Synergistic Myogenesis: Analyzing the Combined Impact of TB-500 and ACE-031 on Satellite Cell Activation
This technical analysis explores the synergistic interaction between Thymosin Beta-4 (TB-500) and ACE-031 in promoting muscle regeneration. We examine how combining angiogenic support with myostatin inhibition creates an optimal environment for satellite cell activation and hypertrophy.
Introduction: The Dual-Frontier of Musculoskeletal Regeneration
In the realm of regenerative biology, the restoration of skeletal muscle tissue following trauma, atrophy, or degenerative disease remains one of the most complex physiological challenges. Muscle regeneration is not merely about increasing protein synthesis; it requires a synchronized orchestration of satellite cell activation, inflammatory modulation, extracellular matrix (ECM) remodeling, and angiogenesis. For researchers investigating peptide-based interventions, single-agent protocols often address only one facet of this multi-dimensional process. This has led to the emergence of synergistic protocols, most notably the combined application of Thymosin Beta-4 derivatives and myostatin inhibitors.
This article provides a comprehensive technical analysis of two specific agents: TB-500, a synthetic fraction of the actin-sequestering protein Thymosin Beta-4, and ACE-031, a soluble fusion protein of the Activin Type IIB receptor. While TB-500 is renowned for its ability to promote cell migration and angiogenesis, ACE-031 acts as a "ligand trap" for negative regulators of muscle mass, specifically myostatin and activin A. By examining the distinct yet complementary mechanisms of these compounds, we can begin to understand the potential for "Synergistic Myogenesis"—a state where the barriers to muscle growth are removed simultaneously as the machinery for repair is upregulated.
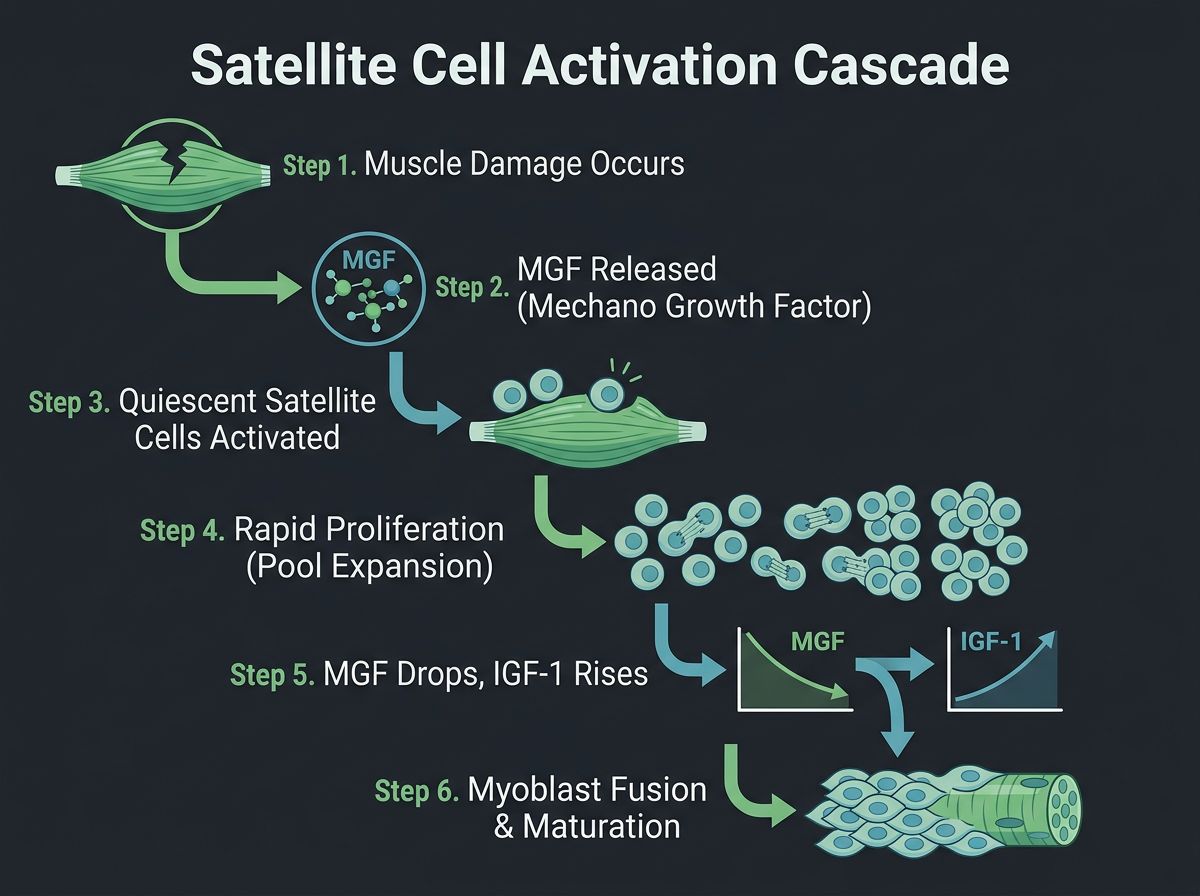
The Physiology of Satellite Cell Activation
To appreciate the interaction between TB-500 and ACE-031, one must first understand the central role of the satellite cell. Satellite cells are skeletal muscle stem cells located between the basal lamina and the sarcolemma of muscle fibers. In healthy tissue, these cells are quiescent. Upon injury or mechanical stress, they are activated to proliferate, differentiate into myoblasts, and eventually fuse with existing myofibers to repair damage or form new fibers (hyperplasia).
