Comparative Affinity: Evaluating Retatrutide in the Context of Modern Incretin Science
Explore the scientific differences between Retatrutide, Tirzepatide, and Semaglutide. This comprehensive guide analyzes triple-receptor affinity, metabolic mechanisms, and research applications for next-generation incretin mimetics.
 Retatrutide with increasing receptor targets and efficacy" style="max-width: 100%; height: auto; border-radius: 8px;" />
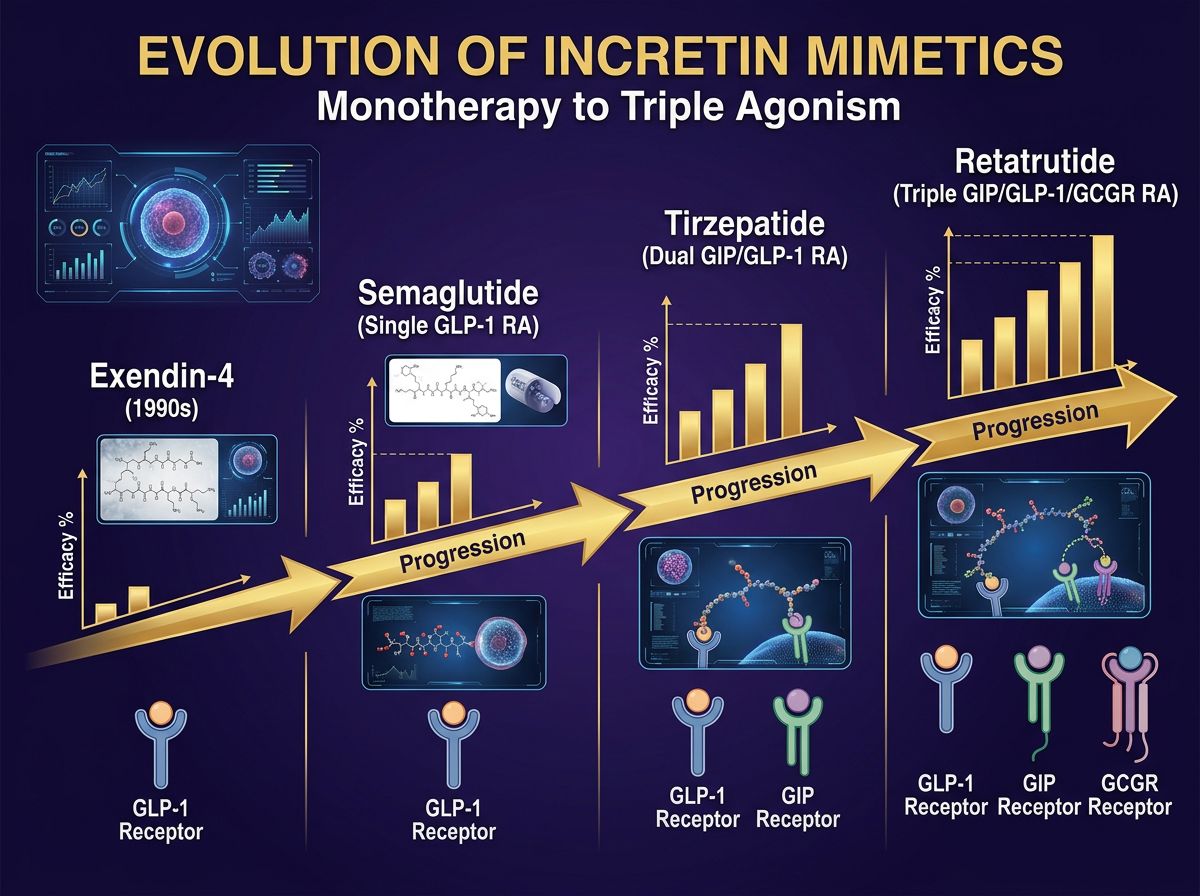
Retatrutide with increasing receptor targets and efficacy" style="max-width: 100%; height: auto; border-radius: 8px;" />The Evolution of Incretin Mimetics: From Monotherapy to Triple Agonism
In the landscape of pharmacological research, few fields have moved as rapidly as incretin science. What began with the isolation of Exendin-4 has evolved into a sophisticated era of multi-receptor poly-agonists. Researchers selecting compounds must now navigate a complex hierarchy of affinity, ranging from the highly specific Semaglutide to the dual-agonist Tirzepatide, and now the experimental frontier of triple-agonists like Retatrutide.
