Beyond Dual Agonism: Comparing the Pharmacological Profiles of Retatrutide and Tirzepatide
A deep dive into the molecular differences between Retatrutide and Tirzepatide, exploring how triple-receptor agonism (GLP-1/GIP/Glucagon) compares to dual-incretin pathways in metabolic research.
Introduction: Evolution of the Incretin Mimetic
In the landscape of metabolic research, the progression from mono-agonist peptides to multi-receptor ligands represents a paradigm shift in how researchers approach obesity, type 2 diabetes (T2D), and metabolic syndrome. While Semaglutide established the potency of Glucagon-Like Peptide-1 (GLP-1) receptor agonism, the subsequent development of dual agonists like Tirzepatide introduced the synergistic potential of Gastric Inhibitory Polypeptide (GIP) integration. Now, the research community is looking toward "triple-agonism" with Retatrutide, a peptide that adds Glucagon receptor (GCGR) activation to the GLP-1/GIP framework.
To understand the efficacy of these compounds, it is essential to analyze the molecular pharmacology, pharmacokinetic (PK) profiles, and the distinct signaling pathways they engage. This article explores the nuanced differences between the dual-agonist Tirzepatide and the triple-agonist Retatrutide, providing researchers with a technical foundation for investigating these potent secretagogues.
Molecular Architecture and Receptor Affinity
Tirzepatide: The Unbiased Dual Agonist
Tirzepatide is a 39-amino acid synthetic peptide based on the native GIP sequence. Its primary innovation lies in its "twincretin" nature—acting as a full agonist at the GIP receptor (GIPR) and a potent agonist at the GLP-1 receptor (GLP-1R). At Alpha Carbon Labs, we observe that the structural integrity of these long-chain peptides is paramount for maintaining this balanced affinity.
The peptide includes a C20 fatty diacid moiety attached via a linker to a lysine residue at position 20. This modification allows for reversible albumin binding, resulting in a prolonged half-life of approximately 5 days. Crucially, Tirzepatide’s affinity for the GIPR is equal to native GIP, whereas its GLP-1R affinity is approximately 20-fold lower than native GLP-1. This "imbalanced" agonism is intentional, designed to maximize the insulinotropic effects of GIP while minimizing the gastrointestinal side effects often associated with excessive GLP-1R activation.
Retatrutide: The Triple Agonist (GIP/GLP-1/Glucagon)
Retatrutide (LY3437943) represents a further evolution. It is a 39-amino acid backbone engineered to target three distinct receptors: GIPR, GLP-1R, and GCGR. The inclusion of glucagon agonism is a bold departure from previous strategies. Traditionally, glucagon was viewed as an antagonist to insulin, raising blood glucose. However, research now demonstrates that when co-administered with GLP-1 and GIP, glucagon agonism increases energy expenditure and promotes hepatic lipid oxidation without the glycemic penalty.
Retatrutide’s potency profile is highly specific:
- GIPR: High potency (comparable to Tirzepatide).
- GLP-1R: Moderate potency.
- GCGR: Significant potency, facilitating thermogenesis.
