Regulatory Shifts: Analyzing the Impact of FDA Compounding Lists on Research Peptide Availability
An in-depth analysis of how recent FDA compounding list updates (Category 2) impact the availability of research peptides like BPC-157 and Thymosin Alpha-1. This guide explores the scientific reasoning behind the regulations and what they mean for researchers.
Introduction: The Changing Landscape of Peptide Regulation
In the rapidly evolving world of biotechnology and biochemistry, the regulatory framework governing the synthesis and distribution of peptide compounds plays a critical role in shaping the research landscape. For years, peptides such as BPC-157 and Thymosin Alpha-1 existed in a unique niche, widely utilized in research settings to explore tissue repair, immune modulation, and cytoprotection. However, recent shifts by the U.S. Food and Drug Administration (FDA)—specifically regarding the categorization of bulk drug substances for compounding—have sent shockwaves through the industry.
The FDA's updates to the 503A and 503B compounding lists have fundamentally altered how certain peptides can be legally prepared by compounding pharmacies. While these regulations are primarily aimed at clinical administration and patient safety, their ripple effects are felt acutely in the research community. The reclassification of key peptides into "Category 2" (signaling safety concerns) or their removal from "Category 1" (allowed for compounding) has created confusion regarding availability, legality, and the future of peptide research.
This article aims to provide a comprehensive, scientifically grounded analysis of these regulatory shifts. We will explore the mechanisms of the FD&C Act sections 503A and 503B, analyze the specific scientific reasoning cited by the FDA for targeting compounds like BPC-157, and discuss the implications for researchers looking to source high-purity peptides for laboratory models. Understanding these distinctions is vital for maintaining compliant, rigorous research protocols in an era of heightened scrutiny.
Understanding the FDA Compounding Framework: 503A vs. 503B
To comprehend the current state of peptide availability, one must first understand the legal distinction between standard pharmaceutical manufacturing and pharmacy compounding. Under the Federal Food, Drug, and Cosmetic Act (FD&C Act), compounding was traditionally a localized practice intended to create tailored medications for patients with specific needs (e.g., removing an allergen from a formula). However, as the demand for peptides grew, the scale of compounding expanded, leading to tighter federal oversight.
Section 503A: Traditional Compounding
Section 503A generally regulates traditional community pharmacies. These entities prepare patient-specific prescriptions. To be exempt from the stringent new drug approval (NDA) requirements that major pharmaceutical companies face, 503A pharmacies must comply with specific rules, one of which restricts the bulk drug substances (active pharmaceutical ingredients) they can use. They are generally limited to substances that have a USP monograph, are components of FDA-approved drugs, or appear on the "503A Bulks List" developed by the FDA.
Section 503B: Outsourcing Facilities
Section 503B was established to regulate "outsourcing facilities"—larger scale operations that can compound drugs without patient-specific prescriptions essentially serving as a bridge between pharmacy and manufacturer. These facilities are subject to Current Good Manufacturing Practice (CGMP) requirements. Crucially, they can only compound with bulk substances that are on the "503B Bulks List" or are used in FDA-approved drugs (provided there are no safety issues). The FDA’s recent actions have largely focused on refining these lists, determining which substances are permitted and which are excluded.