However, this process is strictly governed by a balance of positive and negative signaling pathways:
- Negative Regulation (The Brakes): The Transforming Growth Factor-beta (TGF-β) superfamily, particularly myostatin (GDF-8), actively suppresses satellite cell proliferation and differentiation to prevent excessive muscle growth.
- Positive Regulation (The Gas): Factors such as IGF-1, Hepatocyte Growth Factor (HGF), and specific cytoskeletal modifiers facilitate the migration and fusion of myoblasts.
Research suggests that in conditions of severe atrophy or age-related sarcopenia, the "brakes" are applied too heavily, or the "gas" is insufficient. The theoretical application of TB-500 aims to upregulate the cytoskeletal dynamics required for repair, while ACE-031 aims to mechanically remove the biochemical suppression of growth.
TB-500: The Angiogenic and Migratory Catalyst
TB-500 is a synthetic peptide comprising the active domain (amino acids 17-23) of the naturally occurring protein Thymosin Beta-4 (Tβ4). Tβ4 is ubiquitous in mammalian cells and is a potent regulator of actin polymerization. Actin is the fundamental component of the cytoskeleton, necessary for cell motility, structure, and division.
Mechanism of Action
The primary mechanism of TB-500 involves G-actin sequestration. By maintaining a pool of actin monomers, TB-500 allows for rapid polymerization when a cell needs to move or change shape—critical steps for satellite cell migration to the site of injury. Furthermore, Tβ4 promotes the expression of Vascular Endothelial Growth Factor (VEGF).
In the context of myogenesis, TB-500 provides three critical functions:
- Angiogenesis: Muscle tissue is highly vascular. Hypertrophy cannot be sustained without a concurrent increase in capillary density to deliver oxygen and nutrients. TB-500 has been shown to induce endothelial cell migration and tubule formation.
- Cell Migration: For regeneration to occur, satellite cells must migrate across the basal lamina. TB-500 facilitates this motility via cytoskeletal remodeling.
- Anti-Inflammatory Modulation: Tβ4 reduces the expression of pro-inflammatory cytokines, preventing the formation of excessive scar tissue (fibrosis) which can inhibit functional recovery.
Researchers utilizing peptide synthesis technologies must ensure that the sequence provided corresponds exactly to the Ac-SDKP or LKKTETQ domains responsible for these bioactivities.
