The Paradigm Shift: Transitioning Existing Metabolic Research Models to Retatrutide Protocols
Explore the transition from GLP-1 and dual-agonist models to the triple-agonist Retatrutide framework, focusing on the synergistic roles of GLP-1, GIP, and Glucagon receptors in metabolic research.
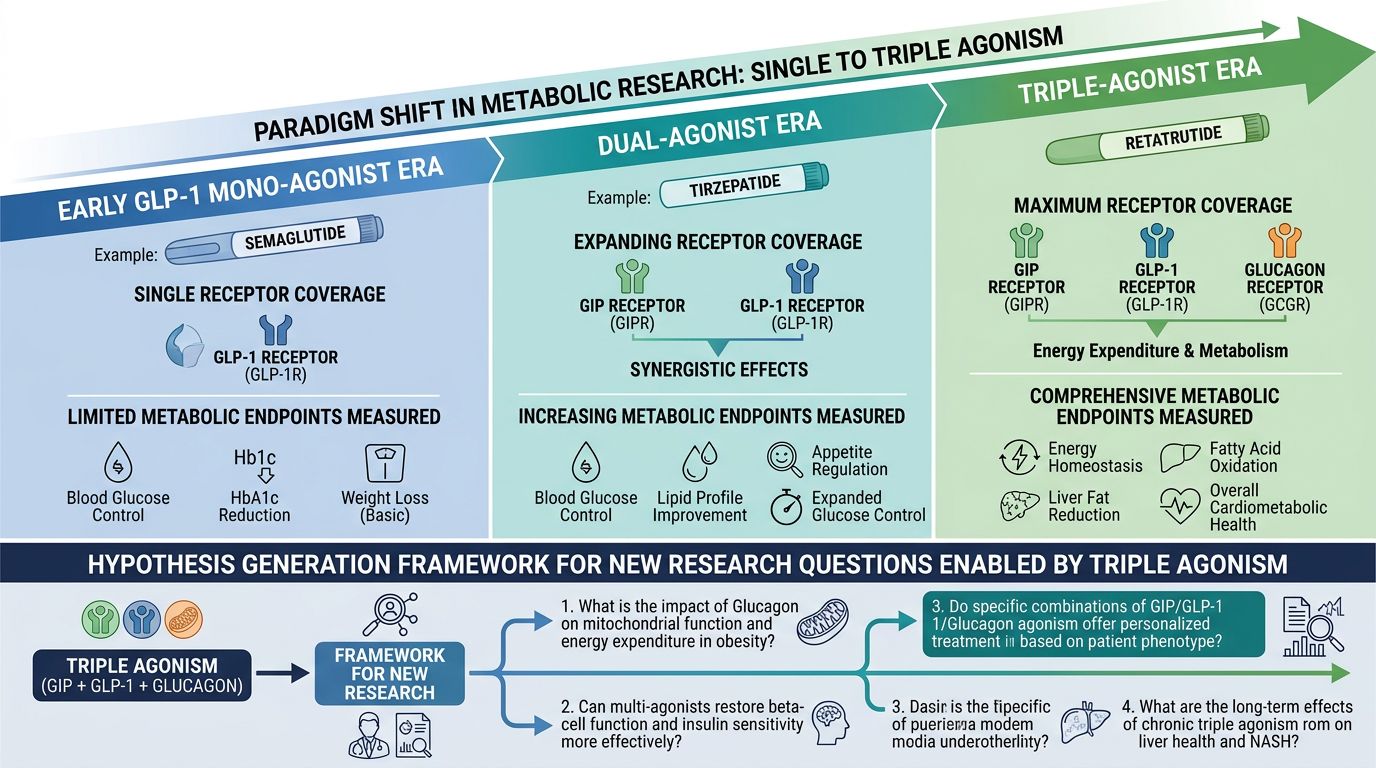
The landscape of metabolic research is currently undergoing its most significant evolution since the introduction of synthetic insulin. For years, researchers focused on the GLP-1 (Glucagon-Like Peptide-1) receptor as the primary lever for glycemic control and weight reduction. This era, defined by compounds like Semaglutide, provided a robust foundation for understanding incretin-based therapies. However, the subsequent emergence of dual-agonists like Tirzepatide introduced the synergistic potential of GIP (Glucose-dependent Insulinotropic Polypeptide). Today, we stand at the precipice of a new paradigm: the triple-agonist framework, lead by the investigational peptide Retatrutide.