The core challenge for investigators lies in understanding the nuance of receptor binding. While high affinity is often desired, the synergy between independent metabolic pathways—namely GLP-1 (Glucagon-like peptide-1), GIP (Glucose-dependent insulinotropic polypeptide), and GCGR (Glucagon receptor)—determines the ultimate metabolic outcome. Retatrutide represents the current pinnacle of this "modular" metabolic engineering, but its utility must be measured against the established benchmarks of dual-agonists.
Retatrutide: The Triple-Agonist Mechanism
Retatrutide (LY3437943) is an oxyntomodulin-based peptide that has been engineered to possess potent activity at three distinct receptors. Its primary structure is a 39-amino-acid peptide with a C18 fatty diacid moiety, designed to extend its half-life and allow for prolonged receptor recruitment. Understanding Retatrutide requires a breakdown of its tripartite action:
1. GIP Receptor Agonism
Unlike early GLP-1 monotherapies, Retatrutide places a heavy emphasis on GIP receptor (GIPR) activity. GIP is increasingly recognized as a "buffer" for metabolic health, enhancing insulin secretion in a glucose-dependent manner while potentially mitigating the nausea often associated with GLP-1 receptor activation. In the context of Retatrutide, GIPR affinity is balanced to optimize lipid metabolism and energy expenditure.
2. GLP-1 Receptor Agonism
The GLP-1 receptor (GLP-1R) remains the cornerstone of weight and glucose regulation. Agonism here suppresses appetite by delaying gastric emptying and modulating satiety centers in the hypothalamus. Retatrutide’s GLP-1R affinity is designed to be complementary to its GIP and Glucagon actions, ensuring that the anorectic effect remains potent without overwhelming the organism's tolerance.
3. Glucagon Receptor Agonism (GCGR)
The inclusion of the Glucagon receptor is what distinguishes Retatrutide from Tirzepatide. Historically, glucagon was viewed solely as a hyperglycemic hormone. However, modern research indicates that GCGR agonism increases thermogenesis and energy expenditure through the activation of futile cycles in the liver and potentially the recruitment of brown adipose tissue. By combining GCGR with GLP-1 and GIP, Retatrutide aims to "burn" calories while simultaneously "reducing" calorie intake.
Comparative Receptor Affinity: Retatrutide vs. Tirzepatide
For researchers, the choice between Tirzepatide and Retatrutide hinges on the specific metabolic pathways being studied. While Tirzepatide is a GIP/GLP-1 dual agonist, Retatrutide adds the Glucagon component. However, the affinity ratios are not identical.
| Feature | Tirzepatide | Retatrutide |
|---|---|---|
| Primary Type | Dual Agonist (GIP/GLP-1) | Triple Agonist (GIP/GLP-1/GCG) |
| GIP Affinity | High (Potency comparable to native GIP) | High (Potency optimized for GIPR) |
| GLP-1 Affinity | Moderate (Weaker than native GLP-1) | Moderate/High |
| Glucagon Affinity | None | Potent (High GCGR activation) |
| Half-Life | ~5 Days | ~6 Days |
| Primary Outcome | Glucose control + Significant Weight Loss | Aggressive Weight Loss + Energy Expenditure |
In comparative studies, Tirzepatide has shown a "GIP-heavy" bias, which many researchers suspect is the reason for its superior tolerability compared to high-dose GLP-1 monotherapies. Retatrutide maintains this GIP potency but introduces a "Glucagon-biased" metabolic acceleration. This makes Retatrutide particularly interesting for research focusing on Non-Alcoholic Fatty Liver Disease (NAFLD) and metabolic-associated steatotic liver disease (MASLD), where Glucagon's role in hepatic lipid oxidation is paramount.
