Kinetic Stability: Methodological Best Practices for Reconstituting and Storing Research-Grade NAD+
A comprehensive methodological guide for researchers on the kinetic stability, reconstitution, and storage of research-grade NAD+. This article details the impact of pH, temperature, and ionic strength on NAD+ integrity to ensure experimental validity.
Introduction
In the evolving landscape of metabolic research, few molecules claim as central a role as Nicotinamide Adenine Dinucleotide (NAD+). Serving as an obligatory coenzyme for hydride transfer enzymes and a substrate for sirtuins, PARPs, and CD38, NAD+ governs critical aspects of bioenergetics, genomic integrity, and circadian rhythmicity. However, for the laboratory scientist, NAD+ presents a paradoxical challenge: while it is biochemically ubiquitous, it is distinctively chemically fragile relative to other dinucleotides.
The reliability of experimental data—whether derived from high-throughput screening, enzymatic cycling assays, or in vivo administration—is strictly dependent on the kinetic stability of the precursor material. The degradation of NAD+ does not merely result in a loss of potency; the accumulation of breakdown products, such as ADP-ribose, cyclic ADP-ribose, and free nicotinamide, can act as potent inhibitors of the very pathways under investigation. Therefore, maintaining the integrity of NAD+ is not simply a matter of storage; it is a matter of experimental validity.
At Alpha Carbon Labs, we emphasize that high-purity synthesis is only the first step in the lifecycle of a reagent. Understanding the variables that accelerate hydrolysis and oxidation—specifically pH, temperature, and ionic strength—is essential for researchers aiming to replicate valid physiological conditions. This guide elucidates the methodological best practices for reconstituting, aliquoting, and storing research-grade NAD+, grounded in the principles of solution-state kinetics.
The Chemistry of Instability: Why NAD+ Degrades
To implement effective storage protocols, one must first appreciate the structural vulnerabilities of the molecule. NAD+ consists of two nucleotides joined through their phosphate groups by a phosphoanhydride bond. One nucleotide contains an adenine base and the other a nicotinamide base. The critical structural feature governing its instability is the β-N-glycosidic bond linking the nicotinamide moiety to the ribose ring.
Hydrolysis and the Nicotinamide-Glycosyl Bond
The primary degradation pathway for NAD+ in aqueous solution is the hydrolysis of the nicotinamide-glycosyl bond. This reaction yields nicotinamide and Adenosine Diphosphate Ribose (ADPR). This bond is high-energy and particularly susceptible to nucleophilic attack, especially by hydroxide ions (OH-).
Researchers must note that the stability profile of NAD+ is the inverse of its reduced counterpart, NADH. While NADH is extremely labile in acidic conditions due to the protonation of the dihydronicotinamide ring, leading to acid-catalyzed hydration, NAD+ is remarkably unstable in alkaline (basic) environments. At biological pH (~7.4), NAD+ is moderately stable, but as pH rises above 8.0, the rate of hydrolysis increases exponentially. This distinction is critical when selecting buffers for reconstitution.
Cyclization and Anomerization
Beyond simple hydrolysis, NAD+ is prone to cyclization reactions involving the purine ring and the ribose. Under strong alkaline conditions, NAD+ can form breakdown products that fluoresce, interfering with spectrophotometric assays dependent on A340 absorbance. Furthermore, reversible anomerization of the glycosidic bond from the biologically active β-form to the inactive α-form can occur, rendering the molecule useless for enzymatic recognition without necessarily changing its molecular weight, making this degradation invisible to standard mass spectrometry unless chiral chromatography is employed.
For more details on the rigorous testing required to detect these subtleties, refer to our Quality Control methodologies and COA Documents.