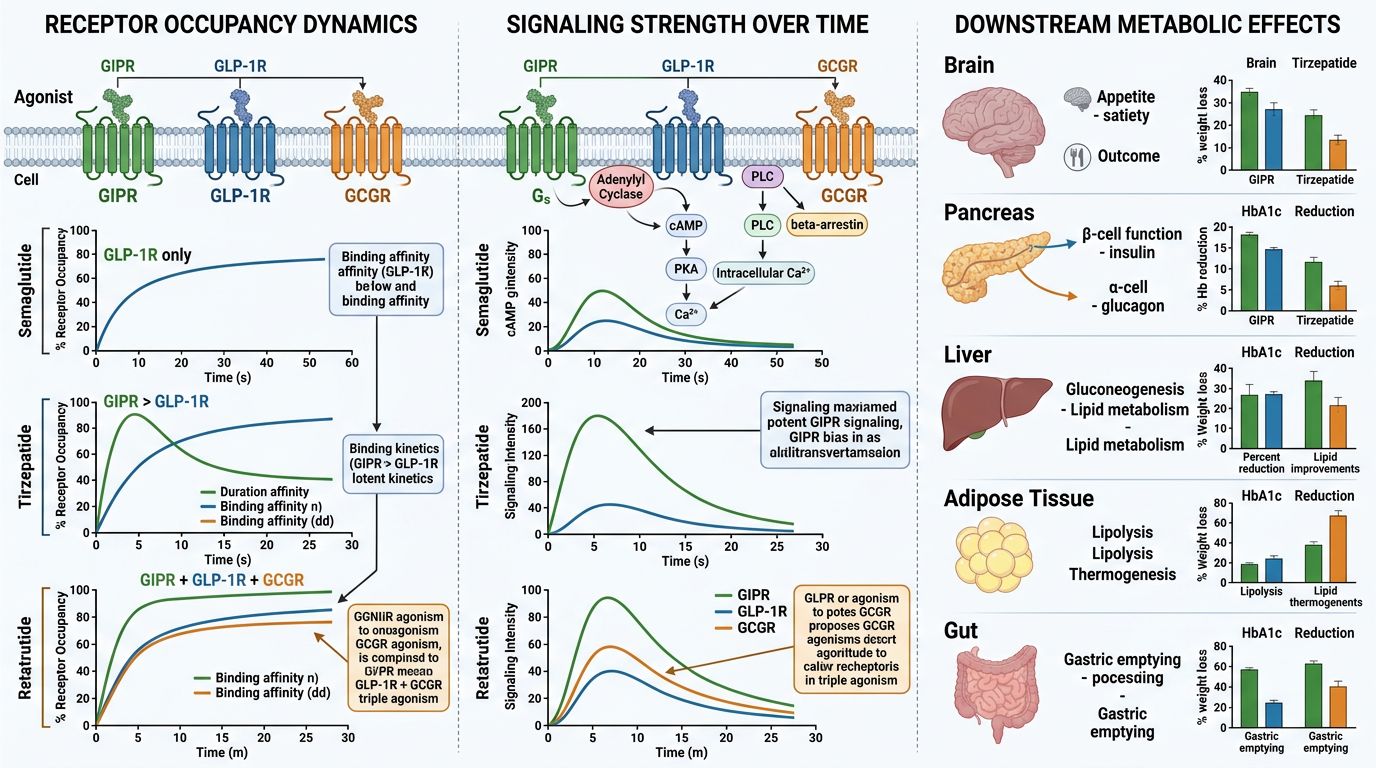
Comparative Pharmacological Mechanisms
1. Synergy of GIP and GLP-1 (The Tirzepatide Baseline)
The primary mechanism of Tirzepatide involves the potentiation of glucose-dependent insulin secretion (GSIS). While GLP-1 acts on pancreatic beta-cells to stimulate insulin, GIP adds a secondary layer of glycemic control. Perhaps more importantly, GIP receptors in the central nervous system (CNS) and adipose tissue are thought to buffer the "nausea" signals of GLP-1 while enhancing insulin sensitivity in white adipose tissue. This dual-action allows for higher dosing and greater weight loss compared to pure GLP-1 agonists like Semaglutide.
2. The Addition of Glucagon (The Retatrutide Advantage)
Retatrutide utilizes the GLP-1 and GIP foundation but overlays a glucagon-mediated metabolic boost. Glucagon receptors are primarily located in the liver. Activation leads to:
- Increased Energy Expenditure: Higher basal metabolic rate via mitochondrial decoupling and thermogenesis.
- Direct Lipolysis: Promotion of breakdown of stored fats into free fatty acids for oxidation.
- Hepatic Fat Clearance: Significant reduction in liver fat content, making it a focus for research into Non-Alcoholic Fatty Liver Disease (NAFLD).
