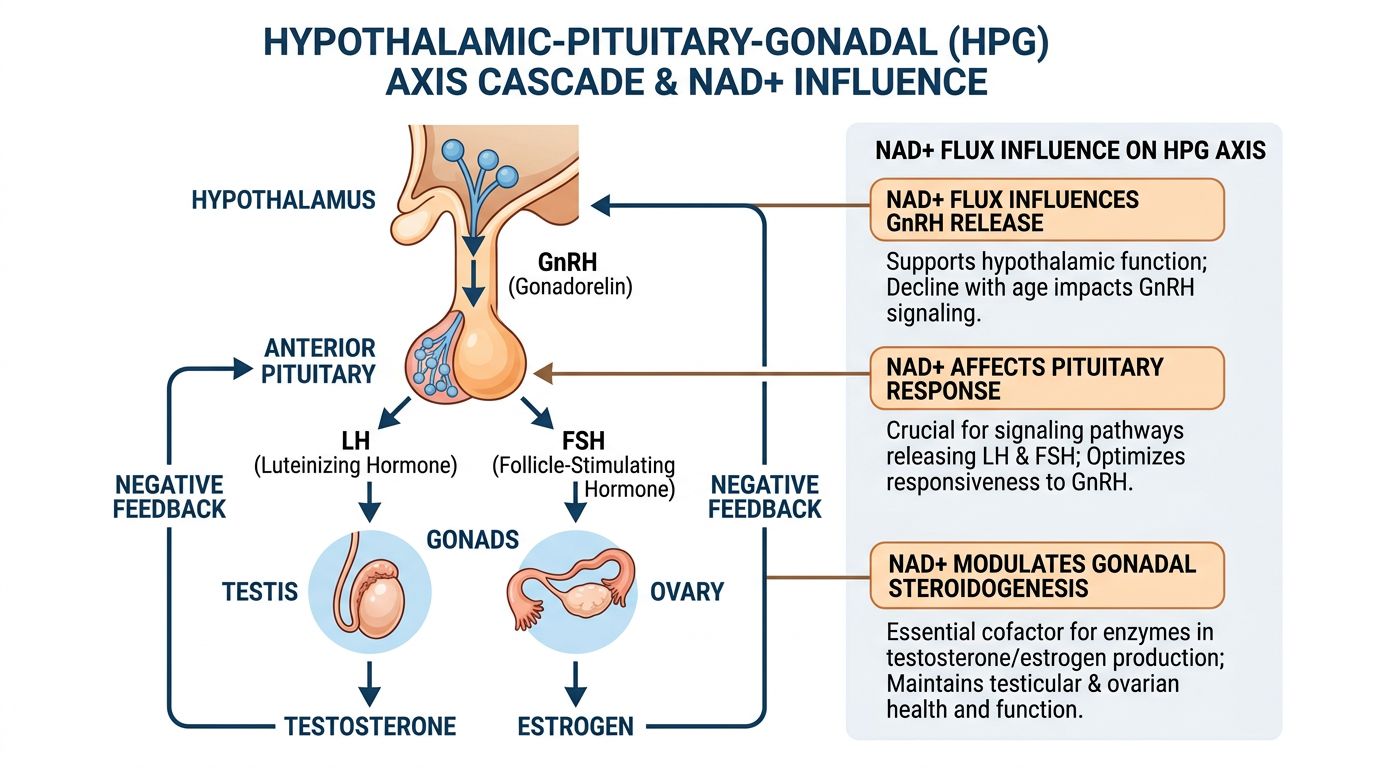
The Convergence: NAD+ Regulation of the HPG Axis
1. Hypothalamic Regulation: SIRT1 and Kisspeptin
The hypothalamus acts as the command center for reproduction. The secretion of GnRH is governed by upstream regulators, most notably Kisspeptin. Recent studies have identified that SIRT1 is highly expressed in hypothalamic neurons and is essential for the pulsatile secretion of GnRH.
SIRT1 acts as a metabolic sensor in these neurons. When NAD+ levels are high (signaling energy availability), SIRT1 is active and deacetylates specific transcription factors (such as FOXO1) that permit the expression of Kiss1 and GnRH. Conversely, in states of low NAD+ (metabolic stress or aging), SIRT1 activity drops, leading to a suppression of central reproductive drive. This is an evolutionary conservation mechanism: reproduction is halted when energy reserves are perceived as low or inefficient.