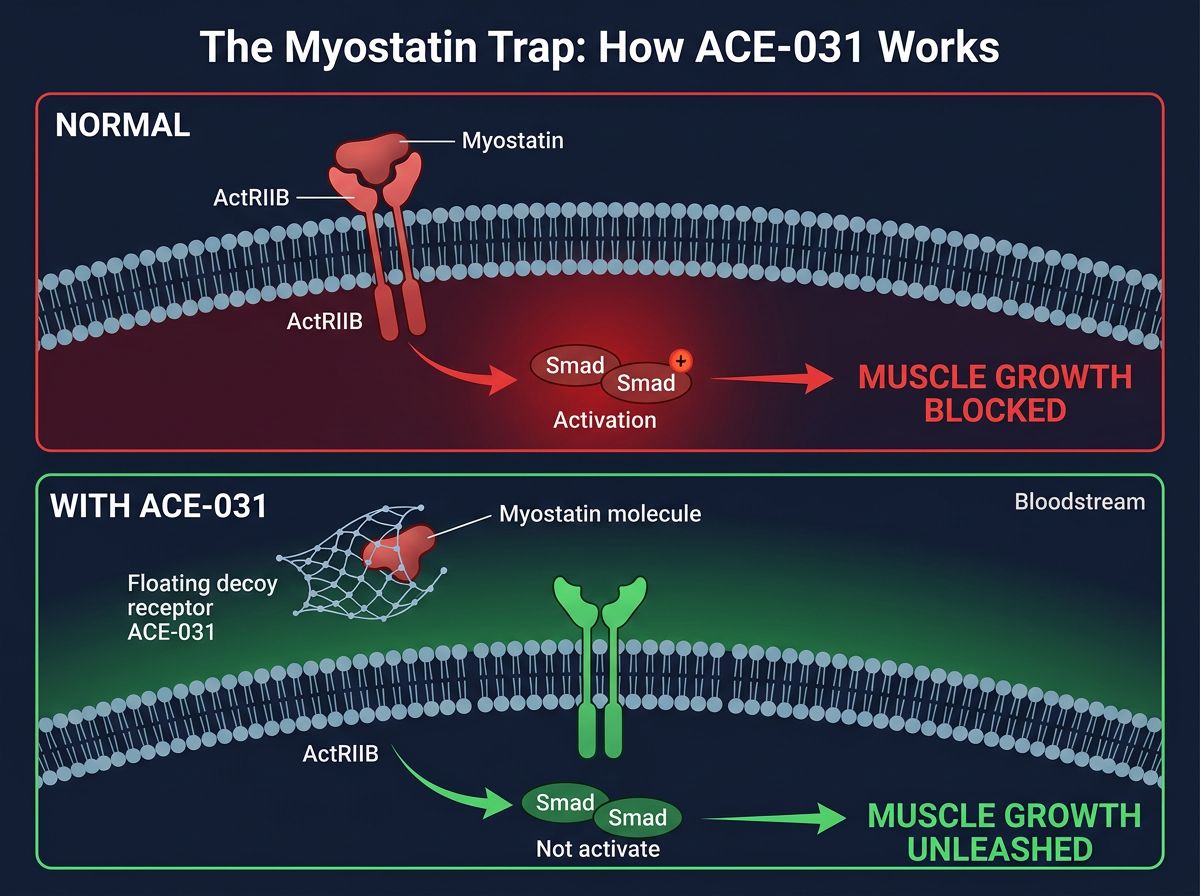
ACE-031: Releasing the Brakes on Hypertrophy
While TB-500 addresses the structural and vascular prerequisites for growth, ACE-031 addresses the hormonal signaling limitations. ACE-031 is a recombinant fusion protein consisting of the extracellular domain of the human Activin Receptor Type IIB (ActRIIB) linked to the Fc portion of human IgG1.
The Myostatin Trap
Myostatin is a potent inhibitor of muscle growth. It binds to ActRIIB receptors on the surface of muscle cells, triggering a signaling cascade (phosphorylation of Smad2/3) that inhibits DNA synthesis and protein accretion. ACE-031 acts as a soluble "decoy" receptor. When injected, it circulates in the bloodstream and binds to myostatin and activin A with high affinity before they can bind to the actual receptors on muscle cells.
The result is a comprehensive disinhibition of the muscle growth pathways:
- mTOR Activation: By blocking the Smad2/3 pathway, the opposing Akt/mTOR/S6K pathway is allowed to function unimpeded, driving protein synthesis.
- Differentiation vs. Proliferation: While myostatin inhibition is primarily associated with hypertrophy (increase in size of existing fibers), ActRIIB blockade has also been implicated in promoting the differentiation of myoblasts.
