Beyond the Pancreas: Investigating Semaglutide’s Neuroprotective Mechanisms in CNS Research Models
Explore the advanced neuroprotective mechanisms of Semaglutide in CNS research models, focusing on neuroinflammation, mitochondrial health, and its potential in Alzheimer's and Parkinson's disease studies.
Introduction to Semaglutide in Neuroscience Research
While Semaglutide has achieved global recognition for its transformative impact on metabolic health and glycemic regulation, the research community is increasingly shifting its focus toward the central nervous system (CNS). The rationale for investigating glucagon-like peptide-1 receptor agonists (GLP-1 RAs) in neurobiology stems from the widespread expression of the GLP-1 receptor (GLP-1R) beyond the pancreas, particularly in regions of the brain associated with cognition, neuroinflammation, and neuronal survival.
Researchers who acquire Semaglutide for laboratory use are often exploring its potential as a disease-modifying agent for neurodegenerative conditions such as Alzheimer’s disease (AD) and Parkinson’s disease (PD). This article provides a technical dive into the molecular pathways, pharmacokinetic properties, and cellular mechanisms that position Semaglutide as a primary candidate for CNS research.
The Molecular Architecture of Semaglutide
To understand its neuroprotective efficacy, one must first examine its structure. Semaglutide is a synthetic GLP-1 analog with roughly 94% sequence homology to human GLP-1. Key modifications include a C18 fatty diacid chain attached via a spacer to Lysine at position 26, and an Alpha-aminoisobutyric acid (Aib) substitution at position 8 to prevent degradation by dipeptidyl peptidase-4 (DPP-4).
Quality in synthesis is paramount for these sensitive investigations. Researchers often consult COA documents and verify quality control protocols to ensure the purity of the peptide, as impurities can confound the observation of nuanced neurobiological signaling pathways.
Brain Penetration and Pharmacokinetics
A critical question in CNS research is whether peripherally administered Semaglutide can cross the blood-brain barrier (BBB). Research indicates that while the peptide remains relatively large, it accesses the brain via several mechanisms:
- SFO and AP Entry: Subtle entry through circumventricular organs like the subfornical organ (SFO) and area postrema (AP), which lack a traditional BBB.