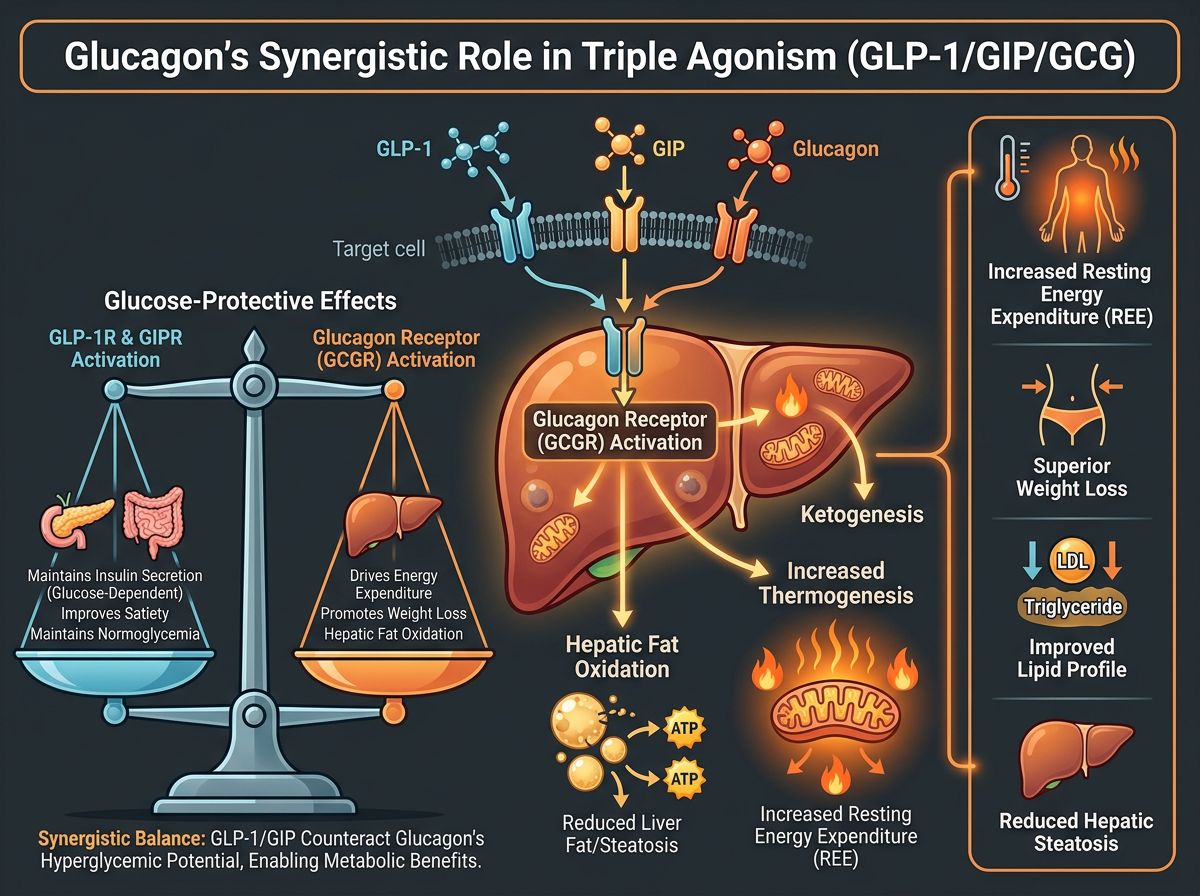
The Synergistic Role of Glucagon Agonism
The major physiological leap represented by Retatrutide is the controlled use of Glucagon. Researchers have long feared that GCGR agonism would lead to hyperglycemia. However, the concurrent activation of the GLP-1 and GIP receptors creates a "glucose-protective" environment. The insulinotropic effects of GIPR and GLP-1R agonism effectively neutralize the glucose-elevating potential of GCGR activation. What remains is the metabolic benefit: increased resting energy expenditure (REE).
This "Triple Threat" approach is often compared to a "push-pull" system:
- The Push (GLP-1/GIP): Pushing energy intake down through satiety and appetite suppression.
- The Pull (Glucagon): Pulling energy out of storage via increased thermogenesis and hepatic fat oxidation.
Research Applications and Comparison to Other Metabolic Modulators
While Retatrutide is currently a leader in triple-agonism research, it is often studied alongside other metabolic peptides available at Alpha Carbon Labs. Depending on the research objective, other compounds may be more appropriate.
AOD9604 and Adipotide
For studies focused strictly on lipolysis without the insulin-modulating effects of incretins, AOD9604 offers a focused look at the C-terminus of Human Growth Hormone, which stimulates fat breakdown without affecting blood sugar. Conversely, Adipotide works via a completely different mechanism—targeting the vascularity of white adipose tissue. Retatrutide differs from these by working through the central nervous system and hormonal signaling pathways rather than direct adipocyte lysis.
Energy Expenditure: MOTS-c and 5-Amino-1MQ
Researchers interested in the energy-expenditure aspect of Retatrutide may also find MOTS-c and 5-amino-1mq relevant. MOTS-c is a mitochondrial-derived peptide that regulates metabolic homeostasis, while 5-amino-1mq inhibits NNMT to improve cellular energy metabolism. While Retatrutide increases energy expenditure through hormonal signaling, these compounds act at the mitochondrial and enzymatic levels, respectively.
Therapeutic Benchmarking: Semaglutide vs. Tirzepatide vs. Retatrutide
When evaluating these compounds for research purposes, it is essential to review the clinical weight loss and metabolic markers observed in Phase II and III trials. Although Retatrutide is still in the research phase, initial data suggests a steeper efficacy curve than its predecessors.
- Semaglutide: Demonstrated approximately 15% body weight reduction in 68-week trials.
