Incretin Evolution: Mapping the Receptor Occupancy Differences Between Tirzepatide and Retatrutide
Explore the divergent pharmacological profiles of Tirzepatide and Retatrutide in this comprehensive analysis. We map the nuances of dual vs. triple receptor agonism, examining how Glucagon receptor engagement alters metabolic velocity and adipose tissue remodeling.
Introduction: The Paradigm Shift in Incretin Mimetics
The landscape of metabolic research has undergone a seismic shift over the last decade. For years, the scientific focus was tethered almost exclusively to the Glucagon-Like Peptide-1 (GLP-1) receptor. Mono-agonists like semaglutide redefined what was considered possible in terms of glycemic control and weight modulation. However, as influential as GLP-1 receptor agonists (GLP-1RAs) have been, they represent only the first generation of incretin-based therapeutics. The current frontier involves a more complex, multi-faceted approach: the engineering of unimolecular peptides capable of engaging multiple receptor families simultaneously.
This evolution has birthed two distinct titans in the field of peptide research: Tirzepatide, a dual GIP/GLP-1 receptor agonist, and the emerging Retatrutide, a triple GIP/GLP-1/Glucagon receptor agonist. While both peptides share a lineage rooted in incretin biology, their pharmacological profiles, receptor occupancy patterns, and resulting metabolic velocities are fundamentally distinct. For researchers, understanding the nuance between "dual" and "triple" agonism is not merely a matter of counting receptors; it requires a deep dive into how these peptides differentially remodel adipose tissue, influence hepatic lipid metabolism, and alter systemic energy expenditure.
This article aims to provide a comprehensive mapping of the receptor occupancy differences between Tirzepatide and Retatrutide. We will explore the molecular architecture that allows these peptides to "multitask," analyze the specific contributions of the Glucagon receptor (GCGR) to metabolic velocity, and evaluate the divergence in their mechanisms of action, particularly regarding adipose tissue remodeling.
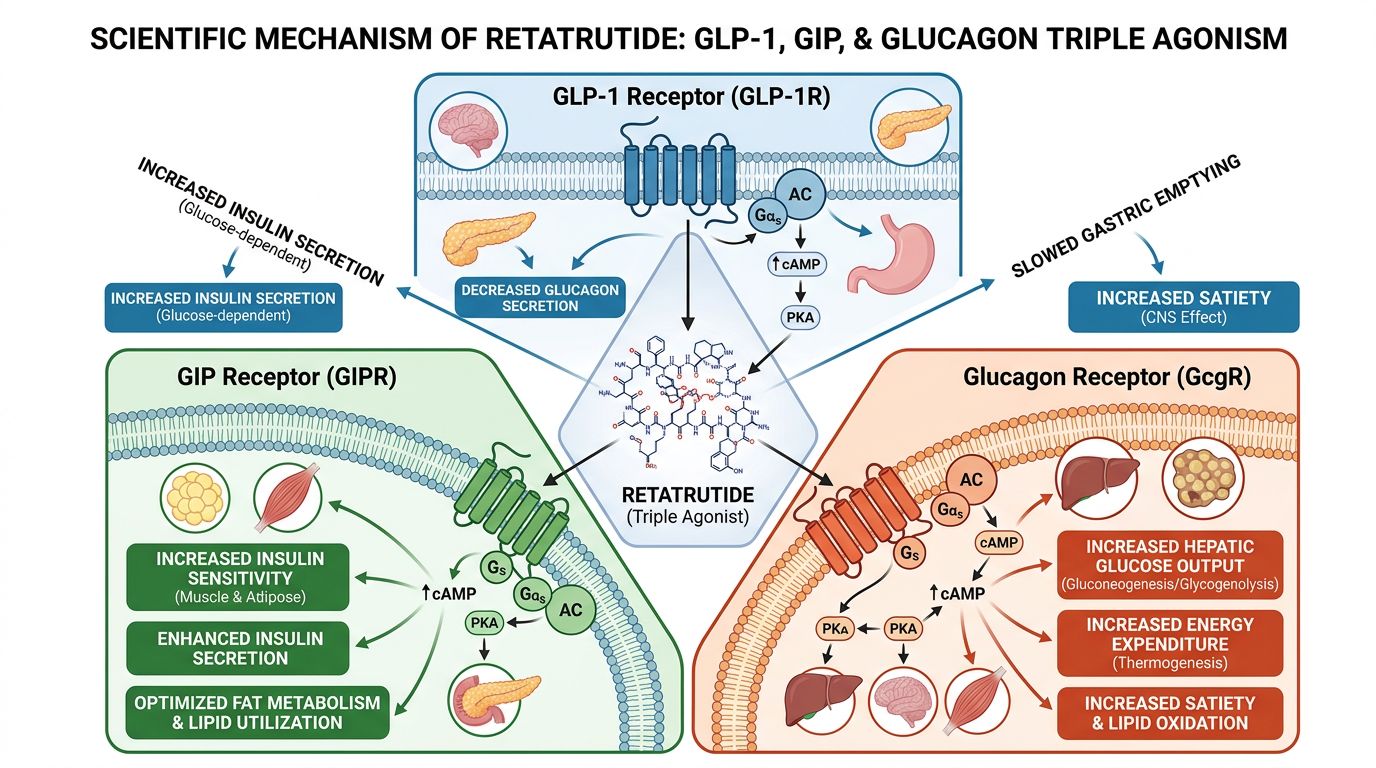
The Molecular Architecture of Multi-Receptor Agonists
