Hepatic Lipid Regulation: The Nuanced Role of Glucagon Reciprocal Activation in Retatrutide Studies
This article investigates the molecular mechanism of Retatrutide, focusing on how its unique Glucagon receptor activation synergizes with GLP-1 and GIP to target hepatic fat accumulation.
Introduction: The Evolution of Incretin Mimetics in Metabolic Research
In the landscape of metabolic research, the progression from mono-agonists to poly-agonists has redefined the therapeutic possibilities for managing obesity and its comorbid conditions. For years, Semaglutide served as the benchmark for GLP-1 (Glucagon-like Peptide-1) receptor agonism, focusing primarily on glycemic control and appetite suppression. This was followed by the emergence of Tirzepatide, a dual GLP-1 and GIP (Glucose-dependent Insulinotropic Polypeptide) receptor agonist, which demonstrated that synergistic activation of multiple pathways could yield superior weight loss and metabolic outcomes.
However, the most recent frontier in this scientific endeavor involves the integration of a third hormone receptor: the Glucagon receptor (GCGR). Retatrutide (LY3437943) represents this new class of "triple agonists" (GLP-1/GIP/GCGR). While GLP-1 and GIP are predominantly recognized for their roles in insulin secretion and anorexigenic signaling, the inclusion of Glucagon agonism introduces a specialized mechanism—reciprocal activation—that fundamentally alters hepatic lipid regulation. This article explores the nuanced role of the Glucagon receptor in Retatrutide studies and how it distinguishes this peptide from its predecessors by targeting hepatic fat accumulation with unprecedented precision.
The Molecular Architecture of Retatrutide
Retatrutide is a multi-receptor agonist engineered to maintain a delicate balance between three distinct signaling pathways. To understand its role in hepatic regulation, one must first examine its structural affinity for its target receptors. Retatrutide is a 39-amino-acid peptide based on the GIP backbone, modified to include specific sequences that engage the GLP-1 and Glucagon receptors. Research indicates that its potency is balanced to ensure that the stimulatory effects of Glucagon do not override the glucose-lowering benefits of the incretin components.
The Triple Agonist Potency Ratio
