Experimental Design for Regenerative Medicine: Measuring Outcomes of the Wolverine Stack in Orthopedic Models
Explore the scientific methodology for designing regenerative medicine experiments using the BPC-157, TB-500, and GHK-Cu 'Wolverine Stack' in orthopedic research models.
Introduction to Multi-Peptide Systems in Regenerative Research
In the evolving landscape of regenerative medicine, the transition from monotherapy to multi-peptide "stacks" represents a paradigm shift in addressing complex orthopedic injuries. Single-molecule interventions often fail to address the multifaceted nature of tissue repair, which necessitates simultaneous modulation of angiogenesis, collagen synthesis, and inflammatory resolution. The combination of BPC-157, TB-500 (Thymosin Beta-4), and GHK-Cu—frequently termed the "Wolverine Stack" in research circles—is currently being scrutinized for its synergistic potential in accelerating connective tissue restoration.
For the rigorous researcher, implementing such a stack requires more than anecdotal dosing protocols. It demands a sophisticated understanding of molar ratios, peptide-peptide interactions, and the specific biological windows where each compound exerts its primary influence. This guide explores the experimental design, pharmacokinetic considerations, and quantitative outcome measures for evaluating the BPC-157 + TB-500 + GHK-Cu Blend within controlled orthopedic injury models.
Pharmacological Profiles of the Stack Components
BPC-157: The Angiomodulatory Agent
BPC-157 (Body Protection Compound 157) is a pentadecapeptide derived from human gastric juice. Its primary mechanism involves the upregulation of Vascular Endothelial Growth Factor (VEGF) and the activation of the FAK-paxillin pathway. In orthopedic models, its role is foundational: establishing the vascular infrastructure necessary for nutrient delivery and waste removal in relatively avascular tissues like tendons and ligaments. Quality control in these experiments is paramount; researchers are encouraged to review Alpha Carbon Labs Quality Control standards to ensure peptide purity exceeds 99%.
TB-500 (Thymosin Beta-4): The Migratory Driver
TB-500 is a synthetic version of the naturally occurring 43-amino acid peptide Thymosin Beta-4. Its primary mechanism is actin-sequestering, which facilitates cell migration (chemotaxis) and prevents apoptosis. In tendon-to-bone healing models, TB-500 is responsible for the recruitment of progenitor cells to the site of injury. This peptide operates via a distinct biological pathway from BPC-157, providing the "migration" signal that complements BPC-157’s "vascular" signal.
GHK-Cu: The Remodeling Catalyst
