Methodological Rigor: Analytical Techniques for Reconstituting and Assaying Tirzepatide
A technical guide for laboratories on the analytical techniques required for the reconstitution, molarity calculation, and stability maintenance of Tirzepatide in research settings.
Introduction: The Complexity of Dual-Agonist Peptides
In the evolving landscape of incretin mimetics, Tirzepatide represents a watershed moment in peptide engineering. As a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, its molecular architecture necessitates a high degree of precision during laboratory reconstitution and assay preparation. For researchers, understanding the biophysical properties of this 39-amino acid linear peptide—and its C18 fatty acid diacid moiety—is critical for maintaining potency and ensuring reproducible experimental results.
At Alpha Carbon Labs, we emphasize that scientific discovery is predicated on the quality of raw materials and the rigor of the methodology applied to them. This guide explores the analytical techniques required for the successful handling of lyophilized Tirzepatide, from initial solvation to the maintenance of chemical stability in complex in vitro environments.
Molecular Profile and Structural Considerations
Sequence and Modification
Tirzepatide is a synthetic peptide based on the GIP sequence, modified with two aminoisobutyric acid (Aib) residues at positions 2 and 13 to enhance stability against dipeptidyl peptidase-4 (DPP-4) degradation. Most importantly, it features a C20 fatty diacid moiety attached via a linker to a lysine residue at position 20. This acylation is the primary driver of its extended half-life, allowing for high-affinity albumin binding.
Physical State: Lyophilization and Purity
Our research-grade Tirzepatide is provided as a lyophilized (freeze-dried) powder. This state is reached through a controlled sublimation process that preserves the secondary structure of the peptide. To confirm the identity and purity of the sample before beginning an assay, researchers should consult the Certificate of Analysis (CoA). At Alpha Carbon Labs, we utilize High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) to guarantee >99% purity, ensuring that extraneous peptide fragments do not interfere with GIP/GLP-1 receptor signaling assays.
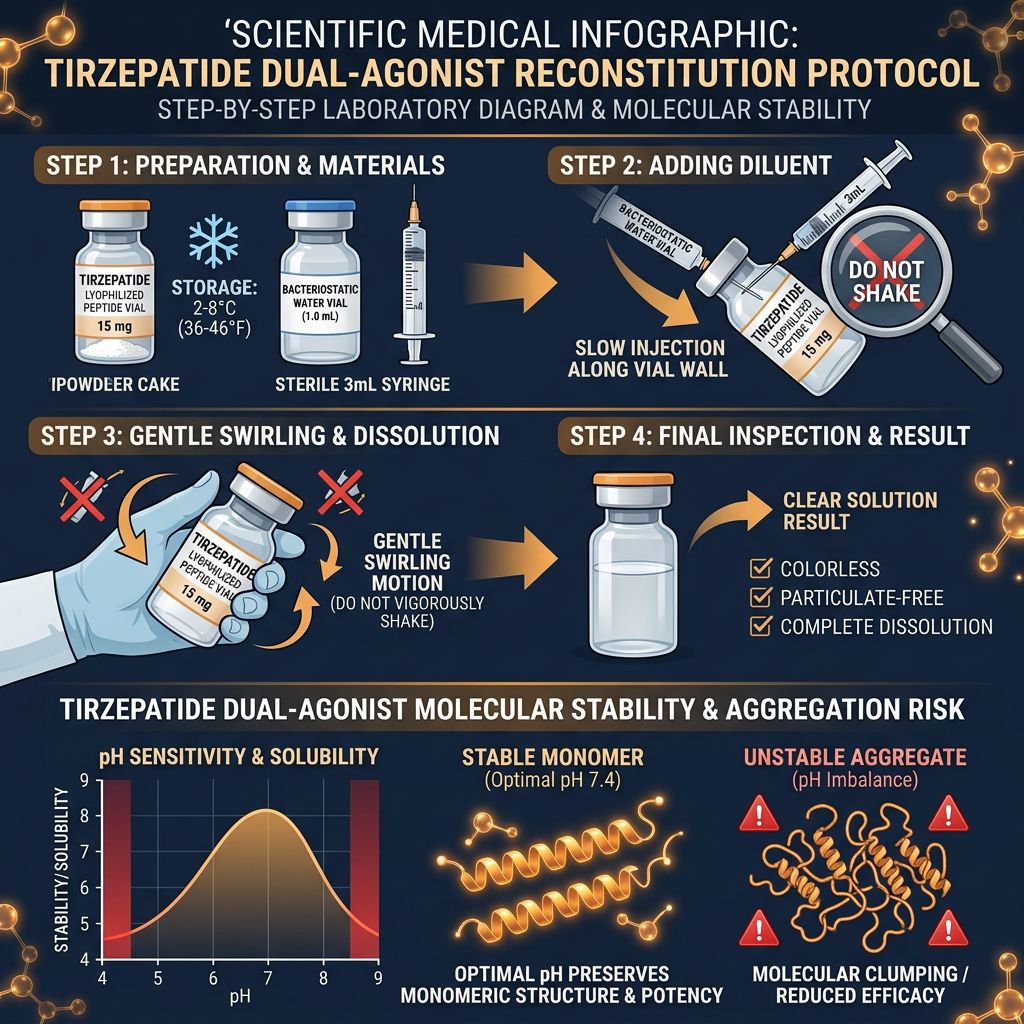
Reconstitution Strategy: Achieving Proper Solvation
The transition of a peptide from a lyophilized state to a bioavailable solution is a critical juncture. Improper reconstitution can lead to aggregation, precipitation, or oxidation of sensitive residues. Unlike simpler peptides like BPC-157, the acylated nature of Tirzepatide requires specific environmental considerations.
Solvent Selection