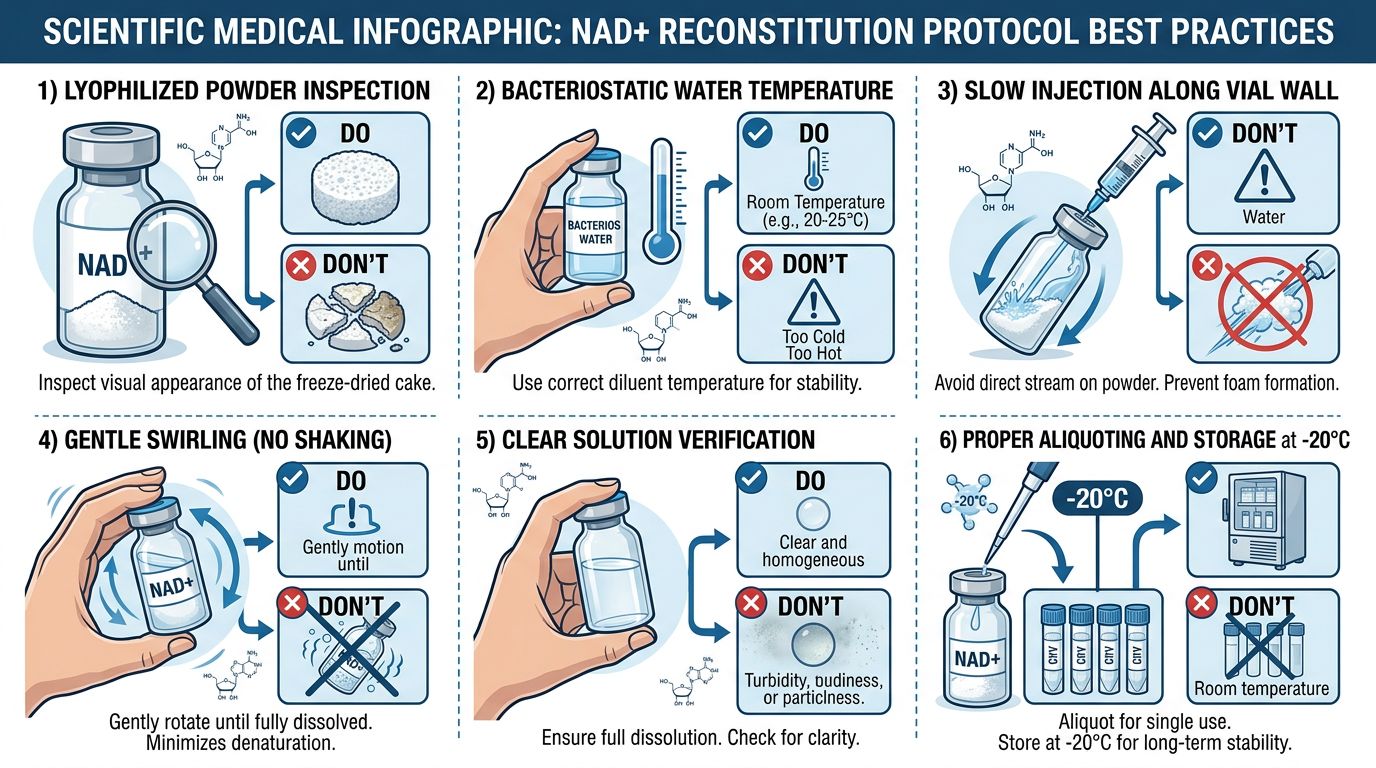
Reconstitution Protocols: The Critical First Step
The moment lyophilized NAD+ interacts with a solvent, a "kinetic clock" begins ticking. The choice of solvent and the physical technique of reconstitution define the longevity of the solution.
