Optimizing Experimental Reproducibility: Technical Criteria for Selecting NAD+ for Long-Term Longitudinal Research
A technical analysis of the chemical stability and selection criteria for NAD+ in longitudinal research. This guide covers lyophilization standards, purity metrics, and protocols for optimizing experimental reproducibility.
Introduction: The Reproducibility Crisis and Reagent Integrity
In the domain of metabolic research, few molecules have garnered as much recent attention as Nicotinamide Adenine Dinucleotide (NAD+). Fundamental to cellular respiration, DNA repair, and sirtuin activation, NAD+ flux is a critical variable in studies ranging from neurodegeneration to mitochondrial biogenesis. However, as the scope of NAD+ research expands into long-term longitudinal studies—experiments tracking subjects over extended periods to observe aging or chronic disease progression—the technical challenge of experimental reproducibility becomes acute.
Reproducibility in life sciences often hinges on variables that are frequently overlooked: reagent stability, batch-to-batch consistency, and the precise chemical form of the compound administered. For researchers utilizing NAD+, the selection of the substrate is not merely a purchasing decision; it is a critical experimental parameter. The inherent chemical reactivity of the dinucleotide structure renders it susceptible to hydrolysis and degradation, potentially introducing variance that can obfuscate subtle longitudinal data trends.
This article provides a rigorous technical analysis of the criteria required for selecting NAD+ for high-quality research. We will explore the biochemistry of NAD+ stability, the comparative advantages of various precursors versus direct NAD+ application, and the strict quality control protocols necessary to ensure data integrity over months or years of experimentation.
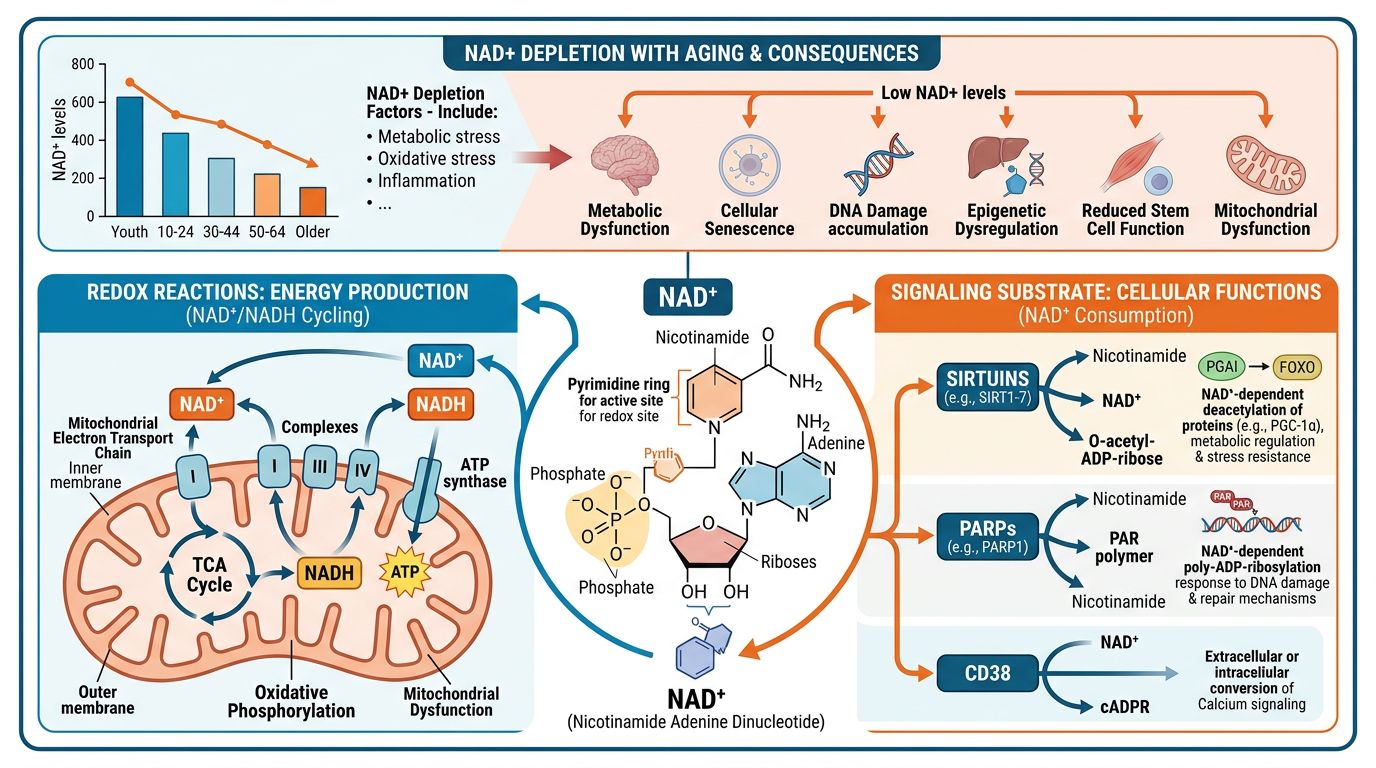
The Biochemistry of NAD+: Structure and Instability
To understand the stability challenges associated with NAD+, one must first examine its molecular architecture. NAD+ consists of two nucleotides joined through their phosphate groups by a phosphoanhydride bond. One nucleotide contains an adenine base and the other a nicotinamide base. The critical feature regarding stability is the glycosidic bond linking the nicotinamide moiety to the ribose sugar.
Hydrolysis and Degradation Pathways
In aqueous solution, NAD+ is thermodynamically unstable. The primary degradation pathway is the hydrolysis of the bond between the nicotinamide ring and the ribose. This reaction is catalyzed by both acidic and alkaline conditions, following a specific pH-rate profile. Furthermore, the phosphoanhydride bond is susceptible to cleavage, particularly in the presence of heat or specific divalent cations.
