Structural Biochemistry: How Retatrutide Achieves High-Affinity Triple Agonism at the Molecular Level
Retatrutide represents a breakthrough in peptide engineering, utilizing a 39-amino acid chimeric sequence to achieve high-affinity binding at the GIP, GLP-1, and Glucagon receptors simultaneously.
Introduction to Triple Agonism
In the rapidly evolving landscape of metabolic research, the transition from mono-agonism to multi-receptor targeting has represented a paradigm shift. While Semaglutide demonstrated the profound efficacy of GLP-1 receptor (GLP-1R) activation, and Tirzepatide introduced the synergistic benefits of dual GIPR/GLP-1R agonism, a new horizon has emerged: the triple agonist. Retatrutide (LY3437943) stands at the pinnacle of this structural evolution, designed to target the Glucagon receptor (GCGR), Glucose-dependent Insulinotropic Polypeptide receptor (GIPR), and Glucagon-like Peptide-1 receptor (GLP-1R) simultaneously.
The biochemical challenge of creating a single peptide capable of high-affinity binding to three distinct Class B G-protein coupled receptors (GPCRs) is immense. It requires precise spatial orientation of amino acid side chains to satisfy three different binding pockets while maintaining metabolic stability. This article provides a deep dive into the structural biochemistry of Retatrutide, analyzing how its molecular blueprint achieves what was once thought impossible in peptide engineering.
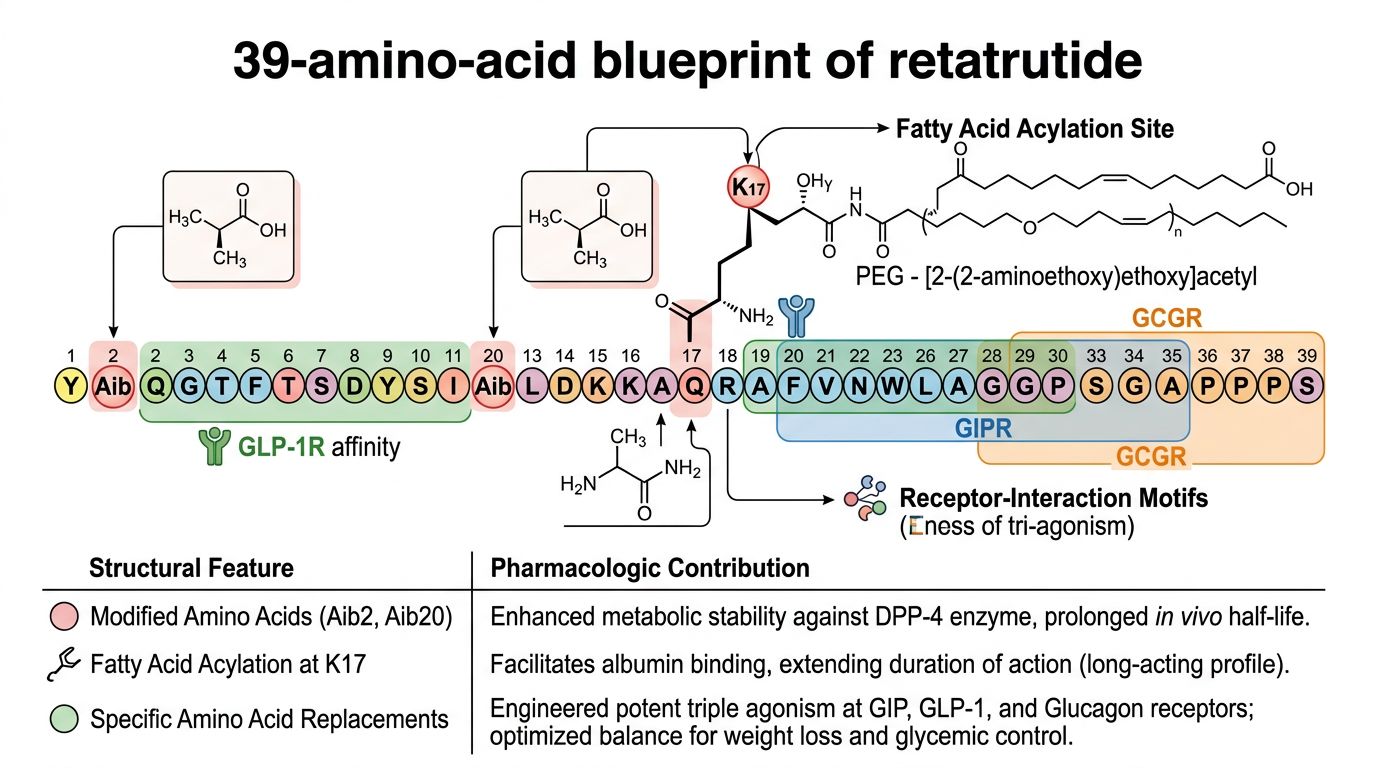
The Molecular Blueprint: A 39-Amino Acid Masterpiece
Retatrutide is a synthetic 39-amino acid peptide. Its primary sequence is derived from the GIP backbone but incorporates critical modifications from GLP-1 and Glucagon sequences to foster cross-reactivity. The engineering of Retatrutide is not merely an exercise in blending sequences; it is a sophisticated optimization of the peptide’s helical structure and lipophilicity.