The Category System: A Tool for Regulation
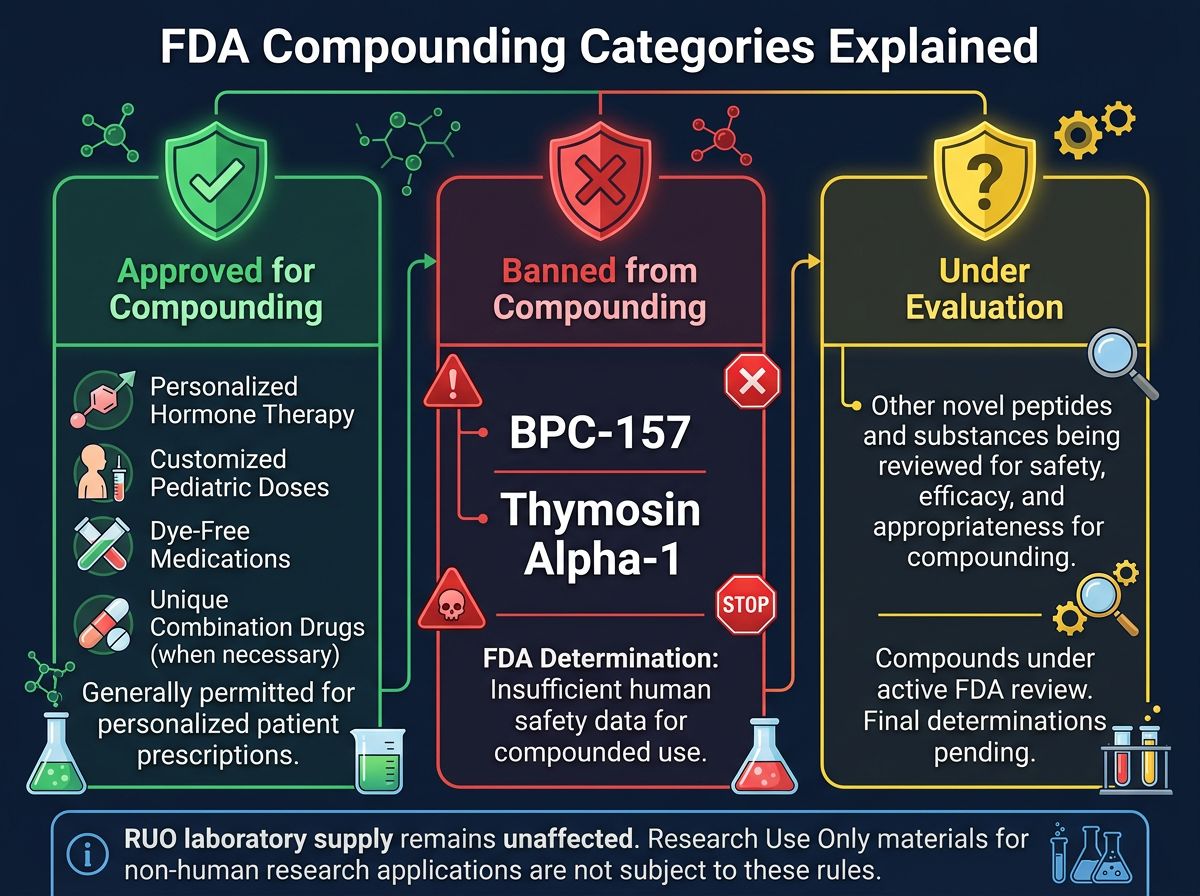
The FDA evaluates nominated bulk drug substances for the 503B list by placing them into one of three categories during the evaluation process. This categorization acts as the primary filter for what is currently accessible through clinical compounding channels.
- Category 1: Substances nominated with sufficient information for FDA evaluation, which the FDA has not yet identified as having significant safety concerns. These are typically under interim policy permitting their use.
- Category 2: Substances requiring an interim ban due to significant safety risks identified by the FDA. Compounding with these substances is prioritized for enforcement action.
- Category 3: Substances nominated without sufficient support information.
The seismic shift in the peptide community occurred when popular research targets were moved into Category 2. This effectively signaled that the FDA considers these compounds unsafe for human compounding, regardless of their popularity in clinical practice or potential therapeutic benefits suggest by preliminary studies.
The BPC-157 Controversy: From Cytoprotection to Category 2
Perhaps no peptide has faced greater scrutiny in recent regulatory updates than BPC-157 (Body Protection Compound-157). A pentadecapeptide derived from a protective protein found in human gastric juice, BPC-157 has been the subject of extensive animal research.