In research models involving Gonadorelin, the responsiveness of the pituitary is partly dictated by the history of endogenous GnRH exposure. Chronic lack of endogenous GnRH (due to low central NAD+/SIRT1 activity) can lead to receptor downregulation or desensitization, potentially altering the expected response to exogenous administration.
2. Leydig Cell Efficiency and the StAR Protein
Perhaps the most direct link between NAD+ flux and peptide efficacy occurs in the Leydig cells of the testes. As mentioned, the rate-limiting step of testosterone production is the transport of cholesterol into mitochondria via the StAR protein. This process is highly sensitive to oxidative stress and mitochondrial potential.
Research published in Antioxidants & Redox Signaling has shown that SIRT1, activated by adequate NAD+, deacetylates and activates PGC-1α (Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha). PGC-1α is the master regulator of mitochondrial biogenesis. Increased PGC-1α leads to:
- Increased mitochondrial density in Leydig cells.
- Upregulation of StAR protein expression.
- Enhanced expression of steroidogenic enzymes (CYP11A1, CYP17A1).
This creates a clear synergy: HCG provides the signal (via cAMP/PKA) to start steroidogenesis, but NAD+/SIRT1 builds the machinery (Mitochondria/StAR) required to execute it. In a research subject with depleted intracellular NAD+, HCG administration may result in a sub-optimal rise in testosterone because the metabolic machinery cannot keep pace with the hormonal demand. This phenomenon is often termed "steroidogenic arrest."
3. Oxidative Stress and Redox State
Steroidogenesis is inherently inflammatory; the P450 enzymes generate Reactive Oxygen Species (ROS) as a byproduct. If these ROS are not neutralized, they damage the Leydig cells and inhibit the StAR protein, blocking testosterone synthesis. This is where the reducing power of the cell becomes critical.
NAD+ is a precursor for NADPH (via pathways like the Pentose Phosphate Pathway). NADPH is crucial for recycling Glutathione, the cell's master antioxidant. Therefore, high NAD+ flux ensures a robust antioxidant defense system, protecting Leydig cells from the ROS generated during HCG-stimulated steroidogenesis. Without this protection, HCG stimulation can actually become cytotoxic to Leydig cells in the long term.
Comparative Dynamics: Peptide Interactions
To visualize how these components interact, we can compare the mechanisms of action and where NAD+ flux exerts its influence.
| Component | Primary Function in Research | Primary Energy Requirement | NAD+ interaction Point |
|---|---|---|---|
| Gonadorelin | GnRH Receptor Agonist (Pituitary) | Protein synthesis (LH/FSH), Vesicle exocytosis | SIRT1 regulation of pituitary gene expression; Calcium signaling modulation. |
| HCG | LH Receptor Agonist (Gonads) | Cholesterol Transport, Enzymatic conversion | Mitochondrial biogenesis (PGC-1α); ROS scavenging (Glutathione/NADPH). |
| NAD+ | Coenzyme & Signaling Substrate | ATP production, DNA Repair, Deacetylation | Activates SIRT1/PGC-1α pathway; Supports antioxidant defense. |
| KissPeptin-10 | GnRH Secretagogue (Hypothalamus) | Neuronal firing, Peptide synthesis | Hypothalamic metabolic sensing; SIRT1-mediated Kiss1 expression. |
Research Applications and Future Directions
The Aging HPG Axis
One of the most active areas of investigation is the "hypogonadal-metabolic" phenotype seen in aging models. Aging is characterized by a decline in NAD+ and a concurrent decline in HPG axis function (andropause/menopause). Researchers are now investigating whether restoring NAD+ levels via NAD+ administration or precursors can "re-sensitize" the axis to HCG or Gonadorelin therapies.
Preliminary rodent studies suggest that co-administration of NAD+ precursors with HCG significantly improves testosterone output compared to HCG alone in aged subjects. The proposed mechanism is the restoration of StAR protein expression and mitochondrial function in Leydig cells, effectively removing the metabolic bottleneck.
Metabolic Disorders and Fertility
Metabolic syndrome causes a suppression of the HPG axis, often leading to hypogonadism. High caloric intake coupled with sedentary behavior leads to a "pseudohypoxic" state where NADH is high but NAD+ is low (low redox ratio). This inhibits SIRT1, leading to reduced mitochondrial efficiency.