The GIP-Based Backbone
The choice of a GIP backbone is strategic. GIP is a longer peptide than GLP-1, and its native structure provides a robust scaffold for the addition of an acylation tail, which is necessary for albumin binding and extended half-life. Scientists utilized the GIP scaffold to maintain high potency at the GIP receptor, which plays a dominant role in the metabolic synergy of these "nutrient-stimulated" hormones. Retatrutide maintains several key residues from native GIP that ensure it remains the most potent component of its triple-action profile.
Specific Amino Acid Substitutions
To achieve triple agonism, several non-proteogenic and specific amino acid substitutions were implemented at the N-terminus and along the mid-section of the peptide:
- Aib (α-aminoisobutyric acid) at positions 2 and 20: These synthetic amino acids provide resistance to Dipeptidyl Peptidase-4 (DPP-4) enzymatic degradation. Unlike natural L-amino acids, Aib induces a specific helical constraint (310-helix vs α-helix) that protects the peptide bonds from cleavage, a standard technique also seen in Semaglutide.
- Glucagon-specific residues: To recruit the Glucagon receptor, specific residues such as Gln3 and Lys10 were optimized. These residues interact with the extracellular domain (ECD) of the GCGR, facilitating the conformational change required for G-protein signaling.
- GLP-1 mimicry: The C-terminal portion of Retatrutide includes residues designed to stabilize the peptide's interaction with the GLP-1 receptor's hydrophobic pocket, ensuring that while it is a GIP-backbone peptide, its GLP-1R affinity remains clinically relevant.
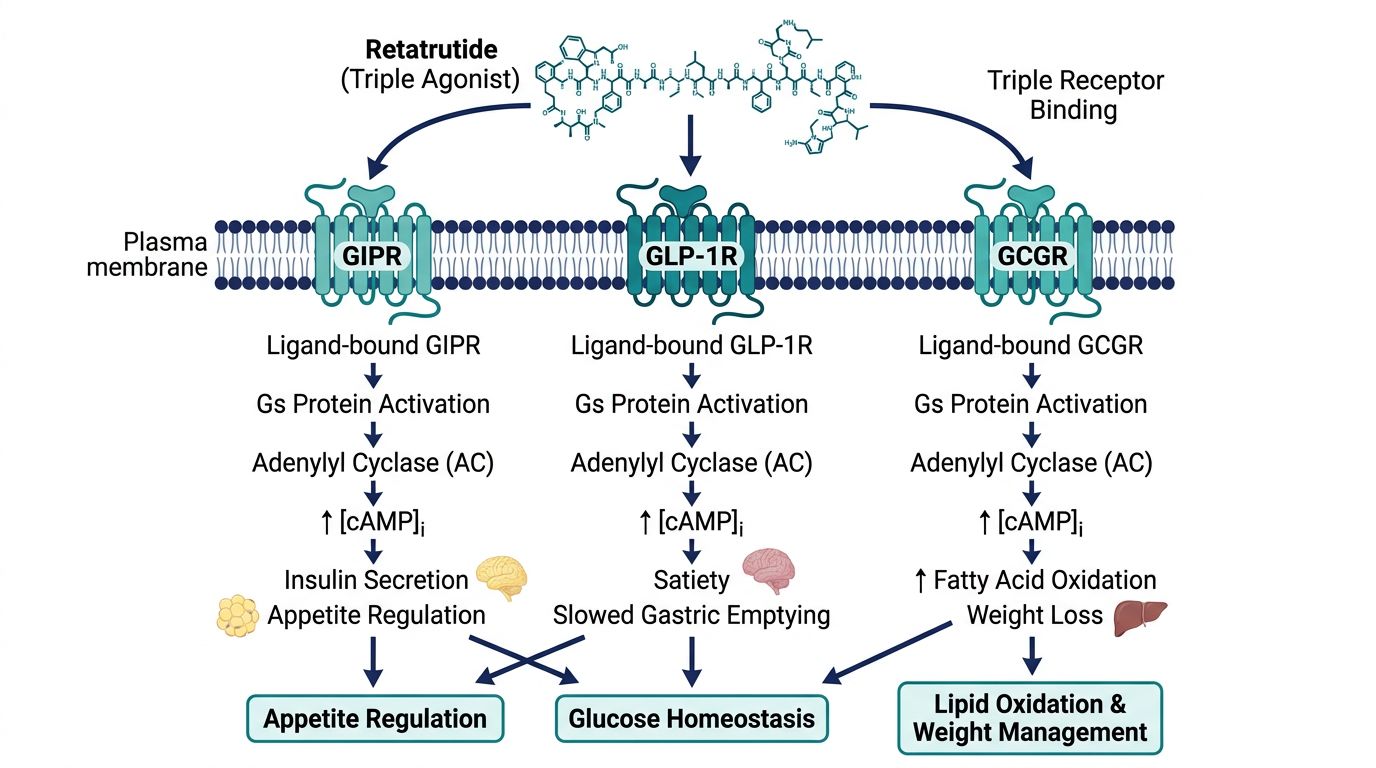
The Mechanics of Triple Binding: Receptor Interactions
The structural biochemistry of Retatrutide relies on the "chimeric" nature of its sequence. When a researcher introduces Retatrutide into a biological system, it must navigate the distinct structural requirements of three receptors:
| Receptor | Primary Physiological Role | Retatrutide’s Structural Adaptation |
|---|---|---|
| GIPR | Insulin secretion, lipid metabolism, neuroprotection | GIP-based N-terminus and helical core preservation. |
| GLP-1R | Appetite suppression, gastric emptying, insulinotropism | Specific substitution of GLP-1-like residues at the mid-section. |
| GCGR | Energy expenditure, thermogenesis, hepatic lipid oxidation | Glucagon-derived residues at the N-terminal signaling domain. |
GIP Receptor (GIPR) Dominance