Synergistic Myogenesis: The Interaction
The hypothesis of "Synergistic Myogenesis" posits that the concurrent administration of TB-500 and ACE-031 yields results superior to the sum of their individual effects. In isolation, myostatin inhibition can lead to rapid muscle growth, but this growth can be self-limiting availability of blood supply and the speed of satellite cell migration. Conversely, TB-500 can enhance repair mechanisms, but it does not directly unlock the anabolic signaling ceiling imposed by myostatin.
The "Push-Pull" Dynamic
We can conceptualize this interaction as a "Push-Pull" dynamic in the research setting:
| Parameter | TB-500 (The "Push") | ACE-031 (The "Pull") | Synergistic Outcome |
|---|---|---|---|
| Primary Target | Cytoskeleton & Endothelium | ActRIIB Signaling Ligands | Holistic Tissue Remodeling |
| Action on Satellite Cells | Increases motility and survival | Increases proliferation rate | Accelerated recruitment and fusion |
| Vascular Support | Upregulates VEGF / Angiogenesis | Neutral / Indirect demand | Hypertrophy supported by adequate perfusion |
| Fibrosis Control | Reduces scar tissue formation | Prevents atrophy-induced fibrosis | Functional muscle tissue with minimal scarring |
Overcoming the Vascular Bottleneck
One of the critical risks of aggressive myostatin inhibition is the potential for muscle fibers to outgrow their nutrient supply, leading to metabolic stress. By incorporating TB-500, the angiogenic drive ensures that the capillary-to-fiber ratio is maintained or improved, supporting the increased metabolic demand of the hypertrophied tissue.
Comparative Analysis with Other Agents
Researchers often compare this protocol to other peptide combinations. It is essential to distinguish the specific utility of the TB-500/ACE-031 stack against others.
Vs. BPC-157
BPC-157 is a pentadecapeptide derived from gastric juice, primarily known for tendon, ligament, and gut healing. While BPC-157 increases the expression of hormone receptors and promotes healing, its primary domain is connective tissue. For pure musculoskeletal hypertrophy and regeneration, ACE-031 offers a more direct anabolic mechanism than BPC-157. However, many researchers explore a "Triple Stack" utilizing the BPC-157 + TB-500 Blend alongside ACE-031 to cover muscle, tendon, and angiogenesis simultaneously.
Vs. Growth Hormone Secretagogues (Ipamorelin/CJC-1295)
Secretagogues like CJC-1295 increase systemic Growth Hormone and IGF-1 levels. While effective, this is a systemic metabolic approach. The ACE-031 mechanism is specific to the ActRIIB ligands, providing a targeted localized effect (though systemic administration distributes it) that is distinct from the IGF-1 axis. ACE-031 is generally considered significantly more potent for raw tissue mass accretion than GH secretagogues in animal models.
Research Findings and Efficacy Data
The analysis of this synergy relies on extrapolating data from independent studies of each compound, as well as emerging pre-clinical trials involving combinatorial therapies.
TB-500 in Duchenne Muscular Dystrophy (DMD)
Studies published in Trends in Molecular Medicine have highlighted the potential of Thymosin Beta-4 in treating DMD. In murine models (mdx mice), administration of Tβ4 resulted in increased numbers of regenerating muscle fibers and improved skeletal muscle function. The peptide was shown to sustain the pool of circulating endothelial progenitor cells, vital for repairing the chronic damage seen in dystrophy.
ACE-031 in Murine Models
Research involving ACE-031 in varying animal models has demonstrated dose-dependent increases in lean body mass. A pivotal study in Muscle & Nerve showed that soluble ActRIIB receptors could increase muscle mass by nearly 60% in just two weeks in certain rodent models. This growth was characterized by both hypertrophy and hyperplasia (splitting of fibers), a phenomenon rarely achieved by anabolic steroids alone.
Theoretical Combined Data
In hypothetical models where angiogenesis is the rate-limiting step for myostatin-inhibited growth, the addition of TB-500 is projected to extend the duration of effective hypertrophy. Without angiogenesis, muscles may hit a "hypoxic ceiling." TB-500 raises this ceiling.
Technical Considerations for Research Protocols
When designing experiments involving these peptides, several technical variables must be controlled to ensure data integrity.