Mechanism of Action and Research Interest
Scientifically, BPC-157 is fascinating due to its proposed role in the "gut-brain axis" and tissue healing. Research by Sikiric et al. suggests that BPC-157 interacts with the nitric oxide (NO) system, promoting angiogenesis (blood vessel formation) and protecting endothelium. In animal models, it has demonstrated efficacy in healing tendon-to-bone injuries, repairing damaged muscle fibers, and mitigating gastric ulcers induced by NSAIDs.
Key research findings include:
- Angiogenic Modulation: Up-regulation of VEGFR2 expression, facilitating vascular repair.
- Anti-inflammatory Effects: Reduction of inflammatory markers in models of colitis and arthritis.
- Neuroprotection: Emerging evidence of neuroprotective effects in traumatic brain injury models.
The FDA's Safety Rationale
Despite the promising animal data, the FDA placed BPC-157 in Category 2. The agency's reasoning primarily revolves around the lack of rigorous human clinical trials and potential immunogenicity. In their definitions of safety risks, the FDA highlighted that peptides, due to their complexity, pose a risk of inducing an immune response (formation of anti-drug antibodies) that simple small molecules do not.
The FDA argued that there is insufficient data to determine if BPC-157 is safe for human injection. Without an approved New Drug Application (NDA) or a USP monograph, the agency concluded that the risks of compounding it outweighed the benefits, citing the absence of standardized biological activity assays and potential impurities in bulk substances.
Impact on Research Availability
It is critical to distinguish between clinical compounding and research supply. The FDA's move bans compounding pharmacies from prescribing BPC-157 to patients. However, it does not ban the synthesis of the molecule for in vitro or in vivo laboratory research. Companies like Alpha Carbon Labs can still supply BPC-157 for non-human research purposes. The regulation limits the "drug" aspect, not the "chemical" aspect used in scientific inquiry.
Thymosin Alpha-1: The Biologics Classification Challenge
Another major casualty of the regulatory updates is Thymosin Alpha-1 (Ta1). Unlike BPC-157, Thymosin Alpha-1 has a history of clinical approval in various countries (marketed as Zadaxin) and has been designated an orphan drug in the US for specific conditions. It is an endogenous peptide fragment that modulates the immune system by enhancing T-cell function.
The "Biologic" Distinction
The FDA's restriction on Thymosin Alpha-1 stems from a complex regulatory definition regarding "biologics." In March 2020, the definition of a biologic was statutorily adjusted. Certain proteins and peptides that were previously regulated as drugs began to fall under the umbrella of biologics. Under the Biologics Price Competition and Innovation Act (BPCIA), biologics generally cannot be compounded by outsourcing facilities unless specific, stringent criteria are met.
The FDA removed Thymosin Alpha-1 from the compounding lists partly because it was re-categorized as a biological product. Biologics are viewed as having higher complexity and greater manufacturing risks than small-molecule drugs. The FDA maintains that compiling biologics presents "demonstrable difficulties," essentially effectively removing them from the allowed list for 503A and 503B facilities.
Research Implications for Immunology
For researchers, Thymosin Alpha-1 remains a vital tool for studying immune viral responses and cancer immunotherapy adjuncts. Its mechanism involves:
- TLR Modulation: Activating Toll-like receptors (TLRs) in dendritic cells.
- MHC Class I Upregulation: Increasing the visibility of infected cells to cytotoxic T-cells.
- Cytokine Balance: Modulating IL-2 and interferon production.
Because the restriction targets compounding for human use, the availability of high-purity Thymosin Alpha-1 for flow cytometry, cell culture studies, and animal models remains legal, provided it is sourced from chemical suppliers rather than compounding pharmacies. This drives home the importance of verifying COA documents to ensure the research material has not been degraded or misrepresented as a result of market shifts.