GHK-Cu (Glycyl-L-histidyl-L-lysine copper) acts as a remodeling signal. It is known to modulate both TGF-beta expression and matrix metalloproteinases (MMPs). This ensures that the collagen deposited during the proliferative phase—often disorganized Type III collagen—is remodeled into the structurally superior Type I collagen. For researchers focused on the final stages of tissue integration, theinclusion of GHK-Cu is critical for restoring tensile strength.
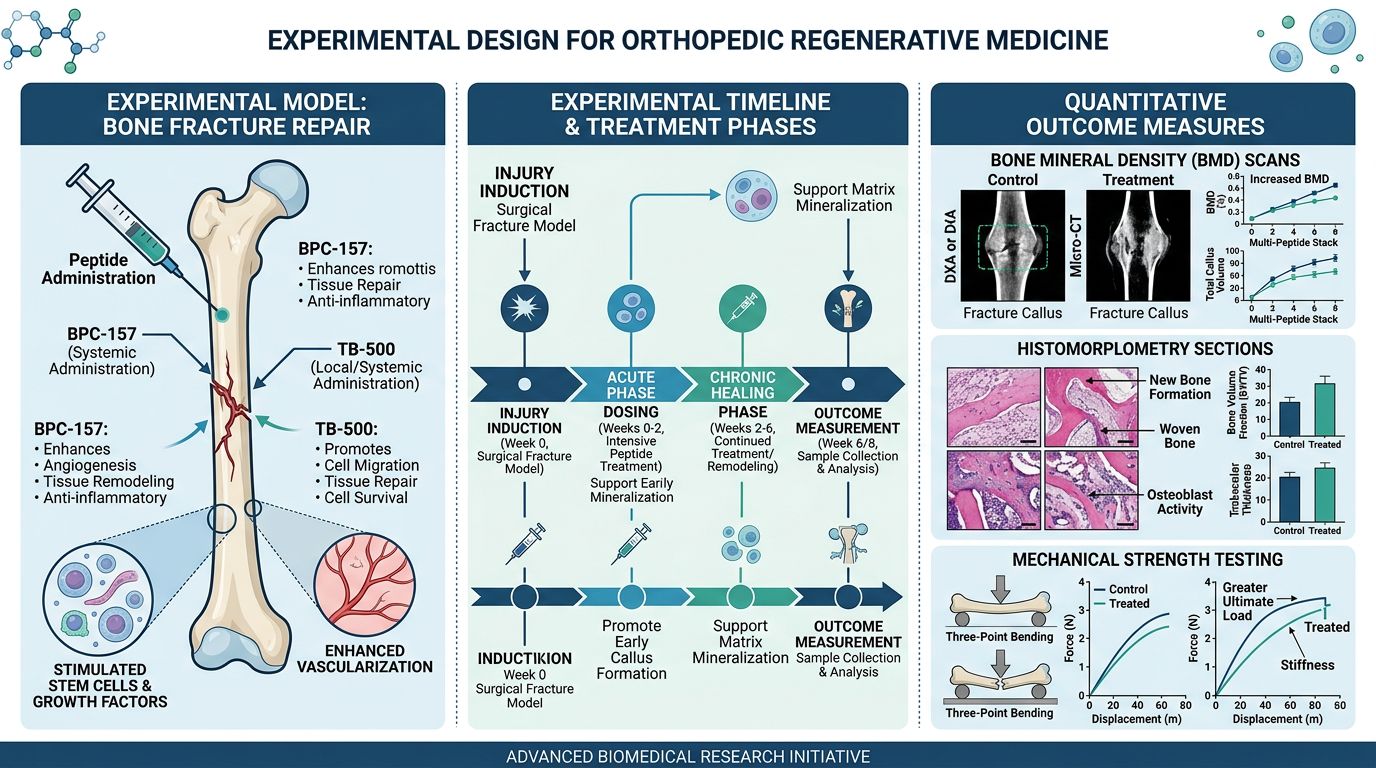
Experimental Design: Calculating Molar Ratios and Dosing
When designing a multi-peptide experiment, equating mass (e.g., 500mcg of each) is physiologically imprecise because the molar masses of these peptides vary significantly. To achieve true synergy, researchers should calculate the molar concentration (micromoles) to ensure the target receptors are proportionately saturated.
Peptide Molecular Weights Comparison
| Peptide | Molecular Weight (approx. g/mol) | Proposed Molar Ratio (Standard Model) | Role in Orthopedic Model |
|---|---|---|---|
| BPC-157 | 1419.5 | 2 | Angiogenesis / NO Modulation |
| TB-500 | 4963.5 | 1 | Actin-sequestering / Cell Migration |
| GHK-Cu | 340.4 (tripeptide) | 5 | Extracellular Matrix Remodeling |
High-precision research requires meticulous documentation; researchers should refer to COA Documents to verify the exact TFA salt content, which can affect the net peptide weight used in these calculations.
Methodology for Orthopedic Injury Simulation
1. Model Selection: The Rat Achilles Rupture
The rodent Achilles tendon rupture remains the gold standard for testing the Wolverine Stack. This model allows for the measurement of both histological healing and biomechanical load-to-failure. The experiment should be divided into four cohorts to determine the additive effect:
- Control Group: Saline vehicle.
- Binary Group A: BPC-157 + TB-500 Blend.
- Binary Group B: BPC-157 + GHK-Cu.
- Synergy Group: BPC-157 + TB-500 + GHK-Cu Blend.
2. Administration Protocols
In regenerative models, local vs. systemic administration is a key variable. While BPC-157 exhibits strong systemic activity, TB-500’s migratory effects are often enhanced when administered in the vicinity of the injury. For researchers utilizing custom peptide synthesis, the stability of the blend in various pH environments must be considered to prevent premature degradation of the GHK-Cu complex.
Quantitative Outcome Measures
Biomechanical Testing
The ultimate metric of orthopedic success is tensile strength. Following the sacrifice of the animal model (typically at Day 14, 28, and 56), the tendon is harvested and subjected to a motorized longitudinal pull.
Key Variables:
- Ultimate Tensile Strength (UTS): The maximum stress the tendon can withstand.
- Young’s Modulus: Measurement of the stiffness of the newly formed tissue.
- Energy to Failure: Total energy absorbed by the tissue before rupture.