To understand the functional differences between these peptides, one must first appreciate the complexity of their synthesis and structural design. The challenge in creating multi-agonists lies in balancing affinity. Native hormones like GLP-1, GIP (Glucose-dependent Insulinotropic Polypeptide), and Glucagon have high specificity for their respective receptors. Designing a single peptide chain that can bind to two or three distinct G-protein coupled receptors (GPCRs) with optimal potency requires sophisticated engineering.
Tirzepatide: The Imbalanced Dual Agonist
Tirzepatide is a 39-amino acid linear peptide. Its structure is based on the native GIP sequence, modified to allow for GLP-1 receptor binding. A C20 fatty diacid moiety is attached via a hydrophilic linker, which binds to serum albumin, extending the peptide's half-life significantly. What makes Tirzepatide unique in peptide synthesis and pharmacology is its "imbalanced" nature.
Contrary to early assumptions that a 50:50 balance is ideal, Tirzepatide exhibits affinity for the GIP receptor that is comparable to native GIP, but its affinity for the GLP-1 receptor is actually lower than that of native GLP-1. This is a deliberate design choice that leverages the synergistic potential of GIP signaling to enhance the tolerability and efficacy of GLP-1 agonism. By anchoring the molecule in GIP efficacy, Tirzepatide achieves a metabolic modulation that mono-agonists cannot replicate.
Retatrutide: The Triple "G" Agonist
Retatrutide elevates the complexity by integrating activity at the Glucagon receptor (GCGR). It is a single peptide backbone modified with a C20 fatty acid chain, similar to Tirzepatide, to ensure long duration of action. However, the sequence is engineered to bind GLP-1, GIP, and GCGR.
The inclusion of Glucagon agonism is the critical differentiator. Historically, Glucagon was viewed solely as a counter-regulatory hormone that raised blood glucose—a "diabetic" hormone. Modern research has rehabilitated Glucagon's reputation, identifying its powerful role in increasing energy expenditure (thermogenesis) and promoting hepatic lipid oxidation. Retatrutide balances the insulinotropic effects of GLP-1 and GIP with the catabolic, energy-burning effects of Glucagon.