- Active Transport: Evidence suggests saturable transport mechanisms across the BBB.
- Enhanced Stability: The prolonged half-life of approximately 165 hours allows for sustained therapeutic concentrations, facilitating gradual accumulation in the brain parenchyma compared to shorter-acting analogs.
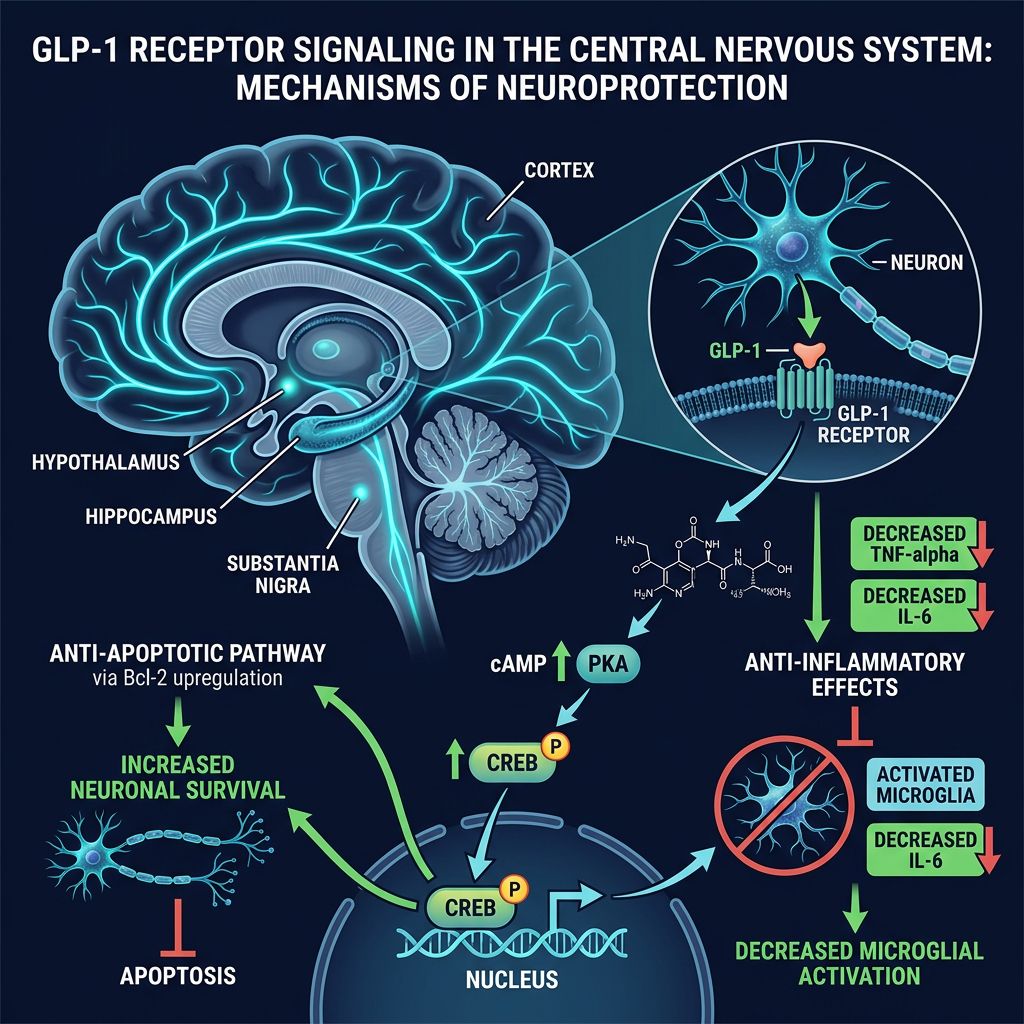
Mechanism of Action: GLP-1R Signaling in the CNS
The neuroprotective effects of Semaglutide are mediated through the activation of GLP-1Rs on neurons and glial cells (astrocytes and microglia). The downstream effects are complex and multi-modal.
1. Modulation of Neuroinflammation
Chronic neuroinflammation, characterized by overactive microglia, is a hallmark of neurodegeneration. Semaglutide has been shown to shift microglial polarization from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype. By reducing the secretion of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, Semaglutide preserves the integrity of the neural environment.
2. Mitochondrial Protection and Bioenergetics
Neurodegenerative diseases often involve mitochondrial dysfunction and oxidative stress. Semaglutide activates the PI3K/Akt pathway and the cAMP/PKA signaling cascade, which leads to:
- Increased expression of Bcl-2 (anti-apoptotic).
- Reduced expression of Bax (pro-apoptotic).
- Maintenance of mitochondrial membrane potential.
- Increased levels of antioxidants like Superoxide Dismutase (SOD).
In models of metabolic-induced cognitive decline, researchers may also explore adjunctive therapies such as SS-31 for targeted mitochondrial stability or MOTS-c for exercise-mimetic metabolic signaling.
3. Proteostatic Regulation: Clearing Beta-Amyloid and Tau
In Alzheimer’s models, GLP-1R activation has been linked to the reduction of amyloid-beta (Aβ) plaque accumulation and the prevention of tau hyperphosphorylation. This is hypothesized to occur through the activation of autophagy and the inhibition of glycogen synthase kinase 3 beta (GSK3β), a kinase responsible for tau phosphorylation.
Comparative Analysis: Semaglutide vs. Other Incretin Mimetics
The evolution of metabolic research has introduced multi-receptor agonists. While Semaglutide is a potent selective GLP-1 RA, other peptides like Tirzepatide (a GLP-1/GIP dual agonist) and the emerging Retatrutide (triple agonist) are now being scrutinized for their potentially synergistic neuroprotective effects.