Histomorphometric Analysis
Tissues should be stained using Masson’s Trichrome to visualize collagen density and orientation. Researchers should quantify the ratio of Type I to Type III collagen using polarized light microscopy. A successful Wolverine Stack intervention typically shows an accelerated transition from the chaotic Type III fiber arrangement to a parallel, organized Type I structure by Day 21.
Immunohistochemistry (IHC)
To confirm the mechanism of action, IHC should target:
- CD31 expression (indicator of endothelial vessel density).
- Alpha-SMA (Smooth Muscle Actin) for myofibroblast activity.
- PCNA (Proliferating Cell Nuclear Antigen) to measure cellular proliferation at the injury site.
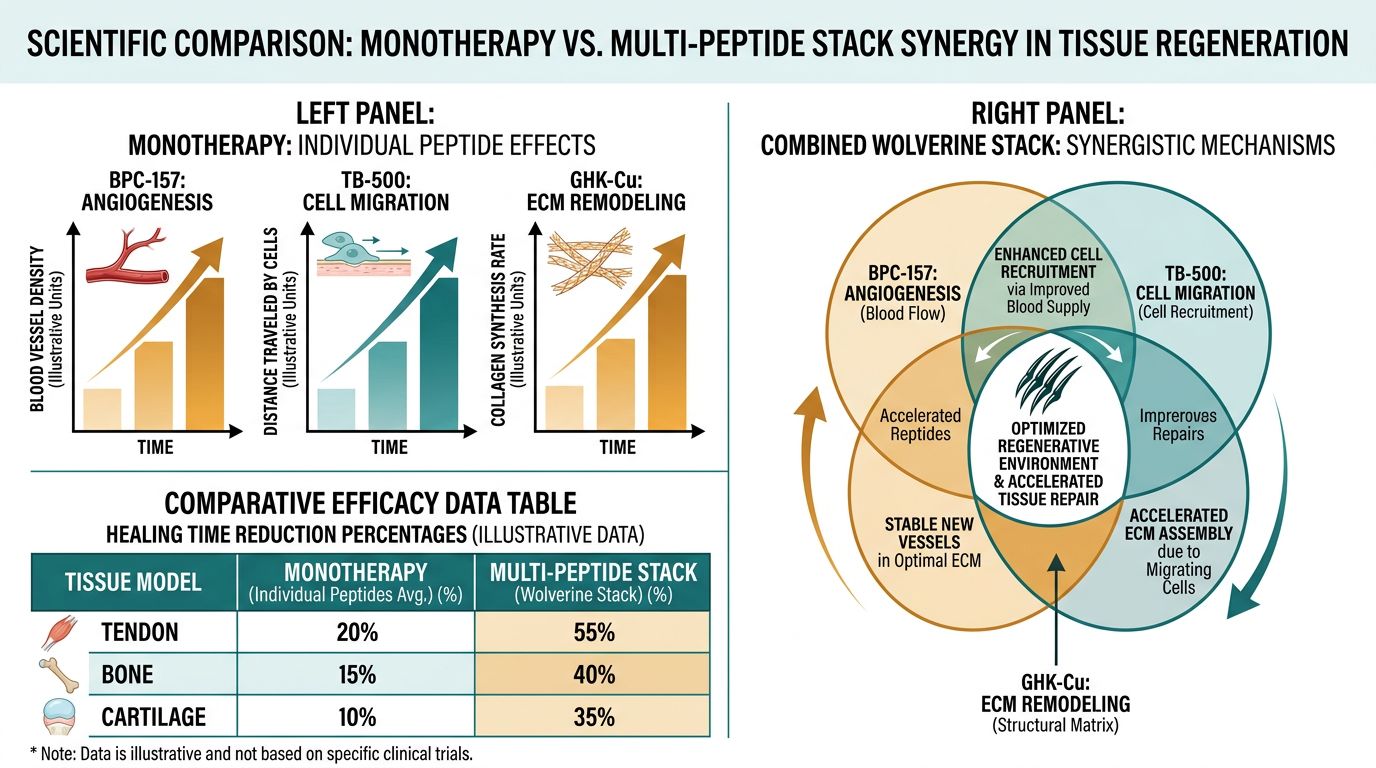
Synergistic Interactions: Why the Stack Outperforms Monotherapy
The theoretical superiority of the Wolverine Stack lies in the "Cascade Effect." While BPC-157 initiates the repair by upregulating receptors like VEGFR2, TB-500 provides the raw cell movement necessary to populate the newly vascularized area. Finally, GHK-Cu ensures that this flurry of cellular activity results in functional tissue rather than hypertrophic scarring. This is particularly relevant in chronic models where the healing process has stalled, a condition often addressed by adding metabolic catalysts like MOTS-c or mitochondrial supporters like SS-31 to the research protocol.
