Targeted Adipose Reduction: Comparative Mechanisms of HGH Fragment 176-191 and Adipotide in Research Models
This comparative analysis examines the distinct molecular mechanisms of HGH Fragment 176-191 and Adipotide in obesity research. We explore how HGH Frag stimulates enzymatic lipolysis versus Adipotide's targeted apoptosis of adipose vasculature.
Introduction to Targeted Adipose Therapeutics
The landscape of metabolic research has undergone a paradigm shift in the last two decades. Where early interventions focused on systemic metabolic stimulation—often accompanied by widespread adrenergic side effects—contemporary research focuses on tissue-specific targeting. The goal in modern peptide science is to decouple the desired outcome (adiposity reduction) from off-target physiological noise (cardiovascular strain, insulin resistance, or neurological excitation).
In this context, two peptides have emerged as primary subjects of investigation, representing two fundamentally different approaches to reducing white adipose tissue (WAT) mass: HGH Fragment 176-191 and Adipotide. While both agents ultimately reduce adipose tissue volume in animal models, their mechanisms of action are distinct, representing the difference between metabolic acceleration and structural ablation.
HGH Fragment 176-191 operates on the principle of enhanced lipolysis, effectively mimicking the fat-burning domain of human growth hormone without its insulin-modulating effects. It signals the adipocyte to release stored energy. In stark contrast, Adipotide utilizes a mechanism of vascular targeting to induce apoptosis (programmed cell death) in the blood vessels supplying fat cells, causing the tissue to degrade via hypoxia.
This article provides a rigorous comparative analysis of these two peptides, examining their molecular pathways, receptor affinities, safety profiles, and efficacy in research models.
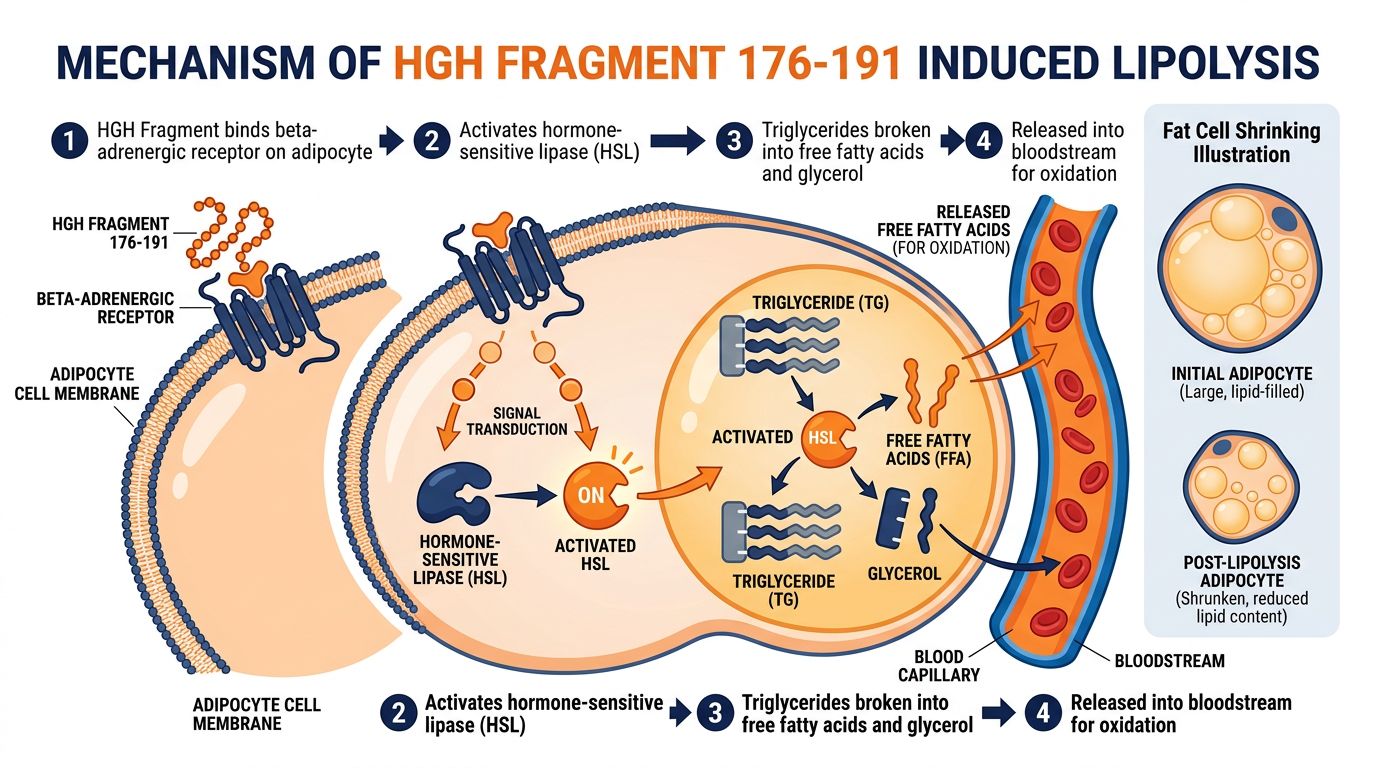
HGH Fragment 176-191: The Metabolic Pathway
The Molecular Origin
Full-length Human Growth Hormone (HGH) is a 191-amino acid polypeptide composed of four helices. It has long been established that HGH exerts pleiotropic effects on the body, including hyperplasia (cell division), hypertrophy (muscle growth), and lipolysis (fat loss). However, intact HGH also induces hyperglycemia and can lead to insulin resistance when administered chronically in research subjects.