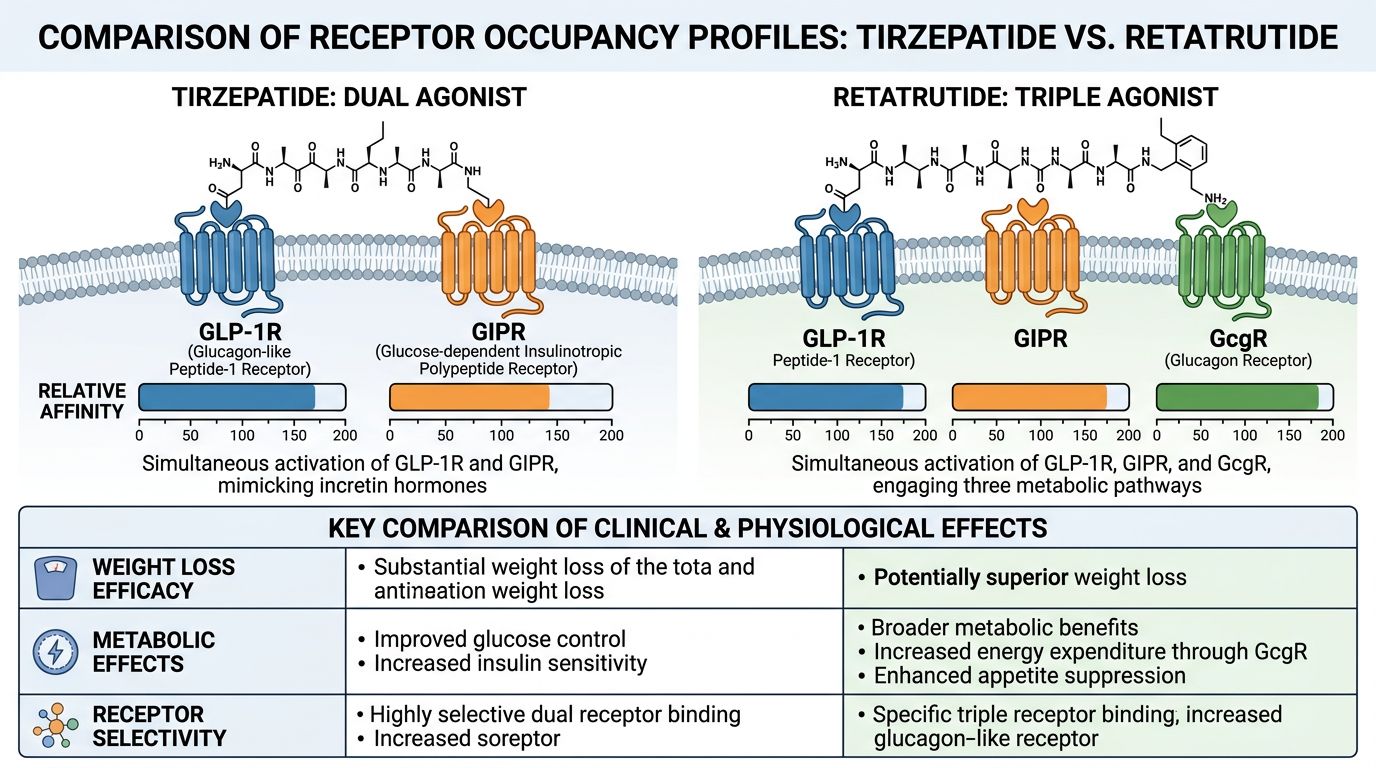
Receptor Occupancy Profiles: Mapping the Divergence
The "personality" of a research peptide is defined by its receptor occupancy—essentially, how tightly it binds to a receptor and what intracellular signals it triggers once bound. The divergence between Tirzepatide and Retatrutide is most evident when analyzing their EC50 values (the concentration required to induce a 50% maximal response) and their bias toward different signaling pathways (cAMP vs. beta-arrestin recruitment).