For most in vitro research applications, Bacteriostatic Water or sterile Phosphate-Buffered Saline (PBS) are the standard diluents. However, researchers must consider the target pH. Tirzepatide is most stable at a slightly acidic to neutral pH (approx. 7.0–7.4). Extreme pH shifts can lead to the deamidation of asparagine or glutamine residues.
- Bacteriostatic Water (0.9% Benzyl Alcohol): Ideal for multi-use vials intended for short-term refrigerated storage. The preservative inhibits microbial growth but may interact with certain high-sensitivity cell-based assays.
- Sterile Normal Saline (0.9% NaCl): Often preferred for physiological modeling, as it maintains osmotic balance.
- Acetic Acid (0.1%): In rare cases where solubility issues arise due to peptide-peptide interactions, a very dilute acid solution may be used, though this is rarely necessary for Tirzepatide compared to more hydrophobic peptides like AICAR.
The "Slow Handoff" Technique
To avoid the formation of air bubbles and mechanical shear—which can denature the peptide’s secondary structure—the solvent should be introduced by allowing it to trickle down the side of the glass vial. Physical agitation should be limited to gentle swirling; never shake the vial. Shaking increases the surface area of the liquid-air interface, which can promote oxidation and the irreversible aggregation of the acylated peptide chains.
Molarity Calculations and Quantitative Precision
Precision in research requires a transition from mass-based dosing (mg) to molar-based dosing (µM or nM), especially when comparing Tirzepatide to other agonists like Semaglutide or Retatrutide. Experiments involving competitive binding assays or EC50 determinations rely entirely on the accuracy of these calculations.
Formula for Concentration
The standard formula for determining the molarity (M) of the reconstituted solution is:
M = (m / MW) / V
Where:
- m: Mass of the peptide (grams)
- MW: Molecular Weight of Tirzepatide (approx. 4813.5 g/mol)
- V: Volume of the solvent (Liters)
Table 1: Dilution Guide for 5mg Tirzepatide
| Volume of Diluent (mL) | Resulting Concentration (mg/mL) | Approx. Molarity (mM) |
|---|---|---|
| 1.0 | 5.0 mg/mL | 1.04 mM |
| 2.0 | 2.5 mg/mL | 0.52 mM |
| 5.0 | 1.0 mg/mL | 0.21 mM |
Stability and Degradation Pathways
Research peptides are inherently metastable. The longevity of Tirzepatide in a liquid state is influenced by temperature, light exposure, and the Presence of secondary additives. Understanding these variables is essential for maintaining the integrity of longitudinal studies.
Thermodynamic Stability
Once reconstituted, Tirzepatide should be stored at 2°C to 8°C. For long-term storage exceeding one week, researchers often aliquot the solution into single-use tubes and freeze them at -20°C or -80°C. Repeated freeze-thaw cycles must be avoided, as the resulting ice crystal formation can cause mechanical cleavage of the peptide backbone.