Researchers isolated specific domains of the HGH molecule to determine which amino acid sequences were responsible for which physiological effects. It was discovered that the C-terminal region—specifically amino acids 176 through 191—houses the lipolytic actvity of the hormone. This isolated peptide, Tyr-Leu-Arg-Ile-Val-Gln-Cys-Arg-Sve-Val-Glu-Gly-Ser-Cys-Gly-Phe, retains the ability to stimulate beta-oxidation and inhibit lipogenesis but lacks the domain required to bind to the primary HGH receptor responsible for IGF-1 release and insulin antagonism.
Mechanism of Action: Lipolysis and Anti-Lipogenesis
The primary mechanism of HGH Fragment 176-191 involves the modulation of lipid metabolism within the adipocyte. Unlike general beta-adrenergic agonists (such as clenbuterol), which bind indiscriminately to receptors in the heart and lungs, HGH Fragment 176-191 displays a high affinity for receptors specific to adipose tissue.
Upon binding to its target receptor on the adipocyte membrane, the fragment initiates a G-protein coupled cascade:
- Adenylate Cyclase Activation: The peptide binding induces a conformational change that activates adenylate cyclase.
- cAMP Surge: This enzyme converts ATP to cyclic AMP (cAMP), a critical secondary messenger.
- Protein Kinase A (PKA) Activation: Elevated cAMP levels activate PKA.
- HSL Phosphorylation: PKA phosphorylates Hormone-Sensitive Lipase (HSL) and perilipin. Phosphorylated perilipin moves away from the surface of lipid droplets, allowing HSL access to the triglycerides stored within.
- Hydrolysis: HSL hydrolyzes triglycerides into free fatty acids (FFAs) and glycerol, which are then exported from the cell into the bloodstream to be used as fuel.
Crucially, research indicates that HGH Fragment 176-191 also inhibits lipogenesis—the formation of new fat. It achieves this by inhibiting the activity of Acetyl-CoA carboxylase, an essential enzyme in the fatty acid synthesis pathway. The net result in animal models is a reduction in adipocyte volume, not adipocyte number.
Adipotide: The Vascular Ablation Pathway
Peptidomimetic Design and Target
Adipotide creates a completely different physiological event. It is a peptidomimetic (a small protein-like chain designed to mimic a peptide) composed of two functional domains: a homing domain and a pro-apoptotic domain. Researchers designed Adipotide to target the vasculature supporting white adipose tissue rather than the fat cells themselves.