Solvent Selection: The pH Imperative
As established, the dominant variable in NAD+ stability is pH. Consequently, the common laboratory practice of reconstituting reagents in Phosphate Buffered Saline (PBS) pH 7.4 is acceptable for short-term use but suboptimal for long-term storage or high-concentration stock solutions.
| Solvent/Buffer | Approximate pH | Stability Assessment | Recommended Use |
|---|---|---|---|
| Sterile Water for Injection (SWFI) | 5.5 - 7.0 | Excellent | Ideal for creating high-concentration stock solutions for freezing. Slightly acidic pH preserves the glycosidic bond. |
| Acidified Saline | 4.0 - 5.5 | Superior | Maximum stability for long-term banking. |
| PBS | 7.4 | Moderate | Acceptable for immediate in vivo injection or cell culture, but hydrolysis accelerates over days. |
| TRIS / HEPES | 7.0 - 8.5 | Poor | Avoid for storage. The common pH ranges of these buffers often accelerate alkaline hydrolysis. |
| Bacteriostatic Water (0.9% Benzyl Alcohol) | 5.0 - 7.0 | Good | Suitable for multi-dose protocols, providing the benzyl alcohol does not interfere with the specific biological assay. |
Concentration and Ionic Strength
Research indicates that NAD+ is more stable at higher concentrations. The principle of molecular crowding suggests that in a highly concentrated solution, the relative availability of free water molecules to facilitate hydrolysis is marginally reduced, and intermolecular interactions may stabilize the conformation. We recommend reconstituting stock solutions at concentrations ranging from 50mM to 100mM, rather than diluting immediately to working concentrations (e.g., 100μM).
Mechanical Handling
While NAD+ is not a long-chain peptide prone to shear stress aggregation like AOD9604 or large proteins like Botulinum Toxin, it is hygroscopic. When handling the lyophilized cake:
- Allow the vial to equilibrate to room temperature before opening. Opening a cold vial in humid lab air causes condensation to form on the hygroscopic powder, initiating immediate hydrolysis before the solvent is even added.
- Add the solvent gently down the side of the vial.
- Swirl gently; vortexing is generally safe for small molecules like NAD+, but gentle inversion is preferred to minimize aeration and oxidation.
Storage Methodologies: Arresting Kinetics
Once in solution, temperature control becomes the primary lever for maintaining stability. The Arrhenius equation dictates that for every 10°C drop in temperature, the rate of chemical reaction (degradation) is reduced by approximately half, though this varies by reaction type. For NAD+, the "Cold Chain" must be unbroken.
Lyophilized Storage
In its powder form, NAD+ is remarkably stable if kept dry.
- Store at -20°C for routine use.
- Store at -80°C for long-term archiving (>1 year).
- Shield from light. While NAD+ is less photosensitive than FAD or certain porphyrins, UV exposure can induce radical formation that attacks the ribose rings.
For researchers managing libraries of metabolic peptides, such as MOTS-c or SS-31, separating lyophilized stocks into single-use aliquots is best practice to avoid repetitive temperature cycling of the main supply.
Aqueous Solution Storage
Once reconstituted, NAD+ must be aliquoted immediately.
- 4°C (Refrigeration): Stable for 2-7 days in acidic to neutral water. Degradation ranges from 1-5% per week depending on pH.
- -20°C (Standard Freezer): Stable for 1-3 months.
- -80°C (Ultracold): Stable for 6-12 months.
The Freeze-Thaw Menace
Repeated freeze-thaw cycles are deleterious to NAD+ integrity. During freezing, water crystallizes as pure ice, creating "freeze concentration" effects where solutes (including protons and buffer salts) become concentrated in the remaining liquid phase before it solidifies. This can cause transient but drastic pH shifts (e.g., phosphate buffers can shift pH by 2-3 units during freezing) and extremely high local concentrations, accelerating second-order degradation reactions.
Protocol: Upon initial reconstitution, divide the master stock into single-use aliquots (e.g., 20μL, 50μL, 100μL) using PCR tubes or low-binding microcentrifuge tubes. Thaw only what is required for the day's experiment.
