Mitochondrial Bioenergetics: Analyzing the Synergistic Potential of SS-31 and MOTS-c
Explore the synergistic mechanisms of SS-31 and MOTS-c in optimizing mitochondrial bioenergetics, focusing on cardiolipin protection, AMPK signaling, and ATP production.
Introduction to Mitochondrial Bioenergetics
In the hierarchy of cellular physiology, mitochondria occupy the pivotal position. Often described as the "powerhouses of the cell," their role extends far beyond the simple production of Adenosine Triphosphate (ATP). They are the primary regulators of metabolic signaling, calcium homeostasis, and apoptosis. However, the byproduct of mitochondrial respiration—Reactive Oxygen Species (ROS)—can lead to a state of oxidative stress that compromises the integrity of mitochondrial membranes and proteins. When mitochondrial bioenergetics fail, the result is a cascade of cellular dysfunction often associated with aging, metabolic disorders, and neurodegeneration.
Recent breakthroughs in peptide research have spotlighted two specific molecules that target these metabolic pathways: SS-31 (Elamipretide) and MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c). While both are mitochondrial-focused, they operate through distinct mechanisms. SS-31 acts primarily as a structural stabilizer of the inner mitochondrial membrane, whereas MOTS-c functions as a nuclear-signaling peptide that regulates systemic metabolism. This article provides a comprehensive analysis of their synergistic potential in modern research environments.
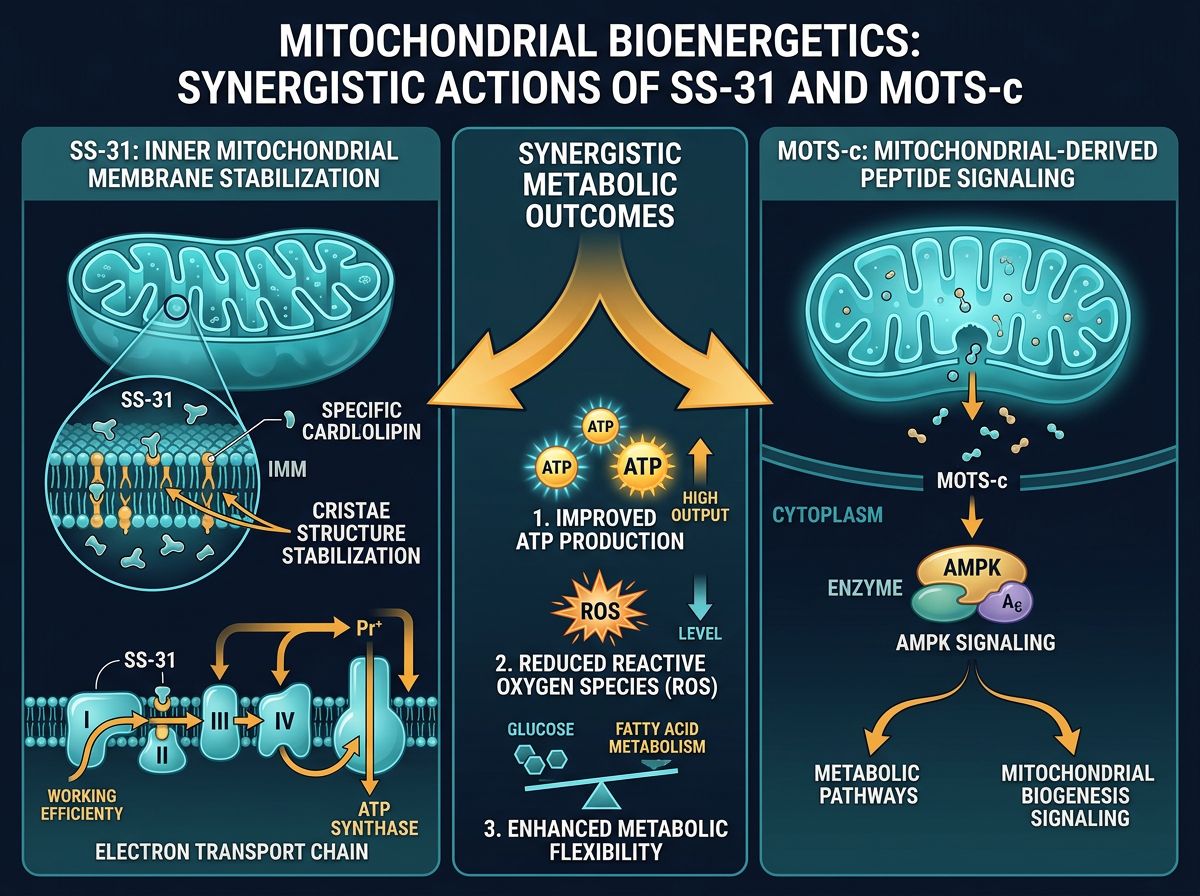
The Physiology of SS-31: Cardiolipin Protection and Membrane Integrity
SS-31 is a member of the Szeto-Schiller (SS) peptide family, characterized by an alternating aromatic-cationic motif. Specifically, it is a synthetic tetrapeptide (D-Arg-2',6'-Dmt-Lys-Phe-NH2) that possesses the unique ability to cross the cell membrane and localize within the inner mitochondrial membrane (IMM).