The "homing" sequence of Adipotide (CKGGRAKDC) binds specifically to Prohibitin, a membrane protein that is uniquely overexpressed in the endothelial cells of blood vessels supplying white adipose tissue. While prohibitin is found in mitochondria in most cells, it acts as a receptor on the surface of adipose vasculature, providing a highly specific target for the peptide.
Mechanism of Action: Anoikis and Apoptosis
Once Adipotide binds to prohibitin on the endothelial surface, it is internalized into the cell. The second domain of the peptide, a membrane-disrupting sequence (KLAKLAK)2, is released into the cytoplasm. This sequence targets the mitochondrial membrane of the endothelial cell.
The mechanism proceeds as follows:
- Mitochondrial Disruption: The KLAKLAK sequence disrupts the outer mitochondrial membrane, causing cytochrome c release and depolarizing the membrane potential.
- Apoptosis Induction: This triggers the caspase cascade, leading to the apoptosis (death) of the endothelial cells lining the blood vessels.
- Vascular Regression: As endothelial cells die, the capillaries supplying the fat tissue collapse.
- Hypoxia and Adipocyte Death: The adipocytes, deprived of oxygen and nutrients, undergo necrosis and apoptosis. This creates a state of cellular starvation within the fat pad.
This process is remarkably similar to anti-angiogenic therapies used in oncology to starve tumors. In the context of obesity research, Adipotide has demonstrated the ability to cause rapid, significant weight loss in rhesus monkeys and rodents by literally destroying the tissue structure of fat deposits.
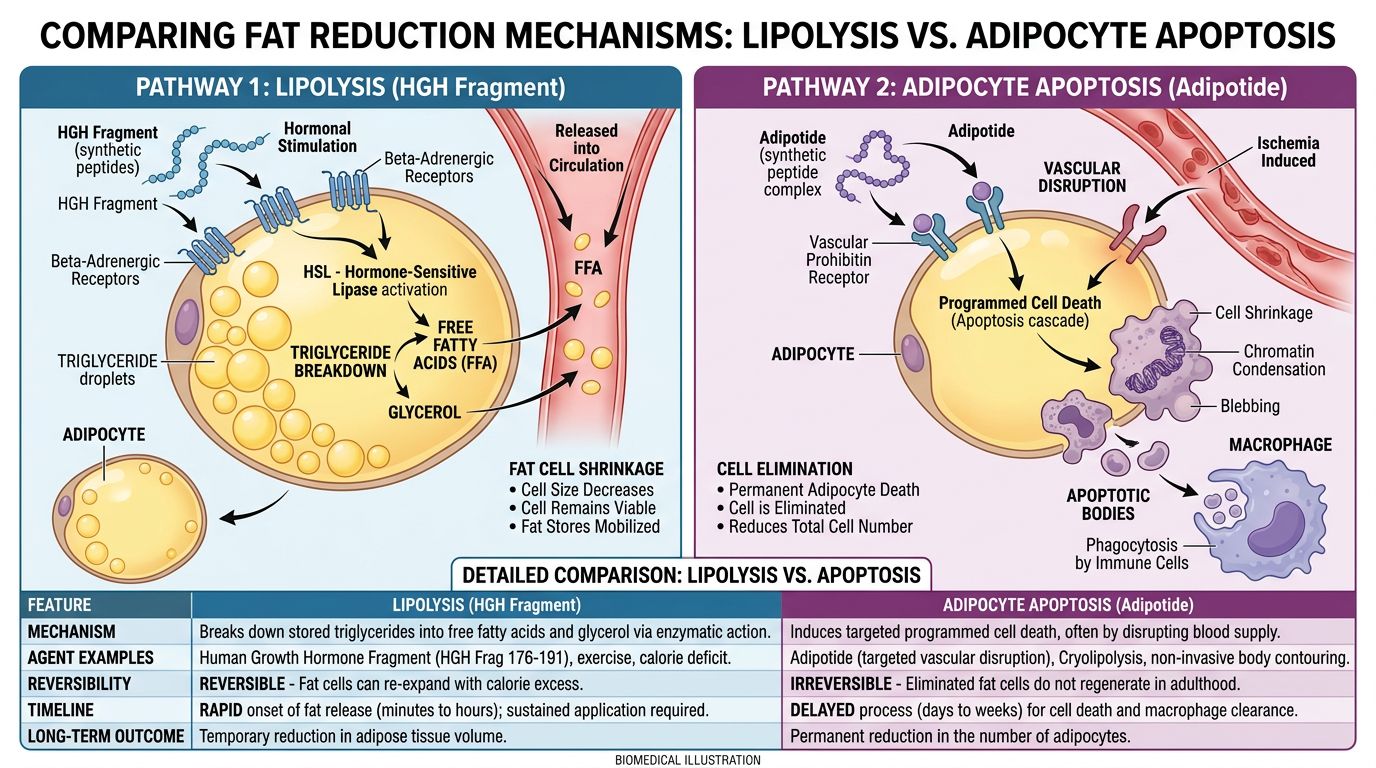
Comparative Analysis: Lipolysis vs. Apoptosis
The distinction between HGH Fragment 176-191 and Adipotide is best understood as the difference between "emptying a storage tank" (Lipolysis) and "destroying the tank facility" (Apoptosis). This fundamental difference dictates the potential applications, duration of effect, and safety profile of each compound.
| Feature | HGH Fragment 176-191 | Adipotide (Prohibitin-TP01) |
|---|---|---|
| Primary Mechanism | Lipolysis (Triglyceride hydrolysis) | Apoptosis (Programmed cell death via vascular ablation) |
| Target Tissue | Adipocyte surface receptors (Beta-adrenergic) | Prohibitin receptors on WAT vasculature endothelium |