The Scope of Affected Peptides: Beyond BPC-157
While BPC-157 and Thymosin Alpha-1 grabbed headlines, the regulatory net cast by the FDA affects a broader spectrum of research peptides. The agency often groups peptides by mechanism or structural complexity. Researchers should be aware of the status of several other compounds.
| Peptide | Proposed Mechanism | Regulatory/Research Status |
|---|---|---|
| AOD9604 | Lipolytic fragment of HGH (C-terminus). | Often scrutinized alongside other HGH fragments. FDA has raised similar safety concerns regarding lack of human data. |
| MK-677 (Ibutamoren) | Ghrelin mimetic / HGH Secretagogue. | Technically a non-peptide small molecule, but often grouped in enforcement. FDA warns against its use for bodybuilding due to potential adverse effects (heart failure risk). |
| CJC-1295 | GHRH Analog. | Frequently examined for its long half-life. Remains popular in research models for metabolic studies but faces compounding headwinds. |
| TB-500 (Thymosin Beta-4) | Actin sequestration / Tissue Repair. | Subject to similar "biologics" arguments as Thymosin Alpha-1. Often cited in regenerative medicine research. |
| Kisspeptin-10 | GnRH secretion stimulator. | Used in reproductive research. Less enforcement visibility than BPC-157 but falls under similar scrutiny of bulk substances. |
The GLP-1 Anomaly: Shortages and Exemptions
An interesting divergence in the regulatory landscape involves GLP-1 agonists like Semaglutide and Tirzepatide. Unlike the research peptides mentioned above, these molecules have full FDA approval as drugs (under brand names like Ozempic, Wegovy, and Mounjaro).
However, under the FD&C Act, if an approved drug is listed on the FDA's "Drug Shortages List," compounding pharmacies are permitted to compound copies of the drug to meet public demand. This creates a unique dynamic where Semaglutide is widely compounded legally, while unapproved research peptides like BPC-157 are restricted.
For researchers, this distinction is crucial. It highlights that the FDA's primary lever of control is the approval status and shortage status of the compound. For non-approved molecules (like BPC-157), there is no "shortage" exemption because there is no approved drug to be short of. This relegates these compounds almost exclusively to the domain of "Research Use Only" (RUO) suppliers.
Research Use Only (RUO): The Lifeline for Scientific Inquiry
The term "Research Use Only" is not merely a label; it is a regulatory categorization that defines the intended use of a substance. In the wake of the crackdown on compounding pharmacies, the role of RUO suppliers has become paramount for the scientific community.
When the FDA restricts a substance from compounding, they are restricting its preparation for clinical use (treatment of humans). They do not restrict the sale of the chemical for discovery, toxicity testing, or basic chemical analysis. This ensures that scientific progress is not halted by clinical safety regulations.
Navigating the Grey Market vs. Legitimate Research Supply
With compounding pharmacies exiting the market for peptides like BPC-157, a vacuum has been created. This inevitably leads to the rise of grey-market vendors selling directly to consumers under the guise of "research." For legitimate laboratory professionals, distinguishing between a compliant chemical supplier and a grey-market vendor is essential.
Hallmarks of a Legitimate RUO Supplier:
- Strict adherence to labeling: Products are explicitly labeled "Not for Human Consumption."
- Domestic Quality Control: Utilization of HPLC (High-Performance Liquid Chromatography) and MS (Mass Spectrometry) to verify purity.
- Transparency: Availability of up-to-date peptide synthesis data and Certificate of Analysis (COA) documents.
- No Medical Claims: Legitimate suppliers do not offer dosing protocols for humans or make therapeutic claims.

The Science of Safety: Immunogenicity and Impurities
To understand the FDA's aggressive stance, one must look closely at the biochemistry of peptide synthesis. The FDA often cites "immunogenicity" as a primary risk. What does this mean in a research context?
Peptide-Related Impurities
During solid-phase peptide synthesis (SPPS), various impurities can arise, including:
- Deletion Sequences: Peptides missing one or more amino acids.
- Insertion Sequences: Peptides with extra amino acids.
- Racemization: Conversion of L-amino acids to D-amino acids, altering structure and function.
- Oxidation: Particularly in peptides containing Methionine or Cysteine.
Even a 99% pure peptide contains 1% impurities. If that impurity is a truncation that acts as an antigen, it could theoretically trigger an immune response in a test subject. In a clinical setting, this can lead to anaphylaxis or the neutralization of endogenous proteins (e.g., antibodies against native erythropoietin causing pure red cell aplasia).