Unlike earlier iterations of metabolic peptides, the efficacy of Retatrutide lies in its unbalanced yet optimized potency across the three receptors. Current research suggests a ratio favoring GIP potency, followed by GLP-1 and GCGR. This hierarchical activation is designed to maximize lipid mobilization (via Glucagon) and metabolic efficiency (via GIP) while mitigating the hyperglycemic risk naturally associated with isolated Glucagon activation.
For researchers interested in high-purity synthesis of such complex molecules, our peptide synthesis facility ensures that the precise amino acid sequence and fatty acid acylation required for Retatrutide's long half-life are strictly maintained. Verification of these sequences can be found in our COA documents.
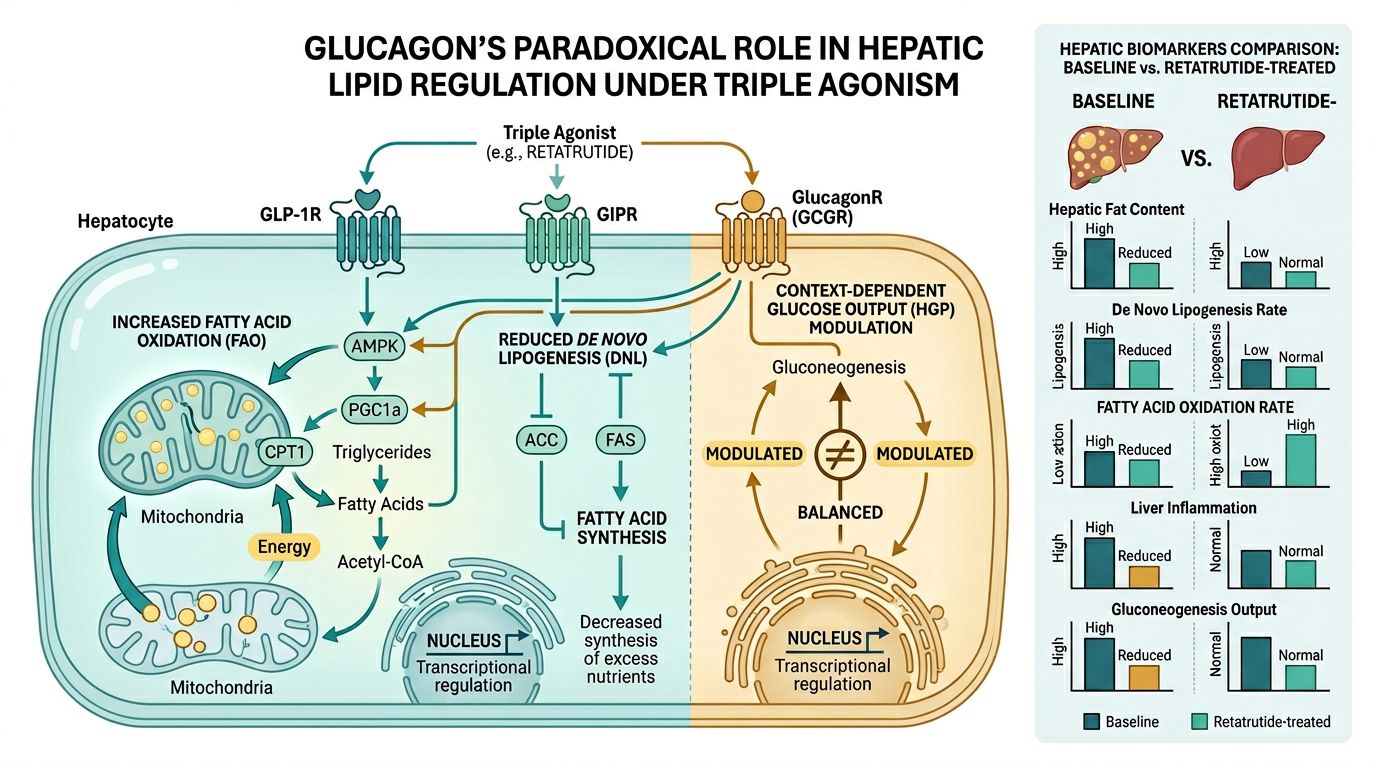
Glucagon’s Paradoxical Role in Metabolism
In traditional endocrinology, Glucagon is often viewed as the "antagonist" to insulin—a hormone that increases blood glucose levels through glycogenolysis and gluconeogenesis. However, recent studies in Retatrutide highlight a different facet of Glucagon: its powerful role in energy expenditure and hepatic lipid metabolism. When integrated into a triple agonist framework, Glucagon acts as a metabolic "accelerator."
Lipid Mobilization and Fatty Acid Oxidation
The Glucagon receptor is highly expressed in the liver. Its activation signals the mitochondria to increase the rate of beta-oxidation—the process by which fatty acids are broken down to produce energy. In models of metabolic-associated steatotic liver disease (MASLD, formerly NAFLD), Retatrutide has demonstrated the ability to significantly "clear" hepatic fat deposits. This is hypothesized to occur through two primary mechanisms:
- Increased Mitochondrial Oxygen Consumption: GCGR activation upregulates mitochondrial thermogenesis, consuming excess lipids as a fuel source.
- Inhibition of De Novo Lipogenesis: By shifting the liver's metabolic state toward oxidation, the synthesis of new fatty acids is suppressed.
Synergy with GIP and GLP-1
This is where the "reciprocal activation" becomes critical. While Glucagon promotes the release of glucose from the liver, the GLP-1 and GIP components of Retatrutide act on the pancreas to stimulate insulin secretion in a glucose-dependent manner. This creates a physiological loop where the liver is stimulated to burn fat, yet the systemic blood sugar remains stable because the incretin components provide a compensatory "buffer."