Experimental Considerations: In Vivo vs. In Vitro
In Vitro Enzymatic Assays
For assays measuring sirtuin activity or PARP kinetics, the presence of ADP-ribose (a breakdown product) impacts results significantly as it is a potent PARP inhibitor. If using stored NAD+ for kinetic studies:
- Verify concentration using spectrophotometry (Molar Extinction Coefficient ε260 ≈ 17,800 M⁻¹cm⁻¹).
- Check the A260/A340 ratio. Pure NAD+ absorbs strongly at 260nm but negligible at 340nm. A rise in A340 indicates reduction to NADH or formation of adducts.
- Ensure the buffer system of the final reaction mix compensates for the acidic nature of the NAD+ stock solution.
In Vivo Administration
Researchers investigating metabolic interventions often pair NAD+ with mitochondrial peptides. When preparing protocols involving NAD+, Epithalon, or 5-amino-1mq (an NNMT inhibitor that raises endogenous NAD+), the delivery vehicle is crucial.
While acidic water is best for storage, injecting a highly acidic solution into animal models can cause tissue necrosis or pain.
- Method: Store NAD+ as a high-concentration stock in SWFI (pH ~6).
- Action: Immediately prior to injection, dilute the stock into PBS or physiological saline to neutralize the pH.
- Warning: Do not store the diluted, neutralized ready-to-inject volume for more than 24 hours.
Synergistic Handling: NAD+ and Mitochondrial Peptides
Current longevity and metabolic research frequently explores the synergy between cytosolic NAD+ pools and mitochondrial-derived peptides. Handling these distinct classes of molecules requires specific attention to their varying stabilities.
SS-31 (Elamipretide) and NAD+
SS-31 acts by targeting cardiolipin on the inner mitochondrial membrane. Unlike NAD+, SS-31 is a peptide and has different solubility and stability characteristics. SS-31 is less pH-sensitive than NAD+ but more prone to surface adsorption. They should not be mixed in the same storage vial. If co-administration is required, mix them immediately prior to use or administer sequentially.
MOTS-c and NAD+
MOTS-c is a mitochondrial-derived peptide regulating metabolic homeostasis. It is highly hydrophobic and prone to aggregation. Mixing MOTS-c with high-ionic strength NAD+ solutions in storage can precipitate the peptide. Always store MOTS-c and NAD+ separately. View our Peptide Synthesis page to understand the structural differences that dictate these handling requirements.
GHK-Cu and NAD+
While GHK-Cu is primarily known for tissue remodeling, some researchers investigate its systemic effects alongside metabolic modulators. Copper ions are transition metals that can catalyze redox reactions. Introducing free copper ions (even complexed in GHK) to an NAD+ solution could theoretically accelerate oxidation-reduction cycles or radical formation. Absolute separation until the point of introduction is mandatory.
Quality Assurance and Verification
At Alpha Carbon Labs, we utilize High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) to verify the purity of our NAD+. However, once the product leaves our facility, verification becomes the researcher's responsibility.
A simple degradation check for the lab scientist involves the Cyanide Addition Assay. Addition of cyanide to NAD+ creates a complex that absorbs at 327 nm. By measuring the absorbance before and after cyanide addition, one can quantify the exact concentration of intact pyridinium rings, distinguishing functional NAD+ from many of its breakdown products (except NMN, which would also react, but is less likely to be a degradation product than Nicotinamide).
Summary of Best Practices
- Keep it Dry: Store lyophilized powder at -20°C or -80°C. Equilibrate to room temp before opening.
- Keep it Acidic: Reconstitute in Sterile Water for Injection or slightly acidified water; avoid basic buffers like TRIS for storage.
- Keep it Cold: Use the "Cold Chain." Thaw on ice.
- Don't Re-freeze: Aliquot immediately upon reconstitution into single-use volumes.
- Check Concentration: Use ε260 = 17,800 M⁻¹cm⁻¹ for quantification.
- Isolate: Do not pre-mix with peptides like Retatrutide, Tirzepatide, or TB-500 during storage.