Mechanism of Action: The Cardiolipin Interaction
The efficacy of SS-31 is rooted in its high affinity for cardiolipin, a unique phospholipid found exclusively in the inner mitochondrial membrane. Cardiolipin is essential for the stabilization of the cristae structure and the organization of the mitochondrial respiratory chain into "supercomplexes." These supercomplexes allow for efficient electron transfer between complexes I, III, and IV.
Under conditions of high oxidative stress, cardiolipin undergoes peroxidation. This causes the supercomplexes to disassemble, leading to electron leakage and a subsequent spike in ROS production. SS-31 binds to cardiolipin, protecting it from peroxidation and maintaining the optimal curvature of the inner membrane. This structural stabilization results in:
- Improved electron transport chain (ETC) efficiency.
- Increased ATP sourcing without increasing oxygen consumption.
- Reduced production of superoxide radicals.
- Prevention of Cytochrome C leakage, thereby inhibiting pro-apoptotic signaling.
Research Findings in Bioenergetics
In various cellular models, SS-31 has demonstrated the ability to restore mitochondrial coupling even after significant damage. Researchers utilizing COA-verified SS-31 have observed that it can reverse bioenergetic deficits in ischemia-reperfusion injury models. By preserving the cristae architecture, SS-31 ensures that the electrochemical gradient remains intact, ensuring cellular survival under metabolic stress.
MOTS-c: The Metabolic Command Center
While SS-31 focuses on the structural hardware of the mitochondria, MOTS-c represents the mitochondrial "software." MOTS-c is a 16-amino acid mitochondrial-derived peptide (MDP) encoded within the mitochondrial DNA (mtDNA) itself. It acts as a signaling molecule that communicates the metabolic status of the mitochondria to the nucleus.
The AMPK Pathway and Glucose Metabolism
The primary mechanism of MOTS-c involves the activation of the AMP-activated protein kinase (AMPK) pathway. In response to metabolic stress, MOTS-c translocates to the nucleus where it regulates genes involved in glucose metabolism and fatty acid oxidation. This makes it a crucial research subject in the study of insulin sensitivity and exercise-mimetic effects.
Unlike traditional hormones, MOTS-c acts locally and systemically to:
- Increase glucose uptake in skeletal muscle via GLUT4 translocation.
- Enhance fatty acid beta-oxidation, reducing lipid accumulation.
- Inhibit the Folate Cycle and De Novo purine synthesis, leading to increased AICAR levels (an AMPK activator).
- Promote mitochondrial biogenesis through PGC-1α upregulation.
For researchers interested in metabolic flexibility, MOTS-c is often studied alongside agents like AICAR or 5-amino-1mq to evaluate the potentiation of lipid metabolism and energy expenditure.
Synergistic Potential: Structural Support Meets Metabolic Signaling
The combination of SS-31 and MOTS-c represents a dual-action approach to mitochondrial health. From a research perspective, using these two peptides together allows for the simultaneous stabilization of mitochondrial structure and the optimization of metabolic output.