Research using peptides like Tesamorelin (which reduces visceral fat) alongside HPG modulators is common, but direct NAD+ modulation represents a more fundamental approach. By correcting the cellular redox state, researchers aim to restore the Leydig cell's ability to respond to LH/HCG signals efficiently.
Synergistic Protocols in Research
Current experimental protocols are moving away from single-vector interventions. Instead of simply pressing the "gas pedal" with HCG, researchers are looking at maintaining the "engine." A theoretical research protocol might involve:
- Basal Support: NAD+ or 5-amino-1mq (to inhibit NNMT and boost endogenous NAD+) to ensure mitochondrial density and enzymatic capacity.
- Axis Stimulation: Gonadorelin to maintain pituitary pulsatility and preventing atrophy of gonadotrophs.
- Direct Agonism: HCG for direct Leydig cell stimulation, timed to coincide with peak metabolic availability.
- Antioxidant Support: Glutathione or SS-31 to protect mitochondria from the increased ROS generation associated with upregulated steroidogenesis.
Sourcing High-Purity Peptides for Research
The complexity of these metabolic pathways underscores the necessity of chemical purity in research reagents. When studying the subtle interplay between mitochondrial flux and hormonal signaling, impurities in peptide synthesis can introduce confounding variables. Truncated peptide sequences or counter-ion contaminants can trigger inflammatory responses (TLR activation) that deplete cellular NAD+ pools via CD38 activation, masking the true experimental results.
At Alpha Carbon Labs, we emphasize stringent Quality Control. Our peptides, including Gonadorelin, HCG, and NAD+, undergo rigorous HPLC and Mass Spectrometry analysis to ensure they meet the specific needs of advanced endocrine research. By eliminating the variable of purity, researchers can focus entirely on the physiological mechanics of the HPG axis.
For more information on the synthesis standards required for sensitive metabolic research, visit our peptide synthesis methodology page or review our Certificates of Analysis for batch-specific data.
Conclusion
The HPG axis does not operate in a vacuum. It is intrinsically tied to the metabolic fate of the organism. The efficacy of Gonadorelin and HCG is governed not just by receptor availability, but by the intracellular flux of NAD+ and the health of the mitochondrial networks in target tissues. By viewing endocrine research through a metabolic lens—recognizing NAD+ as a critical modulator of steroidogenesis and signaling—researchers can design more robust experiments and potentially unlock new therapeutic paradigms for hypogonadism and age-related hormonal decline.
References
- 1. Imai, S., & Guarente, L. (2014). NAD+ and sirtuins in aging and disease. Trends in cell biology, 24(8), 464-471.
- 2. Kolasa, A., et al. (2013). The influence of age and energy restriction on the expression of Sirt1 and iNOS in the rat testis. Reproductive Biology, 13(1), 38-47.
- 3. Tatone, C., & Amicarelli, F. (2013). The aging ovary-the poor granulosa cells. Fertility and sterility, 99(1), 12-17.
- 4. Wang, X., et al. (2020). SIRT1 regulates leydig cell function via crosstalk with the hypothalamus-pituitary-gonadal axis. Frontiers in Endocrinology, 11, 583.
- 5. Navarro, G., & Kaiser, U. B. (2013). Metabolic influences on neuroendocrine regulation of reproduction. Current opinion in endocrinology, diabetes, and obesity, 20(4), 335.
- 6. Yoon, M. J., et al. (2014). SIRT1 mediates melatonin-induced protection against thapsigargin-induced apoptosis in mouse testes. Journal of pineal research, 56(2), 195-206.
- 7. Wu, S., et al. (2019). NAD+ regulates steroidogenesis in Leydig cells via SIRT1‐mediated regulation of the StAR protein. Journal of Cellular Physiology, 234(5), 7245-7256.
- 8. Cohen, D. J., et al. (2009). NAD+ levels regulate sperm mitochondrial function. Molecular Human Reproduction, 15(9), 563-573.
- 9. Vazquez, M. J., et al. (2015). Nutritional regulation of the hypothalamic-pituitary-gonadal axis. Physiological Reviews, 95(3), 953-1002.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.