By adhering to these kinetic principles, researchers ensure that the variability in their data reflects biological reality, not chemical artifact. NAD+ is a powerful tool for understanding cellular age and energy, but it demands respect for its chemical boundaries.
References
- Lowry, O. H., Passonneau, J. V., & Rock, M. K. (1961). The stability of pyridine nucleotides. Journal of Biological Chemistry, 236(10), 2756-2759. https://doi.org/10.1016/S0021-9258(19)61731-2
- Oppenheimer, N. J. (1982). Chemistry and solution conformation of the pyridine coenzymes. In The Pyridine Nucleotide Coenzymes (pp. 51-89). Academic Press. https://doi.org/10.1016/B978-0-12-244750-1.50007-8
- Colpoys, F. & Fogelman, A. (2020). Stability of Nicotinamide Adenine Dinucleotide (NAD+) in aqueous solutions. Journal of Pharmaceutical Sciences, 109(5), 1812-1819. https://doi.org/10.1016/j.xphs.2020.02.012
- Rover Jr, L., et al. (1998). Stability of NADH in solution and in solid state. Analytica Chimica Acta, 366(1-3), 103-109. https://doi.org/10.1016/S0003-2670(98)00151-5
- Yamada, K., Hara, N., Shibata, T., Osago, H., & Tsuchiya, M. (2006). The simultaneous measurement of nicotinamide adenine dinucleotide and related compounds by liquid chromatography–electrospray ionization–tandem mass spectrometry. Analytical Biochemistry, 352(2), 282-291. https://doi.org/10.1016/j.ab.2006.02.016
- Cantó, C., Menzies, K. J., & Auwerx, J. (2015). NAD+ metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metabolism, 22(1), 31-53. https://doi.org/10.1016/j.cmet.2015.05.023
- Yoshino, J., Baur, J. A., & Imai, S. I. (2018). NAD+ intermediates: the biology and therapeutic potential of NMN and NR. Cell Metabolism, 27(3), 513-528. https://doi.org/10.1016/j.cmet.2018.01.038
- Romsicki, Y., & Sharom, F. J. (2001). The ATPase and ATP-binding properties of P-glycoprotein: Evidence for a novel catalytic transition state. Biochemistry, 40(23), 6937-6947. https://doi.org/10.1021/bi002766v
References
- 1. Oppenheimer, N. J. (1982). Chemistry and solution conformation of the pyridine coenzymes. In The Pyridine Nucleotide Coenzymes (pp. 51-89). Academic Press.
- 2. Lowry, O. H., Passonneau, J. V., & Rock, M. K. (1961). The stability of pyridine nucleotides. Journal of Biological Chemistry, 236(10), 2756-2759.
- 3. Yamada, K., Hara, N., Shibata, T., Osago, H., & Tsuchiya, M. (2006). The simultaneous measurement of nicotinamide adenine dinucleotide and related compounds by liquid chromatography–electrospray ionization–tandem mass spectrometry. Analytical Biochemistry, 352(2), 282-291.
- 4. Rover Jr, L., et al. (1998). Stability of NADH in solution and in solid state. Analytica Chimica Acta, 366(1-3), 103-109.
- 5. Romsicki, Y., & Sharom, F. J. (2001). The ATPase and ATP-binding properties of P-glycoprotein: Evidence for a novel catalytic transition state. Biochemistry, 40(23), 6937-6947.
- 6. Yoshino, J., Baur, J. A., & Imai, S. I. (2018). NAD+ intermediates: the biology and therapeutic potential of NMN and NR. Cell Metabolism, 27(3), 513-528.
- 7. Cantó, C., Menzies, K. J., & Auwerx, J. (2015). NAD+ metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metabolism, 22(1), 31-53.
- 8. Colpoys, F. & Fogelman, A. (2020). Stability of Nicotinamide Adenine Dinucleotide (NAD+) in aqueous solutions. Journal of Pharmaceutical Sciences, 109(5), 1812-1819.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.