The FDA argues that compounding pharmacies lack the robust characterization methods required to detect these subtle, complex impurities. For researchers, this underlines the necessity of quality control. When conducting animal studies, using a peptide with high impurity levels can introduce confounding variables, rendering data useless.
Future Perspectives: The Path Forward for Peptide Research
The regulatory shifts we are witnessing are likely the beginning, not the end, of a tighter control framework for bioactive molecules. As the popularity of peptides grows, so too will the oversight.
The Need for Data
The only way for compounds like BPC-157 to eventually leave the regulatory "penalty box" and enter legitimate clinical use is through the traditional Investigational New Drug (IND) and NDA pathway. This requires massive investment in Phase I, II, and III clinical trials. Until that data exists, these compounds will remain research chemicals.
Continued Innovation
Despite regulations, innovation continues. Researchers are exploring modified versions of these peptides to improve stability and reduce immunogenicity. For example, the development of molecules like PT-141 (Bremelanotide), which successfully transitioned from a research peptide to an FDA-approved drug, shows that the pathway is passable with sufficient evidence.
Furthermore, attention is shifting toward mitochondrial peptides like SS-31 and MOTS-c. These represent a new frontier in metabolic research, and their regulatory status will likely depend on the lessons learned from the BPC-157 and Thymosin Alpha-1 sagas.
Conclusion
The FDA's recent updates to the compounding lists have created a bifurcated world for peptides. On one side, clinical access to compounds like BPC-157 and Thymosin Alpha-1 has been severely restricted due to valid, albeit conservative, safety concerns regarding immunogenicity and synthesis complexity. On the other side, the world of basic science remains open.
For the research community, these "Regulatory Shifts" are a call to action for higher standards. The era of assuming all peptides are equal is over. To generate valid, reproducible data in a compliant manner, researchers must rely on suppliers who prioritize rigorous analytical testing and adherence to scientific standards. While the regulatory hurdles for clinical use are high, the potential for discovery in the laboratory remains boundless.
Alpha Carbon Labs is committed to supporting the scientific community with high-purity research peptides, strictly for laboratory use and non-human research applications.
References
- 1. Food and Drug Administration. (2023). Safety Risks Associated with Certain Bulk Drug Substances Nominated for Use in Compounding.
- 2. Sikiric, P., et al. (2010). Focus on the cytoprotective relation of BPC 157 to the underlying mechanism and the relevance of the gut-brain axis. Journal of Physiology and Pharmacology, 61(6), 703-713.
- 3. Matteucci, C., et al. (2017). Thymosin alpha 1: an endogenous regulator of inflammation, immunity, and tolerance. Annals of the New York Academy of Sciences, 1397(1), 29-38.
- 4. Food and Drug Administration. (2018). Evaluation of Bulk Drug Substances Nominated for Use in Compounding Under Section 503B of the Federal Food, Drug, and Cosmetic Act.
- 5. Gwenn, S. M., et al. (2014). Immunogenicity of therapeutic proteins: Clinical implications and future prospects. Clinical and Translational Science, 7(3), 272-279.
- 6. Vukojevic, J., et al. (2022). Pentadecapeptide BPC 157 and the central nervous system. Neural Regeneration Research, 17(3), 482.
- 7. Costantini, E., et al. (2019). Peptide therapeutics: Current status and future directions. Drug Discovery Today, 24(9), 1738-1747.
- 8. Pietramaggiori, G., et al. (2016). Thymosin beta-4 modulates the expression of senescence markers in diabetic mice. Biochemical and Biophysical Research Communications, 477(4), 947-952.
- 9. Lau, J. L., & Dunn, M. K. (2018). Therapeutic peptides: Historical perspectives, current development trends, and future directions. Bioorganic & Medicinal Chemistry, 26(10), 2700-2707.
- 10. Sei, Y., et al. (2020). Brief Review of BPC-157 and Its Potential Therapeutic Applications. Current Pharmaceutical Design, 26.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.