Comparing SS-31 and MOTS-c
| Feature | SS-31 (Elamipretide) | MOTS-c |
|---|---|---|
| Origin | Synthetic Tetrapeptide | Mitochondrial-derived (Natural) |
| Primary Target | Cardiolipin (Inner Membrane) | AMPK Pathway / Nucleus |
| Main Benefit | Reduced ROS & Structural Integrity | Improved Glucose & Lipid Metabolism |
| Method of Action | Electron transport optimization | Genomic signaling & protein synthesis |
| Secondary Effects | Anti-apoptotic (inhibits Cyt C) | Exercise-mimetic properties |
A Hypothetical Research Model for Synergy
In a research setting, the application of SS-31 provides the necessary "foundation." By ensuring that cardiolipin is protected and the ETC is functioning efficiently, the mitochondria are less likely to produce excessive ROS when metabolic demands are increased. Following this, the introduction of MOTS-c can then push the "accelerator," signaling the cell to increase its metabolic throughput. In isolation, increasing metabolic demand via MOTS-c might lead to oxidative stress if the mitochondria are already damaged. However, SS-31 mitigates this risk by fortifying the mitochondrial membrane.
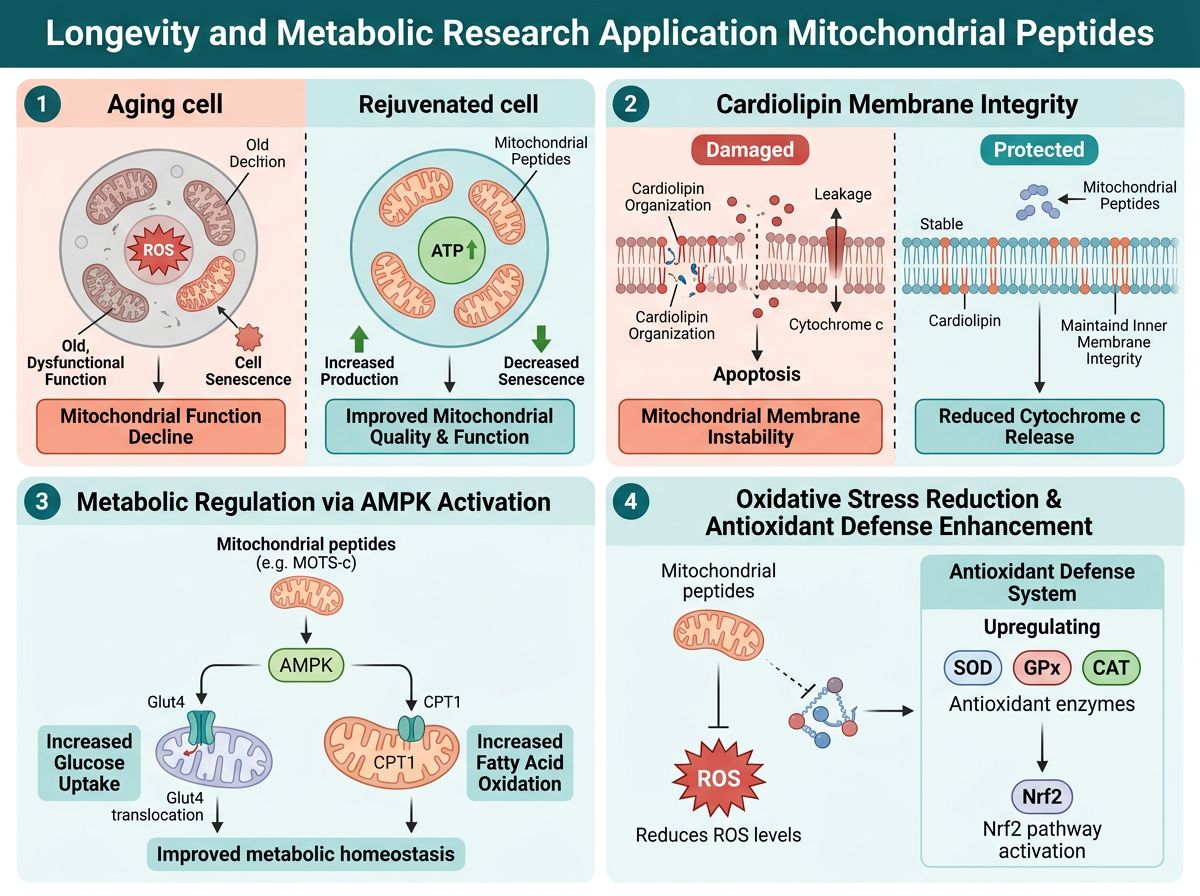
Applications in Longevity and Metabolic Research
Mitochondrial decline is a hallmark of biological aging. As mitochondria age, they become fragmented and less efficient. This leads to a state known as mitinflammation. The synergistic use of mitochondrial-targeted peptides is currently being explored to combat this decline.