Special Considerations in Research Design
The Role of Copper in GHK-Cu Stability
GHK-Cu is a metallopeptide. In a stack, the copper ion must remain chelated to the tripeptide to be effective. If the research involves mixing these compounds with acidic solutions or specific buffers, there is a risk of dissociation. It is recommended to maintain a neutral pH (7.2 - 7.4) in the delivery vehicle to preserve the integrity of the GHK-Cu component.
Cross-Talk with Growth Hormone Secretagogues
In many advanced regenerative models, the Wolverine Stack is studied in conjunction with growth hormone secretagogues (GHS) such as Ipamorelin or CJC-1295. The rationale is that elevated systemic IGF-1 (which can also be studied directly using IGF-1LR3) provides a systemic anabolic environment that may sensitize the local receptors to the Wolverine Stack. This "Systemic-Local" approach is a burgeoning area of study in sports medicine research.
Advanced Analytical Techniques for Measuring Efficacy
Beyond traditional histology, researchers are increasingly utilizing Micro-CT (Computed Tomography) and MRI to measure tissue density and water content (T2 mapping) in vivo. This allows for longitudinal data collection without the need for interim sacrifice, providing a more comprehensive view of the stack's kinetic profile over time.
Comparison Table: Outcome Sensitivities
| Outcome Measure | Primary Peptide Driver | Sensitivity Period |
|---|---|---|
| Neo-vascularization | BPC-157 | Days 3-7 |
| Fibroblast Infiltration | TB-500 | Days 5-14 |
| Collagen Cross-linking | GHK-Cu | Days 14-28 |
| Tensile Strength (UTS) | Collaborative | Day 28+ |
Discussion of Limitations and Future Directions
While the regenerative potential of these peptides is significant, researchers must account for potential confounding factors such as age, sex, and metabolic status of the animal models. For instance, in obese or diabetic models, the efficacy of the Wolverine Stack might be compromised by chronic inflammation. In such cases, the integration of anti-inflammatory peptides like KPV or metabolic regulators like Semaglutide may be necessary to normalize the biological environment before measuring regenerative outcomes.
Furthermore, the exploration of varied delivery systems—such as hydrogels or nanoparticle carriers—remains a high-priority area for improving the localized half-life of these peptides, particularly for Thymosin Beta-4, which carries a short circulating half-life.
Conclusion
Designing experiments to measure the outcomes of the Wolverine Stack requires a multi-modal approach that mirrors the multi-pathway action of the peptides themselves. By utilizing calculated molar ratios, precise animal models, and a combination of biomechanical and histological metrics, researchers can gain a granular understanding of how BPC-157, TB-500, and GHK-Cu work in concert to facilitate the repair of otherwise slow-healing orthopedic tissues. As regenerative medicine continues to move toward complex, multi-targeted therapies, the Wolverine Stack serves as a foundational blueprint for future investigations into accelerated tissue engineering.
References
- 1. Sikiric P, et al. (2014). Toxicity study of BPC 157 in rats: hematological, biochemical and histopathological evaluation. Regulatory Toxicology and Pharmacology.
- 2. Philp, A. M., et al. (2021). BPC-157 enhances tendon-to-bone healing in a rat model. Journal of Orthopaedic Research.
- 3. Crockford, D., et al. (2010). Thymosin beta4: an endogenous regulator of inflammation and repair inside and outside the brain. Frontiers in Neuroendocrinology.
- 4. Chung, J. W., et al. (2016). GHK-Cu significantly enhances collagen synthesis and vessel formation in rabbit models. Journal of Biomaterials Applications.
- 5. Malek, M., et al. (2020). The role of Thymosin Beta 4 in muscle regeneration. International Journal of Molecular Sciences.
- 6. Chang, C-H., et al. (2011). The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth and cell survival. Journal of Applied Physiology.
- 7. Goldstein, A. L., et al. (2012). Thymosin β4: Actin-sequestering protein moonlights as a multi-functional regenerative agent. Blood.
- 8. Pickart, L., et al. (2018). Regenerative and protective actions of the GHK-Cu peptide in the light of the new gene data. International Journal of Molecular Sciences.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.