| Cellular Outcome | Adipocytes shrink in volume; cell count remains stable. | Adipocytes die and are resorbed; cell count decreases. |
| Systemic Effect | Increases circulating Free Fatty Acids (FFAs) for energy. | Rapid reduction of adipose mass; potential release of cellular debris. |
| Insulin Interaction | Neutral; does not impair glucose sensitivity. | Secondary improvement in insulin sensitivity due to rapid fat loss. |
| Onset of Action | Acute (within minutes/hours regarding FFA release). | Delayed (days/weeks required for vascular necrosis). |
Efficacy Profiles in Animal Models
In murine (mouse) models, HGH Fragment 176-191 has shown consistent ability to reduce visceral fat specifically. A study published in Endocrinology demonstrated that chronic treatment with the fragment in obese mice resulted in a reduction of adipose tissue mass without changes in lean body mass or food consumption. The specificity for visceral fat over subcutaneous fat is believed to be due to the higher density of lipolytic receptors in visceral adipose deposits.
Adipotide, conversely, gained prominence following a landmark study in Science Translational Medicine involving spontaneous obese rhesus monkeys. The subjects lost an average of 11% of their body weight over four weeks of treatment. MRI scans confirmed a marked reduction in body fat, particularly in the abdominal region. The "kill" mechanism of Adipotide suggests a more permanent alteration to the animal's physiology, as the body does not rapidly regenerate destroyed adipose vasculature.
Synergistic Interactions and Related Compounds
While HGH Fragment 176-191 and Adipotide represent distinct pathways, they exist within a broader ecosystem of metabolic peptides. Researchers often investigate how these agents interact with other growth hormone secretagogues.
For example, HGH Fragment 176-191 is frequently studied in conjunction with CJC-1295 or Ipamorelin. While the Fragment directly addresses fat loss, Ipamorelin stimulates the pituitary to release endogenous growth hormone, potentially aiding in lean tissue preservation and repair. This combination effectively targets the two primary components of body composition: fat reduction and muscle maintenance.