Table 1: Pharmacological Profiles Comparison
| Feature | Tirzepatide | Retatrutide |
|---|---|---|
| Receptor Targets | GIPR, GLP-1R | GIPR, GLP-1R, GCGR |
| Backbone Origin | GIP-based | GIP/Glucagon-based chimeric |
| Weight Loss Induction | High (~20% in clinical trials) | Very High (~24%+ in clinical trials) |
| Liver Fat Reduction | Significant | Exceptional (up to 80% reduction) |
| Core Mechanism | Appetite suppression + Insulin sensitivity | Appetite suppression + Thermogenesis + Lipolysis |
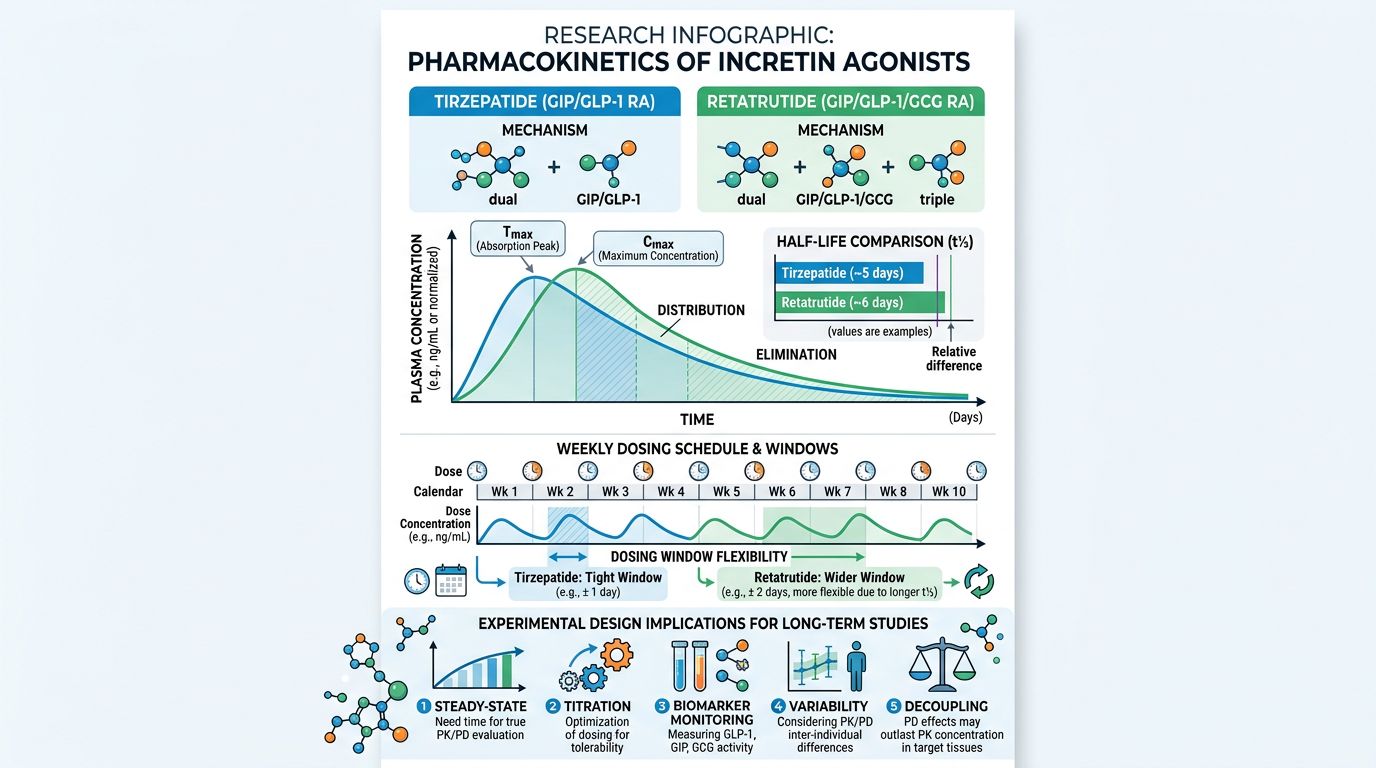
Pharmacokinetics and Research Applications
For researchers, the pharmacokinetic profile dictates the study design. Both Tirzepatide and Retatrutide are designed for a 168-hour (one week) duration of action. However, their metabolic clearance rates and steady-state concentrations vary slightly due to the specific acylation patterns used in peptide synthesis.