In the structural hierarchy of Retatrutide, GIPR affinity remains the highest. Research indicates that Retatrutide acts as a full agonist at the GIPR. This is achieved through the preservation of the N-terminal Tyr1 and the overall hydrophobicity of the peptide's amphipathic helix. The GIPR is particularly sensitive to the N-terminal structure, and Retatrutide’s design ensures that the "key-in-lock" mechanism for GIPR is nearly identical to the endogenous ligand.
Recruiting the Glucagon Receptor (GCGR)
The inclusion of Glucagon agonism is what differentiates Retatrutide from dual agonists like Tirzepatide. To achieve this, engineers focused on the N-terminal signaling domain. The interaction with GCGR requires a specific alignment of the peptide's first 10 amino acids with the receptor’s transmembrane helices (TMs). By modifying positions 12 and 16, Retatrutide gains the ability to trigger the GCGR, which is hypothesized to increase resting energy expenditure and enhance hepatic lipid metabolism—factors that may contribute to the rapid reduction in liver fat observed in research settings.
GLP-1 Receptor (GLP-1R) Interaction
The GLP-1R binding of Retatrutide is slightly "weaker" than its GIPR binding but still highly potent. This "imbalanced" agonism is intentional. Excessive GLP-1R activation can lead to gastrointestinal distress, whereas balancing it with GIPR and GCGR agonism allows for higher total metabolic throughput with potentially better tolerability. The structural affinity for GLP-1R is maintained through the specific arrangement of residues 19-30, which dock into the GLP-1R extracellular domain.
C4-Diacid Fatty Acid Acylation: The Secret to Longevity
A critical component of Retatrutide’s molecular structure is the fatty acid side chain attached to the Lysine residue at position 17. Retatrutide utilizes a C20 fatty diacid moiety. This is an advancement over the C18 side chain found in previous generations of peptides.
Albumin Binding: The C20 fatty acid side chain has a high affinity for human serum albumin (HSA). When the peptide is administered, it binds to albumin in the bloodstream, creating a reservoir. This prevents rapid renal clearance and protects the peptide from proteolysis. This modification is the reason Retatrutide possesses a half-life of approximately 6 days, allowing for consistent receptor activation over a sustained period.
Lipid Spacer: The acylation is connected via a linker (typically containing gamma-Glu and/or AEEA spacers). This spacer provides the necessary flexibility for the fatty acid to bind to albumin without interfering with the peptide's ability to bind to its three target GPCRs. The positioning at Lys17 is "the sweet spot"—it is located on the non-binding face of the alpha-helix, ensuring it does not obstruct the receptor interface.