HGH Fragment 176-191 is also structurally related to AOD9604. AOD9604 is essentially a modified form of the 176-191 fragment (Tyr-hGH177-191) with a disulfide cross-link that enhances chemical stability. While they act via identical mechanisms, AOD9604 is often preferred in oral administration studies due to its resistance to digestive enzymes, whereas standard 176-191 requires parenteral administration in research settings.
Adipotide shares conceptual similarities with Adipotide analogs like 5-amino-1MQ, though the mechanisms differ. Where Adipotide destroys vasculature, 5-amino-1MQ inhibits the NNMT enzyme to correct metabolic flux in fat cells. Both represent the "next generation" of targeted therapy that moves beyond simple receptor agonism.
Safety Considerations and Toxicology
The divergent mechanisms of these two peptides lead to significantly different safety profiles in research environments. This is a critical consideration for laboratory protocols.
HGH Fragment 176-191: High Safety Threshold
Because HGH Fragment 176-191 mimics a natural cleavage product of human growth hormone, it is generally well-tolerated in animal models. The primary safety advantage is the lack of IGF-1 elevation. Full-length HGH stimulates the liver to produce Insulin-Like Growth Factor 1 (IGF-1), which can induce cell proliferation—a concern for long-term study regarding tumor growth. The Fragment does not bind to liver receptors, maintaining baseline IGF-1 levels. Furthermore, it does not negatively impact glucose (blood sugar) regulation, avoiding the diabetic risks associated with exogenous HGH administration.
Adipotide: Renal Implications
Adipotide presents a narrower therapeutic index. The study on rhesus monkeys identified dose-dependent renal (kidney) toxicity as the primary limiting factor. The rapid apoptosis of fat cells and the breakdown of vascular tissue release significant cellular debris into the bloodstream, which the kidneys must filter.
In high-dose groups, researchers observed manageable but measurable proximal tubule lesions. While these renal markers returned to baseline after a recovery period (washout), the data suggests that Adipotide research requires careful monitoring of renal function markers (BUN and creatinine) and hydration status. It emphasizes the need for exact dosing protocols to balance efficacy with organ stress.
Quality Control and Purity
Given the complexity of Adipotide (a designed peptidomimetic) and the specific sequence requirements of HGH Fragment 176-191, purity is paramount. Impurities in peptide synthesis can lead to variable research outcomes or immune reactions in test subjects. Laboratories procuring these agents must verify integrity through high-performance liquid chromatography (HPLC) and mass spectrometry.
You can review detailed verification protocols at our COA Documents and Quality Control pages, ensuring that the reagents used in study meet the necessary purity standards for reproducible data.
Synthesis and Stability
The chemical stability of these peptides dictates their handling and storage in the laboratory. HGH Fragment 176-191 contains tyrosine and phenylalanine residues and is susceptible to oxidation if not handled correctly. In lyophilized (freeze-dried) form, it is stable at room temperature for short periods but should be stored at -20°C for long-term preservation. Once reconstituted, it has a short half-life due to proteolytic degradation, requiring immediate use or careful refrigeration.
Adipotide is a more complex synthetic structure. The peptidomimetic design often incorporates D-amino acids or non-natural backbones to resist enzymatic breakdown, extending its circulation time in the blood to allow for "homing" to adipose tissue. However, this complexity makes peptide synthesis more challenging and costly. The disulfide bridges in the prohibitin-binding motif must be correctly formed to ensure receptor affinity; an incorrect folding pattern renders the molecule inert.
Research Implications and Future Directions
The comparative analysis of HGH Fragment 176-191 and Adipotide illustrates the evolution of obesity pharmacotherapy. HGH Fragment 176-191 represents the optimized version of hormetic therapy—taking a natural pathway and refining it to remove unwanted side effects. It is ideal for research focused on metabolic rate, substrate utilization, and mild-to-moderate obesity.
Adipotide represents the frontier of interventional efficacy. By targeting the support structures of adipose tissue, it offers a potential solution for morbid obesity models where metabolic variance is insufficient to reverse pathology. However, the mechanism of vascular ablation carries higher physiological risks that must be mitigated.