Understanding the Triple-Agonist Framework
Retatrutide represents a biochemical culmination of two decades of research into nutrient-stimulated hormone (incretin) mimetics. While previous generations of metabolic research focused on one or two pathways, Retatrutide targets three distinct G-protein-coupled receptors (GPCRs):
- GLP-1 Receptor (GLP-1R): Primarily responsible for glucose-dependent insulin secretion, gastric emptying delay, and hypothalamic satiety signaling.
- GIP Receptor (GIPR): Acts syncretically with GLP-1 to enhance insulinotropism while potentially mitigating the nausea typically associated with pure GLP-1 agonism and improving lipid metabolism.
- Glucagon Receptor (GCGR): The "wildcard" in triple agonism, glucagon increases energy expenditure through thermogenesis and directly modulates hepatic lipid metabolism.
Transitioning from a Semaglutide or Tirzepatide model to a Retatrutide-centric protocol requires a fundamental shift in how researchers measure metabolic "success." It is no longer purely about caloric restriction and glycemic stability; it is now about the optimization of energy expenditure and hepatic clearance.
The Biochemical Architecture of Retatrutide
Retatrutide is a 39-amino acid backbone peptide, engineered with a specific fatty acid diacid moiety that allows for albumin binding and a prolonged half-life of approximately six days. This structural design ensures that research protocols can maintain steady-state concentrations with once-weekly administration, mirroring the pharmacokinetic profiles of earlier GLP-1 analogs but with vastly different pharmacodynamic outcomes.
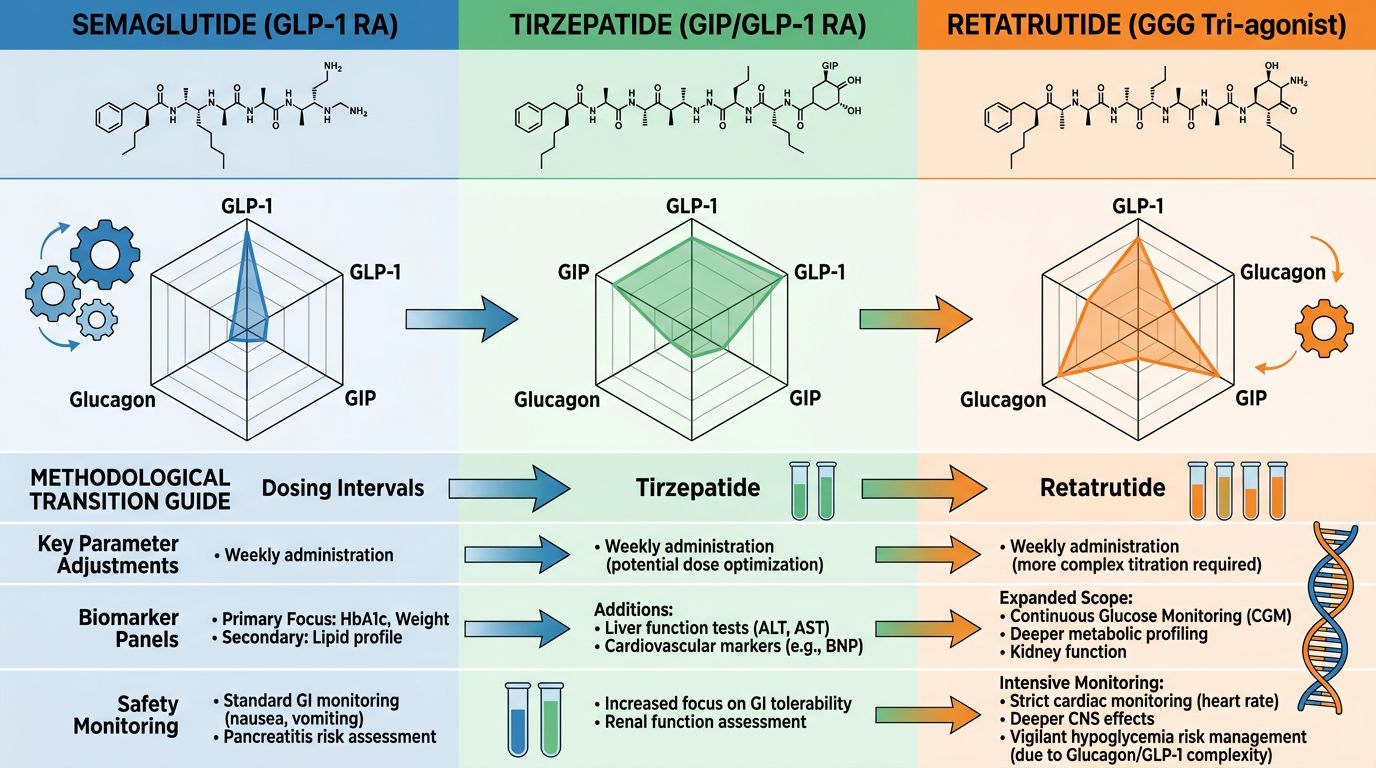
Comparative Analysis: Semaglutide vs. Tirzepatide vs. Retatrutide
To effectively transition research models, it is vital to understand the quantitative leap that Retatrutide offers over its predecessors. The following table summarizes the comparative receptor affinities and reported research outcomes in recent Phase II longitudinal studies.