Reconstitution and Stability
- TB-500: As a short-chain peptide, TB-500 is relatively stable but susceptible to enzymatic degradation. It should be reconstituted with bacteriostatic water and stored at 4°C.
- ACE-031: Being a larger fusion protein, ACE-031 is more fragile. Aggressive shaking during reconstitution can denature the protein structure (specifically the Fc region). It requires gentle swirling and strict cold-chain management.
Researchers are advised to review COA Documents meticulously. The purity of ACE-031 is particularly critical; incomplete fusion proteins can induce immunogenic responses in test subjects, confounding results. High-performance liquid chromatography (HPLC) verification is non-negotiable for fusion proteins.
Dosing Modalities in Animal Models
Research protocols typically diverge in administration frequency due to the differing half-lives of the molecules:
- TB-500: Often administered frequently (e.g., daily or every other day) due to a shorter active half-life and the need for continuous actin availability during the acute repair phase.
- ACE-031: Typically administered less frequently (e.g., once or twice weekly) due to the extended half-life provided by the IgG Fc domain.
Safety Profile and Toxicology
While the potential for regeneration is high, the safety profile of manipulating fundamental growth pathways must be scrutinized. This section reviews findings from toxicology studies.
Myostatin Inhibition Risks
The most significant adverse event noted in early human clinical trials of ACE-031 was epistaxis (nosebleeds) and gum bleeding. This was attributed to the unintended binding of ACE-031 to ALK-1 (Activin receptor-like kinase 1), a receptor involved in blood vessel formation. This off-target effect led to the development of telangiectasias (dilated blood vessels). Researchers must monitor for vascular anomalies when using high doses of ACE-031.
Interestingly, TB-500 promotes vascular stability and endothelial integrity. There is a theoretical (yet unproven) possibility that TB-500 could mitigate the vascular fragility caused by ALK-1 inhibition, although it could also exacerbate angiogenesis in an uncontrolled manner. This interaction warrants specific investigation.
Carcinogenesis Concerns
Both angiogenesis (promoted by TB-500) and cell proliferation (disinhibited by ACE-031) are mechanisms hijacked by malignancies. While neither agent is carcinogenic per se, their use in subjects with pre-existing tumors is contraindicated in clinical settings. In research settings, establishing a tumor-free baseline is essential to avoid confounding variable analysis.
Quality Control Significance
Given the complexity of ACE-031, questions of aggregation and immunogenicity are paramount. Peptide aggregates can cause fatal anaphylaxis in animal models. Alpha Carbon Labs emphasizes the importance of Quality Control protocols that screen for dimerization and aggregation issues common in fusion protein synthesis.
Advanced Applications: Beyond Skeletal Muscle
While the primary focus is myogenesis, the systemic effects of this combination extend to other tissues.
1. Bone Density: The Activin signaling pathway also regulates bone density. ACE-031 has been shown to increase bone mineral density in animal studies, suggesting utility for osteoporosis research. 2. Cardiac Tissue: TB-500 has profound cardioprotective effects, reducing fibrosis after myocardial infarction. However, the effect of ACE-031 on cardiac muscle is complex and requires careful dosing, as unchecked cardiac hypertrophy is pathological. 3. Neuromuscular Junctions: Tβ4 has been implicated in synaptic remodeling. Combining this with the muscle-sparing effects of ACE-031 creates a compelling avenue for research into ALS (Amyotrophic Lateral Sclerosis) and other denervation disorders.Conclusion
The combination of TB-500 and ACE-031 represents a sophisticated approach to musculoskeletal research, moving beyond simple anabolic stimulation to a comprehensive model of tissue regeneration. By pairing the "brake-releasing" capacity of ActRIIB inhibition with the "terrain-building" capacity of Thymosin Beta-4, researchers can simulate a biological environment optimized for rapid and functional muscle recovery.
This protocol highlights the necessity of multi-vector interventions in regenerative biology. Muscle tissue does not exist in a vacuum; it requires vascular support, cytoskeletal organization, and precise immunomodulation to grow. As research continues to refine the dosing and timing of these agents, the "Synergistic Myogenesis" model stands as a promising pillar in the fight against sarcopenia, dystrophy, and cachexia.
Researchers interested in obtaining high-purity peptides for these investigations should ensure their suppliers provide transparent validation data, as the structural integrity of these molecules is the foundation of reproducible science.