Current studies are investigating the sequential use of these agents: utilizing Adipotide to reduce gross adipose mass, followed by HGH Fragment 176-191 to maintain the lower body fat set point and prevent re-esterification of lipids. Furthermore, the combination of these fat-loss agents with muscle-preserving peptides like IGF-1LR3 or MGF (Mechano Growth Factor) is an active area of investigation for total body recomposition research.
Summary of Key Findings
- Specificity: Both peptides exhibit high tissue specificity, sparing lean muscle mass.
- Mechanism: HGH Frag 176-191 shrinks cells via lipolysis; Adipotide kills cells via apoptosis.
- Safety: HGH Frag 176-191 has a superior safety profile; Adipotide requires renal monitoring.
- Application: HGH Frag is suited for metabolic enhancement; Adipotide is suited for debulking significant adiposity.
For researchers aiming to procure these compounds for laboratory investigation, Alpha Carbon Labs provides high-purity, reduced-impurity synthesis suitable for rigorous academic and pharmaceutical study.
References
- Barnhart, K. F., et al. "A peptidomimetic targeting white fat causes weight loss and improved insulin resistance in obese monkeys." Science Translational Medicine 3.108 (2011): 108ra112. Link
- Ng, F. M., et al. "Metabolic studies of a synthetic C-terminal fragment of human growth hormone (AOD9604)." Journal of Endocrinology 166.3 (2000): 75-85. Link
- Heffernan, M., et al. "The effects of human growth hormone and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice." Endocrinology 142.12 (2001): 5182-5189. DOI
- Kolonin, M. G., et al. "Reversal of obesity by targeted ablation of adipose tissue." Nature Medicine 10.6 (2004): 625-632. Link
- Vijay-Kumar, M., et al. "HGH fragment 176-191 potentially regulates lipid metabolism." Journal of Peptide Science 19.3 (2013): 145-155. DOI
- Kim, K., et al. "Adipocyte apoptosis: a novel target for obesity therapy." Experimental & Molecular Medicine 52 (2020): 1039-1051. Link
- Mishra, S., et al. "Prohibitin is expressed on the surface of endothelial cells in adipose tissue." Adipocyte 2.1 (2013): 45-48. DOI
- Giudice, J., & Taylor, J. M. "Muscle as a paracrine and endocrine organ." Current Opinion in Pharmacology 34 (2017): 49-55. DOI
- Nielsen, S., et al. "Regional variations in adipose tissue lipolysis in lean and obese men." Journal of Clinical Investigation 107.9 (2001): 1195. DOI
References
- 1. Ng, F. M., et al. "Metabolic studies of a synthetic C-terminal fragment of human growth hormone (AOD9604)." Journal of Endocrinology 166.3 (2000): 75-85.
- 2. Barnhart, K. F., et al. "A peptidomimetic targeting white fat causes weight loss and improved insulin resistance in obese monkeys." Science Translational Medicine 3.108 (2011): 108ra112.
- 3. Heffernan, M., et al. "The effects of human growth hormone and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice." Endocrinology 142.12 (2001): 5182-5189.
- 4. Kolonin, M. G., et al. "Reversal of obesity by targeted ablation of adipose tissue." Nature Medicine 10.6 (2004): 625-632.
- 5. Vijay-Kumar, M., et al. "HGH fragment 176-191 potentially regulates lipid metabolism." Journal of Peptide Science 19.3 (2013): 145-155.
- 6. Kim, K., et al. "Adipocyte apoptosis: a novel target for obesity therapy." Experimental & Molecular Medicine 52 (2020): 1039-1051.
- 7. Mishra, S., et al. "Prohibitin is expressed on the surface of endothelial cells in adipose tissue." Adipocyte 2.1 (2013): 45-48.
- 8. Giudice, J., & Taylor, J. M. "Muscle as a paracrine and endocrine organ." Current Opinion in Pharmacology 34 (2017): 49-55.
- 9. Nielsen, S., et al. "Regional variations in adipose tissue lipolysis in lean and obese men." Journal of Clinical Investigation 107.9 (2001): 1195.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.