For longitudinal studies where stock solutions might be prepared in bulk or stored for extended durations, this instability presents a significant confounding variable. If the concentration of active NAD+ in a treatment solution decreases by 10% over the course of a 6-week protocol, the resulting data may reflect dosing variance rather than biological reality. This necessitates a rigid adherence to proper synthesis and lyophilization standards to preserve the molecule until the moment of use.
The "M" Shaped Stability Curve
Research indicates that NAD+ exhibits maximum stability at a slightly acidic pH (typically around pH 2.0 to 4.0 for short-term liquid handling), whereas it degrades rapidly in alkaline environments. Conversely, NADH (the reduced form) is unstable in acid and stable in base. This dichotomy complicates the handling of samples meant to measure the NAD+/NADH ratio, but for the administration of exogenous NAD+, maintaining the oxidized form's integrity is the priority.
Technical Criteria for Selecting NAD+ Reagents
When sourcing NAD+ for serious research, "purity" is an insufficient metric on its own. Researchers must evaluate the material based on a triad of factors: Lyophilization quality, Salt Stoichiometry, and Contaminant Profile.
1. Lyophilization and Hygroscopy
NAD+ is highly hygroscopic. Absorbed moisture initiates hydrolysis even in the solid state. High-quality research-grade NAD+ must be lyophilized under precise conditions to remove residual water content to below 1-2%. The physical appearance of the "cake" can be an indicator effectively; a collapsed or "melted" cake often suggests moisture ingress or improper freeze-drying cycles. Storage in desiccated, -20°C (or -80°C for long term) environments is non-negotiable for preserving the integrity of the reagent over a longitudinal timeline.
2. Purity and HPLC Verification
For longitudinal animal models or cell culture systems, purity should be verified via High-Performance Liquid Chromatography (HPLC). A standard of < 98% purity poses risks, not only due to the lower concentration of the active agent, but due to the presence of degradation products like nicotinamide (NAM) and ADP-ribose, which are biologically active. High levels of NAM, for instance, act as a feedback inhibitor of PARP enzymes and sirtuins, potentially inverting the expected results of the experiment. Reviewing COA documents for every batch is essential to track these impurities.