Synergistic Pharmacodynamics at the Cellular Level
The structural ability to bind three receptors results in a unique intracellular signaling profile. While a GLP-1 mono-agonist like Semaglutide primarily increases cAMP through Gαs activation in pancreatic beta cells and the brain, Retatrutide activates multiple pathways:
- GLP-1R/GIPR Synergy: Concurrent activation leads to potentiation of insulin secretion (the incretin effect) while potentially mitigating some of the nausea associated with GLP-1 alone.
- GCGR Activation: This triggers hepatic glucose production (buffered by the insulinotropic effects of GLP-1/GIP) and, more importantly, fatty acid oxidation. It also activates thermogenic pathways in brown adipose tissue (BAT).
- Combined Effect on Homeostasis: The structural integration of these three signals mimics the natural state of "nutrient abundance" more holistically than single-hormone mimics, leading to profound effects on adiposity and glucose regulation.
Comparison with Other Research Peptides
Researchers often compare Retatrutide to other metabolic modulators. While AOD9604 or HGH Frag 176-191 target lipolysis via growth hormone pathways, Retatrutide operates through the physiological incretin and glucagon systems. Similarly, while 5-Amino-1MQ targets NNMT to increase metabolic rate, Retatrutide achieves a similar metabolic acceleration through the GCGR-mediated thermogenic pathway.
Compared to dual agonists like Mazdutide (GCGR/GLP-1R) or Tirzepatide (GIPR/GLP-1R), Retatrutide is the first to successfully integrate all three. The structural complexity required to manage three receptors is significantly higher than two, requiring more precise molecular modeling and quality control during synthesis.
Synthesis and Purity Considerations
Given its 39-amino acid length and complex acylation, the synthesis of Retatrutide requires advanced Solid Phase Peptide Synthesis (SPPS) techniques. Every batch must undergo rigorous testing via HPLC and Mass Spectrometry to ensure the correct sequence and the proper attachment of the C20 diacid. Researchers can verify these parameters by examining COA documents, which confirm the purity and molecular weight consistency of the peptide.
The Future of Triple Agonism
Retatrutide represents a landmark in structural biochemistry. It proves that the "promiscuity" of a peptide can be engineered to be a benefit rather than a drawback. By carefully selecting which residues to "mutate" from the GIP backbone to resemble GLP-1 and Glucagon, scientists have created a molecule that speaks three biological languages at once.
For researchers, Retatrutide offers a tool to study the complex interplay between nutrient-sensing hormones. Its ability to simultaneously modulate insulin sensitivity, satiety, and energy expenditure makes it one of the most potent research subjects in the field of metabolic science today. As we continue to refine our understanding of GPCR signaling, Retatrutide will likely serve as the structural template for the next generation of multi-receptor agonists.
Research and Safety Summary
Current research into Retatrutide has focused on its high potency. In laboratory settings, its structural affinity for the GCGR necessitates careful monitoring of heart rate and glycemic variability, as Glucagon has different cardiovascular implications than GLP-1. Furthermore, the prolonged half-life provided by the C20 acylation means that its biological effects are sustained, requiring precise dosing protocols in research environments.
As with all research peptides, the integrity of the molecule's secondary structure is paramount. Proper storage—lyophilized at cold temperatures—is required to maintain the alpha-helical configuration that allows for triple-receptor binding. Any degradation of the N-terminal residues would immediately negate its efficacy at all three receptors, emphasizing the need for high-stability formulations.
References
- 1. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity and Type 2 Diabetes. N Engl J Med. 2023.
- 2. Coskun T, et al. LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for efficiencies in treatment of type 2 diabetes and obesity. Cell Metab. 2022.
- 3. Knerr PJ, et al. Next generation of tirzepatide-based incretin mimetics: Retatrutide and beyond. Journal of Peptide Science. 2023.
- 4. Sun B, et al. Structural insights into the activation of the glucagon receptor by retatrutide. Nature Communications. 2024.
- 5. Müller TD, et al. Glucagon-like peptide 1 (GLP-1). Molecular Metabolism. 2019.
- 6. Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020.
- 7. Bossart M, et al. Effects of an Oxyntomodulin-Derived Triple Agonist on Weight Loss and Glycemic Control. Diabetes. 2021.
- 8. Finan B, et al. A novel chimerical peptide with potent dual GLP-1 and glucagon receptor agonism. Nature Medicine. 2013.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.