| Feature | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Receptor Targets | GLP-1R | GLP-1R / GIPR | GLP-1R / GIPR / GCGR |
| Mechanism Type | Single Agonist | Dual Agonist | Triple Agonist |
| Weight Loss (24-48 weeks) | ~10-15% | ~15-20% | ~22-24%+ |
| Energy Expenditure | Minimal Change | Moderate Increase | Significant Increase (via GCGR) |
| Hepatic Fat Reduction | Secondary effect | Direct effect | Potent direct effect (up to 80%+) |
Transitioning the Research Protocol: Key Methodological Shifts
Researchers moving from existing models to Retatrutide must account for the introduction of the Glucagon receptor agonist component. This necessitates changes in data collection and monitoring.
1. Monitoring Energy Expenditure (EE)
In Semaglutide models, weight loss is primarily driven by appetite suppression (reduced energy intake). In Retatrutide models, energy expenditure becomes a critical variable. Researchers should utilize indirect calorimetry or metabolic cages in animal models to capture the thermogenic effects triggered by GCGR activation. Early data suggest that Retatrutide may prevent the metabolic slowdown often seen during significant caloric restriction.
2. Hepatic Lipid Profiling
Retatrutide has shown unprecedented efficacy in reducing intrahepatic fat content. Research models focusing on MASLD (Metabolic Dysfunction-Associated Steatotic Liver Disease) will find that Retatrutide necessitates more frequent imaging (MRI-PDFF) or histological analysis compared to Tirzepatide, as the clearance rates appear significantly more rapid.
3. Cardiovascular Parameters
Due to the glucagon component, a transient increase in heart rate has been noted in clinical research. When transitioning models, researchers should implement rigorous telemetry or ECG monitoring to differentiate between metabolic stimulation and potential sympathetic nervous system arousal.