3. Endotoxin Controls
In vivo studies, particularly those involving injection, are sensitive to bacterial endotoxins (lipopolysaccharides). Low-quality NAD+ preparations may contain endotoxin residues from the fermentation or synthesis purification process. Endotoxins trigger potent immune responses that can mask metabolic effects or induce confounding inflammation variables in longevity studies.
Comparative Analysis: Direct NAD+ vs. Precursors
A common dilemma for principal investigators is choosing between direct NAD+ and its precursors, such as Nicotinamide Mononucleotide (NMN) or Nicotinamide Riboside (NR). While precursors have better oral bioavailability, direct NAD+ administration (via IP, IV, or subcutaneous routes) allows for precise control over plasma concentrations, bypassing rate-limiting salvage pathway enzymes like NAMPT.
| Compound | Transport Mechanism | Stability in Solution | Primary Research Application |
|---|---|---|---|
| NAD+ | Extracellular signaling (P2X7 receptors), Connexin 43 channels | Low (requires fresh reconstitution) | Direct elevation of extracellular NAD+, neuroprotection, mitochondrial redox support. |
| NMN | Slc12a8 transporter (tissue specific) | Moderate | Intracellular NAD+ boosting, specifically in tissues expressing Slc12a8 (e.g., gut, kidney). |
| NR | Nucleoside transporters (ENTs) | Moderate to Low | Oral supplementation models, assessing salvage pathway efficiency. |
For studies focused on immediate mitochondrial resuscitation or specific extracellular signaling pathways, direct NAD+ is often the superior choice. Additionally, integrating NAD+ with mitochondrial peptides like SS-31 (Elamipretide) or MOTS-c allows researchers to target mitochondrial function from dual angles: electron transport chain efficiency (SS-31) and metabolic regulation (MOTS-c), alongside substrate availability (NAD+).
Integrating NAD+ with Mitochondrial Peptides
Longitudinal studies are increasingly moving towards combinatorial therapies to address the multifactorial nature of aging and metabolic decline. NAD+ does not exist in a vacuum; its efficacy is often tethered to the functional state of the mitochondrial proteome.
Synergy with SS-31 and MOTS-c
Research suggests that while NAD+ fuels the sirtuin pathways and PARPs, the physical integrity of the inner mitochondrial membrane (IMM) determines the efficiency of the Electron Transport Chain (ETC). SS-31 accumulates in the IMM and binds to cardiolipin, stabilizing cristae structure. In protocols examining energetic decline, the co-administration of NAD+ and SS-31 represents a comprehensive approach: one agent repairs the "machinery" (SS-31) while the other provides the "fuel" (NAD+).
Similarly, MOTS-c, a mitochondrial-derived peptide, acts as a metabolic regulator, influencing insulin sensitivity and fatty acid oxidation. Pairing MOTS-c with NAD+ can simulate a powerful exercise-mimetic effect, useful in obesity or sarcopenia models managed over long durations.
Metabolic Modulators: 5-amino-1mq and GLP-1s
Another angle of NAD+ research involves preventing its degradation. The enzyme NNMT (nicotinamide N-methyltransferase) acts as a sink for nicotinamide, reducing the pool available for NAD+ salvage. The small molecule 5-amino-1mq is an NNMT inhibitor. By inhibiting this enzyme, researchers can theoretically raise intracellular NAD+ levels endogenously. Protocols comparing exogenous NAD+ administration versus NNMT inhibition (via 5-amino-1mq) are gaining traction in obesity research.
Furthermore, in the context of diabetic research, combining NAD+ with GLP-1 agonists like Semaglutide or dual agonists like Tirzepatide allows for the decoupling of weight-loss effects from direct mitochondrial energetics. While Semaglutide drives weight loss through satiety and insulin pathways, NAD+ supports the cellular energy maintenance required during varying metabolic states.
Stability Protocols for the Lab
To ensure longitudinal data validity, the handling of NAD+ must be standardized. Below is a recommended protocol for research laboratories utilizing lyophilized NAD+.
Reconstitution and Storage
- Solvent Selection: Reconstitute lyophilized NAD+ in bacteriostatic water or sterile PBS immediately prior to use. Avoid storing dissolved NAD+ for longer than 48 hours, even at 4°C.