| Peptide | Target Receptors | Primary CNS Research Focus |
|---|---|---|
| Semaglutide | GLP-1R | Neuroinflammation, Amyloid Clearance, PD Models |
| Tirzepatide | GLP-1R, GIP-R | Neuro-metabolic crosstalk, Lipid metabolism in CNS |
| Retatrutide | GLP-1R, GIP-R, GCGR | Energetic homeostasis, severe neuro-metabolic models |
| Cagrilintide | Amylin Receptor | Satiety signaling, potential synergy in weight-centric CNS models |
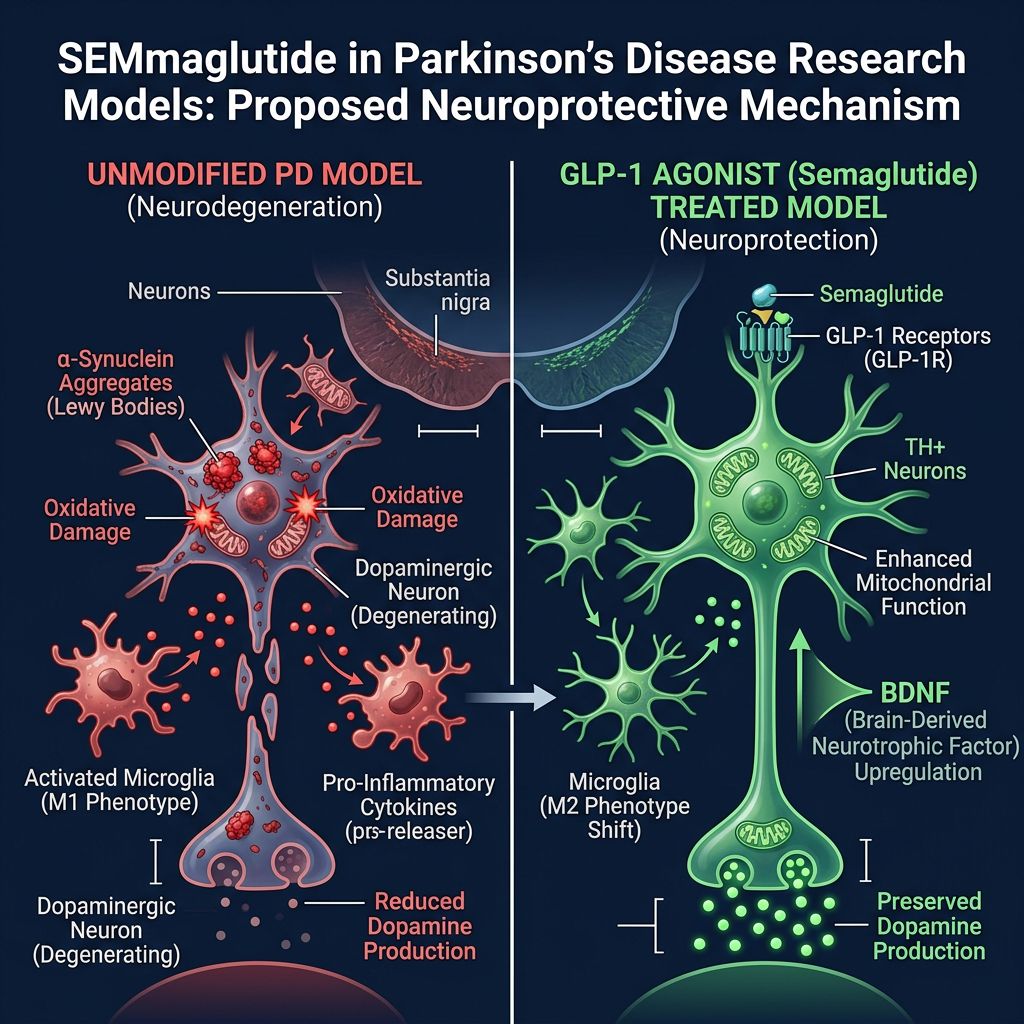
Experimental Evidence in Models of Parkinson’s Disease
Research in Parkinson's disease models focuses heavily on the protection of dopaminergic neurons in the substantia nigra pars compacta. Semaglutide has demonstrated the ability to prevent the loss of tyrosine hydroxylase-positive neurons in MPTP-induced rodent models. Furthermore, it improves motor coordination and reduces the aggregation of alpha-synuclein, the protein responsible for Lewy bodies.