Chemical Degradation Vulnerabilities
- Oxidation: Methionine residues can oxidize to methionine sulfoxide, potentially altering receptor affinity. Vials should be kept away from direct UV light.
- Deamidation: As mentioned, pH control is vital to prevent the conversion of carboxamide groups to carboxylic acids.
- Adsorption: Like many acylated peptides, Tirzepatide can adhere to the plastic walls of microcentrifuge tubes or pipettes. Utilizing low-protein binding (LPB) plastics is a recommended laboratory practice to prevent significant loss of titer during serial dilutions.
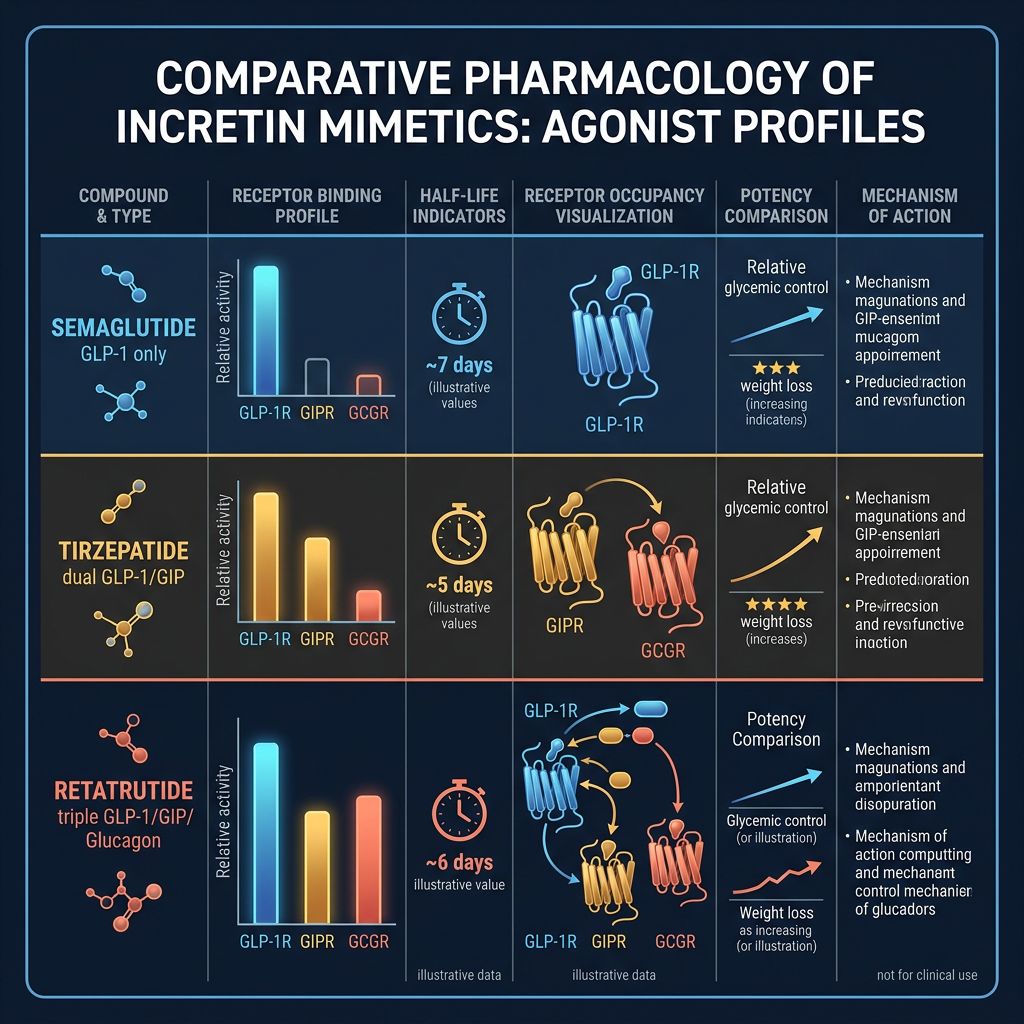
Comparative Analysis: Tirzepatide vs. Other Incretins
When designing an assay, it is often useful to contrast Tirzepatide with other research models to map receptor selectivity. Tirzepatide is unique because it acts as a "biased" agonist at the GLP-1 receptor, favoring CAMP signaling over β-arrestin recruitment, which contributes to its distinct pharmacological profile compared to Cagrilintide or mono-agonists.