- Aliquot Strategy: If a bulk solution must be made, it should be immediately aliquoted into single-use variances and frozen at -80°C. Do not freeze-thaw. Crystallization and thawing shear can accelerate hydrolysis upon re-warming.
- pH Monitoring: Ensure the buffering capacity of the solvent matches the physiological target. For cell culture comparisons, verify that the addition of NAD+ (which can be acidic depending on the salt form) does not shift the culture media pH significantly.
Validation Checks
Researchers should independently verify the concentration of their NAD+ stock periodically. A simple optical density measurement at 260 nm (Adenine peak) is insufficient as it does not distinguish between NAD+, NADH, and ADP-Ribose. An enzymatic cycling assay or HPLC-UV is required to confirm that the substrate remains oxidized and intact.
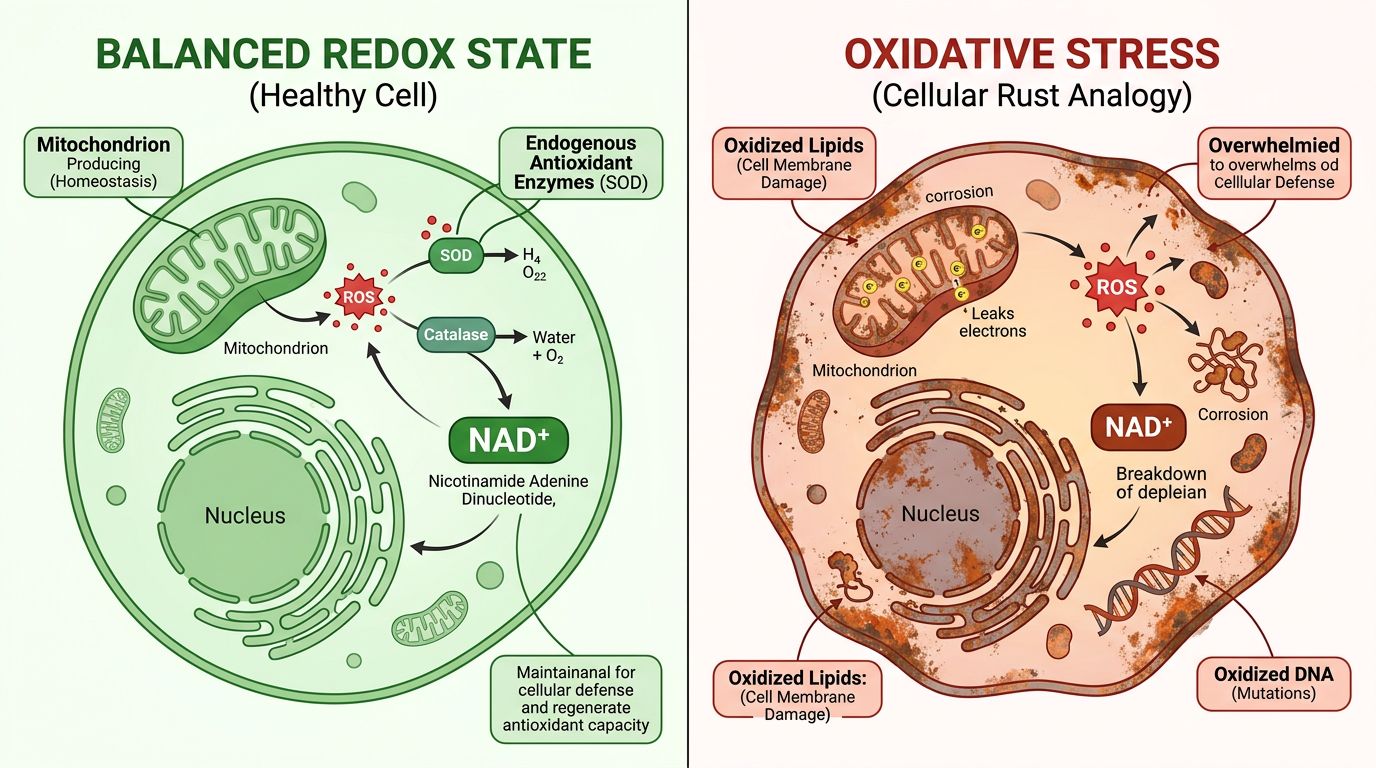
Advanced Applications: Neuroprotection and Longevity
The neuroprotective capacity of NAD+ is a primary focus of longitudinal studies. The brain consumes a disproportionate amount of glucose and oxygen, relying heavily on stable NAD+ levels for neuronal survival. In models of Alzheimer's or Parkinson's, NAD+ depletion is an early hallmark of pathology.