| Parameter | Tirzepatide (Dual) | Retatrutide (Triple) |
|---|---|---|
| GIP Receptor Potency | High (Comparable to native GIP) | High (High potency) |
| GLP-1 Receptor Potency | Moderate (Lower than native GLP-1) | High (Comparable to native GLP-1) |
| Glucagon Receptor Potency | None (negligible) | Moderate/High (tuned for metabolic impact) |
| Signaling Bias | Biased toward cAMP; reduced beta-arrestin recruitment at GLP-1R | Balanced agonism across receptors |
| Primary Metabolic Driver | Insulin sensitization + Satiety | Insulin sensitization + Satiety + Thermogenesis |
The GIP Component: The Foundation of Storage Regulation
Both peptides rely heavily on the GIP receptor. In healthy physiology, GIP is anabolic—it facilitates lipid uptake into white adipose tissue (WAT) to buffer post-prandial nutrients. This prevents ectopic fat deposition (fat storage in the liver or muscles where it causes damage). In the context of pharmacological doses found in research, GIP receptor agonism appears to desensitize this storage signal while enhancing insulin sensitivity and preventing the nausea often associated with high-dose GLP-1 receptor activation.
Tirzepatide's profound occupancy of the GIP receptor is believed to be the reason for its superior efficacy over Semaglutide. It utilizes GIP to allow for higher tolerable signaling through the GLP-1 pathway.
The Glucagon Differentiator: Retatrutide’s Kinetic Advantage
The receptor occupancy map changes drastically with Retatrutide due to the GCGR engagement. When Retatrutide occupies the Glucagon receptor in the liver, it stimulates glycogenolysis and gluconeogenesis (which is counterbalanced by the insulinotropic effects of GLP-1 and GIP) and, crucially, fatty acid oxidation.
In brown adipose tissue (BAT), Glucagon receptor occupancy stimulates uncoupling protein 1 (UCP1) expression, leading to thermogenesis. This suggests that while Tirzepatide achieves results by reducing energy intake and improving storage efficiency, Retatrutide achieves results by reducing intake and actively increasing metabolic outflow. This increase in "metabolic velocity" is the defining characteristic of the triple agonist class.
Metabolic Velocity and Adipose Tissue Remodeling
For researchers investigating obesity and metabolic syndrome, the remodeling of adipose tissue is a primary endpoint. Obesity is characterized by hypertrophic adipocytes (fat cells that are too large), macrophage infiltration, and inflammation. The transition from this pathological state to healthy, smaller adipocytes is termed remodeling.
Tirzepatide: Improving Adipose Functionality
Tirzepatide promotes adipose tissue remodeling primarily through blood flow enhancement and insulin sensitization. Research indicates that GIP receptor signaling in adipocytes increases blood flow to the tissue, facilitating the clearance of inflammatory metabolites. By improving the ability of subcutaneous fat to store lipids safely, Tirzepatide reduces the visceral fat burden—the dangerous fat surrounding organs.
The "velocity" here is moderate. The weight reduction observed in Tirzepatide models is driven largely by caloric deficit (reduced intake). The tissue remodels because the energy surplus is removed, allowing adipocytes to shrink and normalize function.
Retatrutide: Accelerating the Burn
Retatrutide introduces a new vector: increased energy expenditure. By engaging the Glucagon receptor, Retatrutide appears to induce "browning" of white adipose tissue—the conversion of storage-focused white fat cells into energy-burning beige or brown fat cells. This process, thermogenesis, physically wastes energy as heat.
Research models suggest that Retatrutide creates a higher metabolic velocity. Even when caloric intake is clamped (matched) in animal studies, subjects treated with triple agonists often lose more weight than those on dual agonists, implicating the Glucagon-mediated increase within resting metabolic rate. This suggests Retatrutide may be more effective in breaking metabolic plateaus where adaptive thermogenesis (the body's tendency to slow metabolism during weight loss) typically hinders progress.
Comparative Efficacy in Research Findings
When evaluating these peptides in a laboratory or clinical setting, the data reflects the mechanistic differences outlined above. It is crucial to source high-purity peptides to replicate these findings, necessitating review of COA documents for any reagents used.