Comparison of Research Agonists
| Peptide | Receptor Target(s) | Primary Research Use | MW (approx) |
|---|---|---|---|
| Tirzepatide | GLP-1 / GIP | Metabolic Synergy / Dual Signaling | 4,814 Da |
| Semaglutide | GLP-1 | Glucose Homeostasis Models | 4,113 Da |
| Retatrutide | GLP-1 / GIP / GCGR | Triple Agonism / Thermogenesis | 5,429 Da |
| Mazdutide | GLP-1 / GCGR | Incretin-Glucagon Balance | 4,450 Da |
In Vitro Assay Applications
Tirzepatide is frequently utilized in cell culture models to investigate pancreatic beta-cell function, adipocyte differentiation, and hypothalamic signaling. In these settings, ensuring the peptide remains monomeric is paramount.
Cell Culture Preparation
When introducing Tirzepatide to media (e.g., DMEM or RPMI), the presence of Fetal Bovine Serum (FBS) must be accounted for. Since Tirzepatide is designed to bind to albumin, the effective concentration of free peptide in serum-containing media may be lower than intended. Researchers studying receptor-ligand kinetics often utilize serum-free media for short-term incubation to observe direct effects without albumin-mediated sequestering.
Signal Transduction Assays
Commonly, Tirzepatide research involves monitoring cAMP production as a proxy for receptor activation. Because Tirzepatide has a 5-fold higher affinity for the GIP receptor than the GLP-1 receptor, researchers can use it to map the interplay between these two pathways in insulin secretion. For high-throughput screening, ensuring consistent peptide synthesis quality (available through Alpha Carbon Labs) is the difference between meaningful data and statistical noise.
Advanced Handling: Multi-Peptide Blends
In various metabolic research frameworks, Tirzepatide may be combined with other peptides to examine additive effects. For instance, combining incretins with mitochondrial-targeted peptides like MOTS-c or SS-31 is a growing area of study regarding cellular energy expenditure. When assaying blends, it is vital to ensure that the reconstitution solvent is compatible with all components. For example, while Tirzepatide is stable in saline, more complex peptides like IGF-1 LR3 may require more specific acidic environments to prevent precipitation.
Quality Control and Analytical Verification
No research protocol is complete without verification. At Alpha Carbon Labs, our quality control process involves several stages of analysis to ensure that every mg of Tirzepatide delivered meets the strictly defined specifications required for high-impact bioscience.
- HPLC (High-Performance Liquid Chromatography): This technique separates the peptide from any potential synthesis byproducts. A single, sharp peak indicates high purity and correct folding.
- MS (Mass Spectrometry): This confirms the exact molecular mass. For Tirzepatide, this confirms the presence of the fatty acid chain and the correct amino acid sequence.
- Endotoxin Testing: Particularly important for cell culture, ensuring that the peptide is free of bacterial contaminants that could induce inflammatory responses in the host cells.
Conclusion
Tirzepatide remains one of the most sophisticated tools in the modern researcher's arsenal for exploring metabolic health and incretin biology. However, its effectiveness as a research tool is directly proportional to the care taken during its analytical preparation. By adhering to the reconstitution and storage standards outlined—prioritizing pH balance, avoiding mechanical stress, and utilizing high-purity sources—scientific laboratories can maximize the reliability of their data.
For researchers seeking the highest grade of metabolic agonists and supportive peptides like 5-amino-1MQ or CJC-1295, Alpha Carbon Labs provides the transparency and analytical depth required to push the boundaries of medical science.
References
- 1. Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
- 2. Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
- 3. Finan B, et al. A novel chimerical synergy between glucagon-like peptide-1 and glucagon receptor agonism. Nat Med. 2015;21(1):27-36.
- 4. Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes. Mol Metab. 2018;18:3-14.
- 5. D'Alessio D, et al. Continuous subcutaneous insulin infusion and peptide stability in external pumps. Diabetes Care. 2009.
- 6. Manning MC, et al. Stability of Protein Pharmaceuticals: An Update. Pharm Res. 2010;27(4):544-575.
- 7. Nauck MA, et al. Incretin-based therapies: Genus and species. Diabetes Care. 2011;34(Suppl 2):S236–S243.
- 8. Mroz PA, et al. Optimized GIP receptor-selective agonists. J Med Chem. 2016;12;59(21):9889-9908.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.