- Tirzepatide: Demonstrated approximately 21-22% body weight reduction in 72-week trials.
- Retatrutide: Phase II data showed up to 24.2% body weight reduction in only 48 weeks.
The speed and depth of weight loss with Retatrutide suggest that triple-receptor recruitment is more than just an additive effect; it appears to be multiplicative. For researchers, this necessitates a careful consideration of safety protocols, especially regarding heart rate. GCGR agonism has been linked to transient increases in heart rate, a variable that must be closely monitored in any experimental model involving Retatrutide.
Safety and Tolerability in Research Models
All incretin mimetics share a similar side-effect profile, primarily gastrointestinal in nature. Nausea, vomiting, and diarrhea are common during the dose-escalation phases of Semaglutide and Tirzepatide research. Retatrutide is no exception. However, researchers have noted that the "GIP-heavy" profile of Retatrutide and Tirzepatide may actually blunt some of the more severe emetic effects seen with pure GLP-1 agonists.
Specific considerations for Retatrutide research include:
- Cardiac Monitoring: Due to Glucagon’s effect on the heart, monitoring heart rate and rhythm is vital in animal or cellular models.
- Glucose Stability: While early data is promising, the balance between Glucagon and Insulin secretion must be a focal point of any metabolic study.
- LBM Conservation: A significant concern with rapid weight loss is the loss of Lean Body Mass (LBM). Researchers may combine Retatrutide studies with myostatin inhibitors like ACE-031 or growth hormone secretagogues like Ipamorelin to investigate strategies for muscle preservation during aggressive fat loss.
Selecting the Right Peptide for Your Study
At Alpha Carbon Labs, we provide high-purity peptides for laboratory use. Choosing between these molecules depends on your study's primary endpoint:
When to choose Retatrutide:
- Focus is on maximum weight loss efficacy.
- Researching the metabolic effects of triple-hormone agonism.
- Investigating hepatic lipid metabolism or NAFLD/MASLD models.
- Studying the interaction between glucagon and thermogenesis.
When to choose Tirzepatide:
- Seeking a balance between high efficacy and established safety data.
- Researching the synergistic effects of GIP and GLP-1 without Glucagon interference.
- Studying glycemic control as a primary endpoint.
When to choose Semaglutide:
- Requiring a "gold standard" GLP-1R agonist for control groups.
- Researching pure GLP-1 signaling without GIP or GCGR cross-talk.
Quality Assurance in Peptide Research
Regardless of the molecule chosen, the validity of research findings depends entirely on the quality of the compound. At Alpha Carbon Labs, we emphasize rigorous testing protocols. For researchers, ensuring that COA Documents are accessible and that Quality Control standards are met is non-negotiable. Our Retatrutide and Tirzepatide represent the highest levels of purity, ensuring that the receptor affinity observed in your laboratory is a result of the peptide itself and not contaminants.
Conclusion: The Paradox of Agonism
The transition from Semaglutide to Tirzepatide and finally to Retatrutide marks a shift in how we view metabolic regulation. We have moved from simply stimulating a single pathway to orchestrating a symphony of hormonal signals. Retatrutide stands at the forefront of this evolution, offering the most potent metabolic tool currently available to the research community. By understanding the specific receptor affinities of these compounds, researchers can better design studies that push the boundaries of metabolic science.
For those looking to procure these advanced research tools, our catalog includes Tirzepatide, Semaglutide, and the emerging Retatrutide, all backed by our commitment to scientific excellence.
References
- 1. Coskun, T., et al. (2023). 'Retatrutide (LY3437943), a novel triple glucagon, guanidine-dependent insulinotropic polypeptide, and glucagon-like peptide-1 receptor agonist.' The Lancet.
- 2. Jastreboff, A. M., et al. (2023). 'Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.' New England Journal of Medicine.
- 3. Nauck, M. A., & Quast, D. R. (2021). 'Glucagon-like peptide 1 receptor agonists in the treatment of type 2 diabetes—state-of-the-art.' Molecular Metabolism.
- 4. Frias, J. P., et al. (2021). 'Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes.' New England Journal of Medicine.
- 5. Müller, T. D., et al. (2017). 'The visceral-adipose-tissue-homing peptide: Glucagon receptor signaling.' Nature Reviews Endocrinology.
- 6. Knerr, P. J., et al. (2022). 'Next generation of incretin-based peptide therapeutics.' Journal of Peptide Science.
- 7. Smith, N. K., et al. (2022). 'The role of glucagon in energy expenditure and weight loss.' Cell Metabolism.
- 8. Drucker, D. J. (2023). 'GLP-1 receptor agonists: multi-agonists and the future.' Journal of Clinical Investigation.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.