Weight Loss and Glycemic Control
Pre-clinical and clinical data have consistently placed Retatrutide ahead of the curve regarding raw mass reduction. In the rodent models cited by Finan et al. and subsequent human trials by Jastreboff et al., the triple agonist approach yielded reductions in body mass that exceeded 24-25%, figures previously attainable only via bariatric surgery. Tirzepatide, while revolutionary compared to mono-agonists, typically benchmarks at the 20-22% range in similar high-dose protocols.
Hepatic Health: MASH and NASH
One of the most promising applications for these peptides is in the treatment of Metabolic Dysfunction-Associated Steatohepatitis (MASH). Here, the Glucagon component of Retatrutide shines. Glucagon directly stimulates the oxidation of liver fat. While Tirzepatide reduces liver fat indirectly by reducing total body weight and improving insulin sensitivity, Retatrutide attacks hepatic steatosis directly via GCGR signaling.
Researchers focusing on hepatology often prefer the triple agonist profile for studies involving rapid de-lipidation of the liver. However, Tirzepatide remains a robust standard for general metabolic improvement and is frequently compared against other peptides like AOD9604 or analogues in lipid research.
Safety Considerations and Off-Target Effects
The introduction of Glucagon agonism into the equation brings specific safety considerations that researchers must monitor.
Heart Rate and Hemodynamics
The Glucagon receptor is expressed in cardiac tissue (the sinoatrial node). Activation can lead to chronotropic effects (increased heart rate). Research involving Retatrutide has demonstrated a dose-dependent increase in heart rate, generally peaking at 2-4 beats per minute higher than baseline in human subjects, though usually transient.
Tirzepatide also influences heart rate, likely through GLP-1 mediated sympathetic activation, but the absence of direct GCGR stimulation may present a slightly different hemodynamic profile. In research settings, rigorous monitoring of cardiovascular parameters is standard protocol when utilizing these agents.
Gastrointestinal Tolerability
Both peptides share the typical incretin side effect profile: nausea, vomiting, and diarrhea, primarily associated with the initiation of treatment and dose escalation. The GIP component in both Tirzepatide and Retatrutide is hypothesized to mitigate some of the nausea associated with profound GLP-1 activation, potentially by acting on the area postrema in the brainstem (the emetic center) in a counter-regulatory fashion.
Quality and Purity in Research
Given the complexity of these long-chain, fatty-acid-modified peptides, synthesis impurities can significantly alter experimental outcomes. Truncated sequences or incomplete fatty acid attachment can lead to immunogenicity or loss of potency. Researchers should prioritize suppliers that provide transparent transparency regarding quality control processes, ensuring that the receptor occupancy observed in the lab correlates accurately with the dosage administered.
Mechanistic Deep Dive: Intracellular Signaling
To fully grasp the "Incretin Evolution," one must look inside the cell. GPCRs like the GLP-1, GIP, and Glucagon receptors signal primarily through the Gs-alpha subunit, leading to the production of cyclic AMP (cAMP) and the activation of Protein Kinase A (PKA).
Beta-Arrestin and Receptor Internalization
A critical aspect of Tirzepatide's pharmacology is its biased agonism. Studies by Willard et al. have shown that Tirzepatide recruits beta-arrestin—a protein responsible for receptor desensitization and internalization—less potently than native GLP-1. This potentially allows the GLP-1 receptor to remain on the cell surface longer, maintaining signal transduction despite the presence of the ligand. This lack of desensitization may explain why Tirzepatide maintains efficacy over long durations.
Retatrutide’s signaling bias is currently under intense investigation. Whether the triple agonist induces rapid internalization of the Glucagon receptor (which could lead to tachyphylaxis or diminishing returns) is a subject of ongoing inquiry. Understanding this bias is key to optimizing dosing schedules in longitudinal studies.
Future Directions: Beyond Triple Agonists?
The development of Retatrutide marks a high point in peptide engineering, but the field is not static. Research is already looking toward other combinations or refined "tuned" versions of these molecules. For instance, peptides like Mazdutide (a dual GLP-1/GCG agonist) are exploring the omission of GIP to see if the dual "burning and suppressing" mechanism is sufficient without the storage-modulating effects of GIP.