Long-Term Administration in Murine Models
In studies extending beyond 12 months in mice, the method of delivery becomes a variable. Continuous infusion via osmotic pumps offers steady-state levels but presents surgical variables. Daily subcutaneous injections mimic pulsatile clinical therapies. In these designs, the stability of the NAD+ supply over the 12-month procurement period is vital. Utilizing a supplier with robust quality control ensures that the NAD+ injected in month 11 is chemically identical to that in month 1.
Synergistic Neuroprotection
Researchers exploring cognitive decline often combine NAD+ with neurotrophic peptides like Semax or Selank, or peptides that influence telomerase activity such as Epithalon. Epithalon (Epithalamin) has been shown in longitudinal studies to influence lifespan and melatonin secretion. A dual-arm study comparing NAD+ monotherapy vs. NAD+ plus Epithalon could reveal whether mitochondrial support combined with telomere maintenance offers additive benefits.
Conclusion: The Foundation of Reliable Data
As the scientific community delves deeper into the mechanisms of aging and metabolism, the fidelity of our tools must rise to meet the complexity of our questions. NAD+, while a molecule of immense potential, is chemically fragile. Its successful application in long-term longitudinal research requires more than just biological curiosity; it demands chemical vigilance.
By selecting high-purity, properly lyophilized NAD+, validating its integrity through rigorous analytical methods, and storing it with respect for its instability, researchers can eliminate reagent degradation as a confounding variable. Whether investigating the synergy with SS-31 in heart failure models or exploring the metabolic interplay with Semaglutide, the reproducibility of experimental outcomes begins with the quality of the peptide and small molecule inputs.
Alpha Carbon Labs remains committed to providing researchers with the analytical transparency and synthesis quality required for these exacting standards. Through detailed COA verification and advanced synthesis technologies, we support the rigorous exploration of cellular metabolism's most vital molecule.
References
- 1. Canto, C., Menzies, K. J., & Auwerx, J. (2015). NAD+ Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metabolism, 22(1), 31–53.
- 2. Yoshino, J., Baur, J. A., & Imai, S. (2018). NAD+ Intermediates: The Biology and Therapeutic Potential of NMN and NR. Cell Metabolism, 27(3), 513–528.
- 3. Covarrubias, A. J., Perrone, R., Grozio, A., & Verdin, E. (2021). NAD+ metabolism and its roles in cellular processes during ageing. Nature Reviews Molecular Cell Biology, 22(2), 119–141.
- 4. Ratajczak, J., et al. (2016). NRK1 controls nicotinamide mononucleotide and nicotinamide riboside metabolism in mammalian cells. Nature Communications, 7, 13103.
- 5. Trammell, S. A., et al. (2016). Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nature Communications, 7, 12902.
- 6. Camacho-Pereira, J., et al. (2016). CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metabolism, 23(6), 1127–1139.
- 7. Liu, L., et al. (2018). Quantitative Analysis of NAD Synthesis-Breakdown Fluxes. Cell Metabolism, 27(5), 1067–1080.
- 8. Sauve, A. A. (2008). NAD+ and Vitamin B3: From Metabolism to Therapies. Journal of Pharmacology and Experimental Therapeutics, 324(3), 883–893.
- 9. Braidy, N., et al. (2011). Age Related Changes in NAD+ Metabolism Oxidative Stress and Sirt1 Activity in Wistar Rats. PLoS ONE, 6(4), e19194.
- 10. Guo, C., et al. (2020). NAD+ administration attenuates cognitive impairment in aged mice. Aging (Albany NY), 12(11), 10565–10581.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.