Synthesizing New Hypotheses in Metabolic Research
The transition to Retatrutide allows researchers to investigate questions that were previously inaccessible with dual agonists. As we facilitate the supply of high-purity peptides through our advanced synthesis processes, we encourage the academic community to explore these emerging frontiers:
The "Uncoupling" of Caloric Restriction and Thermogenesis
Can Retatrutide maintain basal metabolic rate in the face of massive weight loss? Traditional GLP-1 models suffer from a "plateau effect" where the body adjusts its energy expenditure downward. Investigating whether the GCGR agonism in Retatrutide effectively "uncouples" this survival mechanism is a high-priority research area.
Synergy with Growth Hormone Secretagogues
There is growing interest in how triple-agonists interact with the GH axis. Research models combining Retatrutide with compounds like Tesamorelin or Ipamorelin may provide insights into body composition optimization—specifically the preservation of lean muscle mass during rapid adipose tissue oxidation.
Practical Implications for Laboratory Settings
Precision and purity are non-negotiable when shifting to triple-agonist research. Because Retatrutide acts on three different receptor pathways, any impurity in the peptide sequence could lead to off-target effects that skew data. Researchers are encouraged to review COA Documents and verify the lyophilization quality to ensure that the GLP-1/GIP/GCGR ratio remains stable throughout the study duration.
Reconstitution and Stability
Retatrutide, like other complex incretins, requires careful handling. It should be reconstituted with Bacteriostatic Water and maintained at stable temperatures (2-8°C). Given its long half-life, researchers must be careful to avoid "stacking" effects if the dosing frequency is moved from a weekly to a more frequent schedule in rodent models (which have faster metabolic clearances than humans).
Safety and Secondary Observations
While Retatrutide demonstrates superior efficacy in weight and glucose models, the safety profile requires nuance. The transition from Semaglutide models usually involves a "washout" period or a carefully titrated introduction to minimize gastrointestinal distress. In research environments, titrating the dose over a period of 4-8 weeks has been shown to improve the tolerability of the GLP-1 component, allowing the metabolic benefits of the GIP and Glucagon components to manifest without confounding adverse events.
For researchers specifically interested in muscle preservation, the concurrent use of IGF-1 LR3 or MGF in tandem with Retatrutide protocols is an emerging area of study to counter the catabolic potential of extreme weight loss.
Conclusion: The Future of Incremental Research
Retatrutide is not merely an "incremental" improvement over Tirzepatide; it is a fundamental shift toward multi-pathway metabolic reprogramming. By integrating glucagon receptor agonism, Retatrutide addresses the "other half" of the energy balance equation: expenditure. For the scientific community, transitioning to these protocols represents the next logical step in conquering the global obesity and T2DM crises.
At Alpha Carbon Labs, we remain committed to providing the highest grade research materials to facilitate this shift. We invite researchers to explore our full catalog of quality-tested metabolic peptides and join us in defining the next decade of endocrinology research.
References
- 1. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine. 2023.
- 2. Rosenstock J, et al. Retatrutide, a GIP, GLP-1, and glucagon receptor agonist, for type 2 diabetes: a 24-week, multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. The Lancet. 2023.
- 3. Finan B, et al. A strategically engineered GIP, GLP-1, and glucagon receptor co-agonist with potent anti-hyperglycemic and insulinotropic properties. Nature Medicine. 2013.
- 4. Müller TD, et al. GLP-1, GIP, and Glucagon Receptor Co-agonism: Next Generation Therapeutics for Obesity and Diabetes. Cell Metabolism. 2022.
- 5. Knerr PJ, et al. Next-generation multi-receptor agonists for the treatment of metabolic diseases. International Journal of Molecular Sciences. 2022.
- 6. Heise T, et al. Tirzepatide: A Novel Dual GIP and GLP-1 Receptor Agonist for the Treatment of Type 2 Diabetes Mellitus. Diabetes, Obesity and Metabolism. 2022.
- 7. Thomas MK, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-Cell Function and Insulin Sensitivity in Type 2 Diabetes. J Clin Invest. 2021.
- 8. Bastin M, et al. Glucagon receptor signaling and its metabolic physiological roles. Frontiers in Endocrinology. 2019.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.