Synergistic Research Opportunities
In complex CNS research, Semaglutide is rarely studied in isolation. Researchers may combine it with other neurogenic or neuro-reparative peptides to investigate recovery. For example, the use of Dihexa for synaptic connectivity or Cerebrolysin for global neurotrophic support represents the cutting edge of regenerative neuroscience.
Semaglutide in Stroke and Traumatic Brain Injury (TBI)
The anti-apoptotic and anti-inflammatory properties of Semaglutide extend to acute neurological injuries. In models of ischemic stroke, pre-treatment or early post-ischemic administration has been shown to reduce infarct volume and improve functional outcomes. This is largely attributed to the reduction of glutamate-induced excitotoxicity and the stabilization of the BBB.
For researchers studying peripheral nerve injury alongside CNS models, the combination of Semaglutide with ARA-290 (for neuropathic pain and erythropoietin receptor modulation) or BPC-157 (for tissue repair) offers a comprehensive approach to neural recovery.
Future Directions: Investigating Satiety and Addiction Pathways
Beyond neurodegeneration, Semaglutide is a valuable tool for behavioral neuroscience. By acting on the hypothalamus and the reward systems (ventral tegmental area and nucleus accumbens), it provides a mechanism to study addictive behaviors, cravings, and the psychological aspects of satiety. Researchers are investigating its role in reducing the rewarding effects of ethanol and opioids, suggesting a broader clinical application in addiction medicine.
Technical Considerations for Researchers
When conducting research on GLP-1 RAs like Semaglutide, careful attention must be paid to reconstitution and storage to prevent denaturation. Following strict peptide synthesis standards is essential for reproducibility. Research models should account for the dose-response relationship, as excessively high doses in murine models can lead to profound anorexia, which may serve as a confounding factor in neurobehavioral assays.
Biological Half-Life and Dosing Consistency
One of the primary advantages of Semaglutide in a lab setting is its extended half-life. Unlike Liraglutide, which requires daily administration in many animal models, Semaglutide’s pharmacokinetic profile allows for less frequent dosing, reducing stress on research subjects and maintaining a more stable plasma concentration—critical when observing chronic disease progression such as cognitive decline.
Concluding Summary
The investigation of Semaglutide in CNS research models represents a paradigm shift from treating metabolic symptoms to addressing the root causes of neurodegeneration. Its multifaceted ability to attenuate neuroinflammation, preserve mitochondrial function, and clear toxic protein aggregates makes it a cornerstone of contemporary peptide research.
As research continues to evolve with peptides like Mazdutide or the amylin-agonist Cagrilintide, Semaglutide remains the gold standard for validating the neuroprotective potential of the incretin hormone system. For investigators, the journey beyond the pancreas is just beginning, opening new doors to potential therapies for some of the most challenging disorders of the brain.
References
- 1. Hölscher C. (2022). Protective effects of GLP-1 mimetics in Alzheimer's disease: intracellular signaling and cytoprotective effects. Frontiers in Aging Neuroscience.
- 2. Athelogou M., et al. (2021). Semaglutide, a glucagon-like peptide-1 receptor agonist with protective effects in the central nervous system. CNS Drugs.
- 3. Grieco M., et al. (2019). Glucagon-Like Peptide-1 receptor agonist as a multi-target drug for neurodegenerative diseases. International Journal of Molecular Sciences.
- 4. Salameh T.S., et al. (2020). Blood-brain barrier transport of glucagon-like peptide-1 receptor agonists. Journal of Cerebral Blood Flow & Metabolism.
- 5. Kim D.S., et al. (2017). GLP-1 receptor activation and neuroprotection in ischemia and neurodegeneration. Journal of Neurochemistry.
- 6. McLean B.A., et al. (2021). Revisiting the role of GLP-1 receptor signaling in the brain. Molecular Metabolism.
- 7. Zhang L., et al. (2018). Neuroprotective effects of the GLP-1 analog liraglutide and semaglutide in Parkinson's disease models. Scientific Reports.
- 8. Drucker D.J. (2021). GLP-1 receptor agonists: non-metabolic actions and clinical implications. Cell Metabolism.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.