Steady-State Dynamics
Tirzepatide reaches steady state after approximately 4 weeks of consistent administration. Its PK profile is linear, meaning the concentration in the research subject increases proportionally with the dose. Retatrutide demonstrates similar long-acting properties, but the triple-agonism introduces more complex pharmacodynamics. Because glucagon can transiently increase glucose output from the liver, the GIP and GLP-1 components must "overwhelms" this effect to ensure a net Reduction in blood glucose.
Research in Adiposity and Muscle Preservation
One of the critical areas of current research is the quality of weight loss. Pure GLP-1 agonists often cause a concurrent loss of lean muscle mass. By incorporating GIP (which may have bone and muscle-protective effects) and Glucagon (which favors fat oxidation), Tirzepatide and Retatrutide are being studied for their ability to shift the Rheology of weight loss toward a higher percentage of fat mass loss. Researchers investigating body composition often pair these with growth hormone secretagogues like Ipamorelin or Tesamorelin to investigate synergistic muscle preservation.
The Role of Mitochondrial Health: MOTS-c and SS-31
While Retatrutide increases energy expenditure, this process relies heavily on mitochondrial efficiency. There is growing interest in the research community regarding the co-administration of metabolic peptides with mitochondrial-specific peptides like MOTS-c or SS-31. By optimizing the mitochondrial electron transport chain with SS-31, the thermogenic effects of Retatrutide's glucagon component may theoretically be enhanced while minimizing oxidative stress.
Safety and Tolerability in Research Models
Tirzepatide has a well-documented safety profile, primarily characterized by dose-dependent gastrointestinal (GI) events (nausea, vomiting, diarrhea). Retatrutide, despite its triple action, showed a similar GI profile in Phase 2 trials, though a unique side effect was noted: a dose-dependent increase in heart rate. This is attributed to the presence of glucagon receptors in the sinoatrial node. Researchers monitoring cardiac markers or sympathetic nervous system activity should take particular note of this Retatrutide-specific pharmacodynamic effect.
To ensure research accuracy, utilizing high-purity compounds verified through stringent quality control and COA documentation is non-negotiable, as impurities can exacerbate immunogenic responses in peptide studies.
Conclusion: Selecting the Right Agonist for Research
The choice between Tirzepatide and Retatrutide depends on the specific metabolic pathway under investigation:
- Choose Tirzepatide for studies focusing on dual-incretin synergy, insulin sensitization, and established GLP-1/GIP signaling.
- Choose Retatrutide for studies exploring maximal weight loss, hepatic fat clearance, or the integration of thermogenic pathways via the glucagon receptor.
As we move beyond dual agonism, the potential for triple agonists to "reset" metabolic set points offers a new frontier in peptide science. Whether exploring the mitochondrial synergies with NAD+ or the tissue-repair properties of BPC-157 in the context of rapid weight loss, Alpha Carbon Labs remains committed to providing the highest purity materials for your investigational needs.
References
- 1. Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-217.
- 2. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. N Engl J Med. 2023;389(6):514-526.
- 3. Frias JP, et al. Efficacy and safety of retatrutide, a GIP, GLP-1, and glucagon receptor agonist, in adults with type 2 diabetes: a randomised, double-blind, placebo-controlled and active-controlled, parallel-group, phase 2 trial. Lancet. 2023;402(10401):529-544.
- 4. Müller TD, et al. The evolution of incretin hormones for the treatment of metabolic diseases. Gut. 2023;72:1335-1351.
- 5. Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
- 6. Nauck MA, et al. Incretin hormones: Their role in health and disease. Diabetes Obes Metab. 2018;20(S1):5-21.
- 7. Rosenstock J, et al. Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a 36-week, double-blind, randomised, phase 2 trial. Lancet. 2023;402(10401):529-544.
- 8. Baggio LL, Drucker DJ. Biology of Incretins: GLP-1 and GIP. Gastroenterology. 2007;132(6):2131-2157.
- 9. Heise T, et al. Tirzepatide: Pharmacokinetics and pharmacodynamics. Clin Pharmacokinet. 2022;61(12):1245-1259.
- 10. Knerr PJ, et al. Next-generation multi-receptor agonists for the treatment of obesity and type 2 diabetes. Nat Rev Endocrinol. 2024;20:12-28.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.