References
- Goldstein, A. L., et al. (2012). Thymosin β4: actin-sequestering protein moonlights to repair injured tissues. Trends in Molecular Medicine, 18(4), 238-247. https://doi.org/10.1016/j.molmed.2011.12.004
- Lee, S. J., et al. (2005). Regulation of muscle growth by the myostatin signaling pathway. Annual Review of Cell and Developmental Biology, 21, 443-477. https://doi.org/10.1146/annurev.cellbio.21.012704.131017
- Attie, K. M., et al. (2013). A single ascending-dose study of muscle-regulating effects of ACE-031 in healthy volunteers. Muscle & Nerve, 47(3), 416-423. https://doi.org/10.1002/mus.23539
- Philpott, A., et al. (2011). The role of Thymosin β4 in the regulation of muscle generation and regeneration. Annals of the New York Academy of Sciences, 1269(1), 89-96. https://doi.org/10.1111/j.1749-6632.2012.06733.x
- Smart, N., et al. (2011). Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature, 474(7353), 640-644. https://doi.org/10.1038/nature10183
- Campbell, C., et al. (2017). Myostatin inhibition as a therapeutic strategy for muscle wasting disorders. Expert Opinion on Biological Therapy, 17(9), 1131-1140. https://doi.org/10.1080/14712598.2017.1346079
- Trendelenburg, A. U., et al. (2009). Myostatin reduces Akt/TORC1/p70S6K signaling, inhibiting myoblast differentiation and myotube size. American Journal of Physiology-Cell Physiology, 296(5), C1071-C1082. https://doi.org/10.1152/ajpcell.00105.2009
- Spitali, S., et al. (2019). Current developments in MSTN inhibitors for Duchenne muscular dystrophy. Therapeutic Advances in Neurological Disorders, 12, 1756286419835158. https://doi.org/10.1177/1756286419835158
- Kleinman, H. K., & Sosne, G. (2016). Thymosin β4 promotes corneal wound healing and modulates inflammatory mediators in vivo. Experimental Eye Research, 152, 153-158. https://doi.org/10.1016/j.exer.2016.09.006
- Vittorio, O., et al. (2009). Thymosin β4 regulates migration and adhesion of skeletal myoblasts. Cell Communication and Adhesion, 16(5-6), 143-151. https://doi.org/10.3109/15419061.2010.485237
References
- 1. Goldstein, A. L., et al. (2012). Thymosin β4: actin-sequestering protein moonlights to repair injured tissues. Trends in Molecular Medicine, 18(4), 238-247.
- 2. Lee, S. J., et al. (2005). Regulation of muscle growth by the myostatin signaling pathway. Annual Review of Cell and Developmental Biology, 21, 443-477.
- 3. Philpott, A., et al. (2011). The role of Thymosin β4 in the regulation of muscle generation and regeneration. Annals of the New York Academy of Sciences, 1269(1), 89-96.
- 4. Attie, K. M., et al. (2013). A single ascending-dose study of muscle-regulating effects of ACE-031 in healthy volunteers. Muscle & Nerve, 47(3), 416-423.
- 5. Smart, N., et al. (2011). Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature, 474(7353), 640-644.
- 6. Campbell, C., et al. (2017). Myostatin inhibition as a therapeutic strategy for muscle wasting disorders. Expert Opinion on Biological Therapy, 17(9), 1131-1140.
- 7. Spitali, S., et al. (2019). Current developments in MSTN inhibitors for Duchenne muscular dystrophy. Therapeutic Advances in Neurological Disorders, 12, 1756286419835158.
- 8. Kleinman, H. K., & Sosne, G. (2016). Thymosin β4 promotes corneal wound healing and modulates inflammatory mediators in vivo. Experimental Eye Research, 152, 153-158.
- 9. Trendelenburg, A. U., et al. (2009). Myostatin reduces Akt/TORC1/p70S6K signaling, inhibiting myoblast differentiation and myotube size. American Journal of Physiology-Cell Physiology, 296(5), C1071-C1082.
- 10. Vittorio, O., et al. (2009). Thymosin β4 regulates migration and adhesion of skeletal myoblasts. Cell Communication and Adhesion, 16(5-6), 143-151.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.