Mitochondrial Biogenesis and Repair
While MOTS-c triggers the birth of new mitochondria (biogenesis), SS-31 ensures that those mitochondria remain functional. This interplay is essential for tissues with high energy demands, such as cardiac and skeletal muscle. Researchers are also examining how these peptides interact with NAD+ precursors to further boost cellular energy levels.
Neuroprotection
The brain is the most metabolically active organ, consuming roughly 20% of the body's oxygen. Mitochondrial dysfunction in neurons is a precursor to synaptic loss. Studies involving Cerebrolysin or Dihexa often look at the cognitive benefits of peptide therapy, but the underlying bioenergetic support provided by SS-31 and MOTS-c offers a fundamental layer of protection against neuro-oxidative stress.
Quality Standards in Peptide Research
At Alpha Carbon Labs, we emphasize the importance of high-purity compounds for reproducible research results. The complex structure of MOTS-c and the specific ion-pairing required for SS-31 stability necessitate rigorous quality control. Every batch must undergo HPLC and Mass Spectrometry to Ensure 99%+ purity. For detailed synthesis information, researchers are encouraged to visit our peptide synthesis page.
The Future of Mitochondrial Therapeutics
The transition from looking at mitochondria as simple batteries to viewing them as dynamic signaling hubs has revolutionized peptide therapy. The development of dual-targeting strategies—addressing both the structural (SS-31) and the regulatory (MOTS-c) aspects of mitochondrial function—holds significant promise for future research into sarcopenia, metabolic syndrome, and age-related organ failure.
For researchers focusing on systemic repair, integrating these mitochondrial peptides with tissue-repair agents such as the BPC-157 + TB-500 Blend may provide a comprehensive model for accelerating recovery and enhancing cellular resilience. By stabilizing the bioenergetic core, the efficacy of downstream regenerative peptides is often significantly amplified.
Conclusion
Mitochondrial bioenergetics is a complex field that requires a multi-faceted approach. SS-31 and MOTS-c offer a powerful combination: one preserves the delicate architecture of the mitochondrial membrane, while the other orchestrates a systemic metabolic response. Together, they represent a new frontier in the quest to preserve and enhance cellular energy production in the face of stress and aging.
References
- 1. Szeto HH. First-in-class cardiolipin-protective compound as a therapeutic strategy to restore mitochondrial bioenergetics. Br J Pharmacol. 2014;171(8):2029-50.
- 2. Lee C, et al. The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Obesity and Insulin Resistance. Cell Metabolism. 2015;21(3):443-454.
- 3. Zhang L, et al. SS-31 ameliorates mitochondrial dysfunction and oxidative stress-induced apoptosis in insulin-resistant hepatocytes. Mol Med Rep. 2017;15(4):1753-1759.
- 4. Kim KH, et al. The mitochondrial peptide MOTS-c: A new frontier in metabolic and aging research. Free Radic Biol Med. 2021;177:125-135.
- 5. Brown DA, et al. Mitochondrial-targeted peptides for the treatment of heart failure. JACC Basic Transl Sci. 2017;2(3):293-308.
- 6. Ying W. NAD+/NADH and NADPH in organ failure, aging and insulin resistance. Front Biosci. 2007;12:1861-88.
- 7. Reynolds JC, et al. MOTS-c is an exercise-induced mitochondrial-derived peptide that regulates muscle metabolism and physical performance. Nat Commun. 2021;12(1):437.
- 8. Chavez JD, et al. Mitochondrial Interactome Revealed by Proximity Labeling and Cross-linking Mass Spectrometry. Mol Cell Proteomics. 2018;17(4):726-740.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.