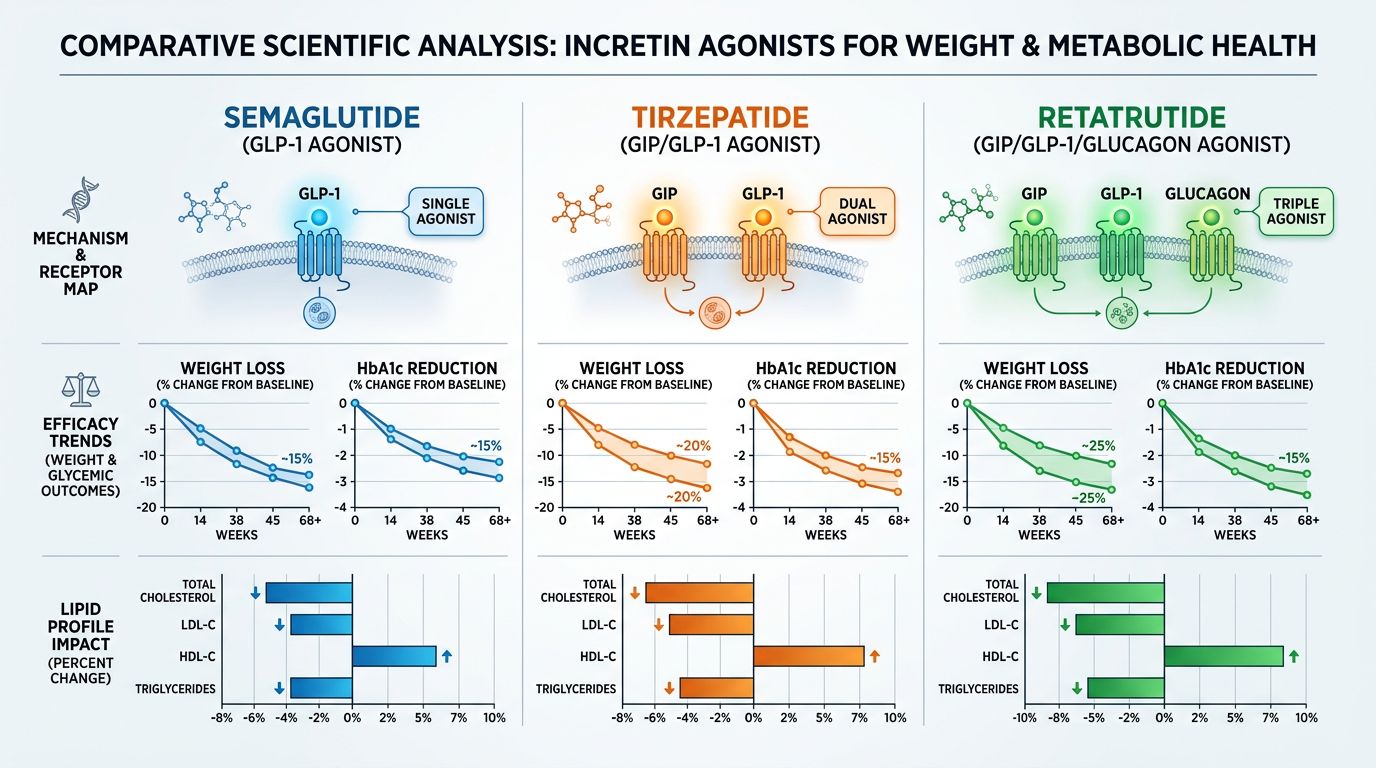
Comparative Analysis: Retatrutide vs. Tirzepatide vs. Semaglutide
To appreciate the specific impact of the Glucagon receptor, it is helpful to compare Retatrutide with the prior gold standards of research.
| Feature | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Receptor Targets | GLP-1R | GLP-1R + GIPR | GLP-1R + GIPR + GCGR |
| Primary Hepatic Effect | Indirect (via weight loss) | Moderate (improved lipid profile) | Direct (targeted fat clearance) |
| Energy Expenditure | Neutral/Minimal change | Mild increase | Significant increase (Thermogenic) |
| Hepatic Fat Reduction | ~30-40% in studies | ~50-70% in studies | Up to 80-90% in Phase 2 |
While Tirzepatide represented a massive leap forward in metabolic efficiency, the addition of the Glucagon receptor in Retatrutide provides a tool for "reprogramming" hepatic energy use that GLP-1/GIP agonists cannot achieve alone. This distinction is vital for researchers focused on liver-specific pathology.
Mitochondrial Health and the Triple Agonist
The role of Glucagon extends beyond simple lipid clearance; it touches upon mitochondrial energetics. Researchers are increasingly investigating how GCGR agonism affects cellular health at the organelle level. Similar to how SS-31 directly stabilizes mitochondrial membranes, the Glucagon component of Retatrutide appears to optimize hepatic mitochondrial efficiency by promoting a high-flux metabolic state.
In research environments where mitochondrial oxidative stress is a variable, utilizing Retatrutide alongside other mitochondrial-targeting agents like MOTS-c or NAD+ precursors may provide a comprehensive view of metabolic restoration. The ability of the Glucagon receptor to "push" the mitochondria to utilize stored lipids is a cornerstone of its efficacy in clearing hepatic steatosis.