Furthermore, the combination of these potent metabolic drivers with agents that preserve muscle mass is a critical next step. Rapid weight reduction often includes lean mass loss. Co-administration research involving agents that inhibit myostatin (like ACE-031) or promote muscle protein synthesis could represent the next "stack" in metabolic research protocols.
Conclusion
The evolution from mono-agonists to the complex poly-pharmacology of Tirzepatide and Retatrutide represents a golden age in metabolic research. While Tirzepatide established the power of the GIP/GLP-1 synergy—effectively rewriting the rules of potency and tolerability—Retatrutide pushes the envelope of metabolic velocity through the addition of Glucagon receptor engagement.
For the researcher, the choice between studying Tirzepatide or Retatrutide often comes down to the specific metabolic pathways of interest. If the focus is on maximizing insulin sensitivity and storage efficiency with high tolerability, Tirzepatide remains the benchmark. If the goal is to investigate maximal energy expenditure, hepatic lipid oxidation, and the limits of pharmacological weight reduction, Retatrutide offers a more aggressive, multi-modal tool. Both peptides underscore the critical importance of receptor occupancy and the intricate balance of the endocrine system.
References
- Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387:205-216. https://doi.org/10.1056/NEJMoa2206038
- Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. N Engl J Med. 2023;389:514-526. https://doi.org/10.1056/NEJMoa2301972
- Coskun T, et al. Ly3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14. https://doi.org/10.1016/j.molmet.2018.09.009
- Finan B, et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nat Med. 2015;21:27-36. https://doi.org/10.1038/nm.3761
- Knerr PJ, et al. Next generation GLP-1/glucagon receptor co-agonists with improved therapeutic potential. Mol Metab. 2022;63:101531. https://doi.org/10.1016/j.molmet.2022.101531
- Müller TD, et al. The New Biology and Pharmacology of Glucagon. Physiol Rev. 2017;97(2):721-766. https://doi.org/10.1152/physrev.00025.2016
- Willard, F.S., et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532. https://doi.org/10.1172/jci.insight.140532
- Gribble FM, Reimann F. Metabolic Messengers: Glucagon-like peptide 1. Nat Metab. 2021;3:142–143. https://doi.org/10.1038/s42255-020-00327-x
- Nauck MA, et al. The broad clinical potential of receptor agonists for glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide. Nat Rev Endocrinol. 2024. https://doi.org/10.1038/s41574-024-00987-w
- Samms RJ, et al. GIPR signaling in immune cells and adipose tissue. Peptides. 2020;125:170252. https://doi.org/10.1016/j.peptides.2019.170252
- Capozzi ME, et al. Glucagon lowers glycemia when β-cells are active. JCI Insight. 2019;5:e129954. https://doi.org/10.1172/jci.insight.129954
References
- 1. Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387:205-216.
- 2. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. N Engl J Med. 2023;389:514-526.
- 3. Coskun T, et al. Ly3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
- 4. Finan B, et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nat Med. 2015;21:27-36.
- 5. Knerr PJ, et al. Next generation GLP-1/glucagon receptor co-agonists with improved therapeutic potential. Mol Metab. 2022;63:101531.
- 6. Müller TD, et al. The New Biology and Pharmacology of Glucagon. Physiol Rev. 2017;97(2):721-766.
- 7. Willard, F.S., et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
- 8. Gribble FM, Reimann F. Metabolic Messengers: Glucagon-like peptide 1. Nat Metab. 2021;3:142–143.
- 9. Nauck MA, et al. The broad clinical potential of receptor agonists for glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide. Nat Rev Endocrinol. 2024.
- 10. Samms RJ, et al. GIPR signaling in immune cells and adipose tissue. Peptides. 2020;125:170252.
- 11. Capozzi ME, et al. Glucagon lowers glycemia when β-cells are active. JCI Insight. 2019;5:e129954.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.