Safety and Quality Control in Triple Agonist Research
The complexity of synthesizing a triple agonist like Retatrutide cannot be overstated. With 39 amino acids and specific acylation patterns, even minor deviations in the sequence can result in a loss of receptor selectivity or an increase in adverse effects (such as uncontrolled tachycardia or hyperglycemia). At Alpha Carbon Labs, we emphasize the importance of quality control through rigorous HPLC and MS testing.
Key Research Considerations:
- Tachycardia Concerns: Glucagon receptors are also present in the heart. High doses of triple agonists in rodent models have shown increases in heart rate. Research must monitor this chronotropic effect closely.
- Glucose Fluctuations: While the incretin components generally balance the Glucagon component, researchers must evaluate the peptide's performance in varied glycemic states (fasting vs. postprandial).
- Gastrointestinal Tolerability: Like all GLP-1 based peptides, dosage titration is essential to manage gastrointestinal motility.
The Future of Hepatic Lipid Research: Beyond Weight Loss
The most compelling data from recent Phase 2 Retatrutide studies is not just the 24% weight loss over 48 weeks, but the near-complete resolution of hepatic steatosis in a significant percentage of patients. This suggests that the Glucagon receptor may be the "missing link" in treating advanced metabolic disorders where weight loss alone is insufficient to reverse organ damage.
For researchers exploring the synergistic effects of peptides on tissue repair and inflammation, combining the metabolic power of Retatrutide with tissue-regenerative peptides like BPC-157 or TB-500 (often found in our BPC-157 + TB-500 Blend) could reveal new insights into how systemic metabolic health influences localized tissue healing.
Experimental Applications
Experimental designs currently utilize Retatrutide to study:
- MASLD/MASH Reversal: Quantifying the rate of lipid droplet reduction via MRI-PDFF (Proton Density Fat Fraction).
- Energy Expenditure: Using indirect calorimetry to measure the thermogenic impact of the GCGR component.
- Muscle-Liver Crosstalk: How increased hepatic lipid oxidation affects glucose availability during physical exertion.
Conclusion
The Glucagon receptor, once feared for its hyperglycemic potential, has been masterfully reclaimed in the form of Retatrutide. By leveraging reciprocal activation—balancing the thermogenic and lipolytic power of Glucagon with the insulinotropic and anorexigenic nature of GIP and GLP-1—retatrutide offers a nuanced approach to hepatic lipid regulation. For the scientific community, this peptide represents more than just a weight-loss tool; it is a sophisticated instrument for probing the limits of human metabolic flexibility.
As research continues, the primary focus remains on the specific molecular modifications that allow these three pathways to coexist. Alpha Carbon Labs remains committed to providing the highest purity research materials to facilitate these breakthroughs. Researchers can access our full range of metabolic peptides, including Cagrilintide and Mazdutide, to continue exploring the next generation of metabolic science.
References
- 1. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. New England Journal of Medicine. 2023;389(6):514-526.
- 2. Sanyal AJ, et al. Retatrutide Phase 2 Data in NAFLD: Significant Reductions in Hepatic Fat Content. Journal of Hepatology. 2023;78(1):S23-S24.
- 3. Finan B, et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nature Medicine. 2015;21(1):27-36.
- 4. Müller TD, et al. Glucagon-like peptide 1 (GLP-1). Molecular Metabolism. 2019;30:72-130.
- 5. Campbell JE, Drucker DJ. Islet α cells and glucagon—critical regulators of energy homeostasis. Nature Reviews Endocrinology. 2015;11(6):329-338.
- 6. Coskun T, et al. LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss. Cell Metabolism. 2022;34(9):1234-1245.e9.
- 7. Evers A, et al. Design of dual and triple agonists of GLP-1, GIP, and glucagon receptors for the treatment of obesity and type 2 diabetes. Journal of Peptide Science. 2017;23(7-8):470-474.
- 8. Lynch L, et al. Glucagon-like Peptide-1 Controls Tumor Necrosis Factor α Expression and Astrogliosis in the Kidney. Journal of the American Society of Nephrology. 2014;25(7):1455-1467.
- 9. Heppner KM, et al. Glucagon regulation of energy expenditure. Physiology & Behavior. 2010;100(5):545-548.
- 10. Rossi MC, et al. Triple agonists in the management of metabolic dysfunction-associated steatotic liver disease (MASLD). Trends in Endocrinology & Metabolism. 2024;35(2):112-125.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.