Metabolic Synergy: Investigating the Combined Effects of NAD+ and 5-amino-1mq on Lipid Oxidation
This article investigates the synergistic potential of combining NAD+ with the NNMT inhibitor 5-amino-1mq to accelerate lipid oxidation. We explore the biochemical mechanisms, the salvage pathway, and the impact on mitochondrial flux in adipocytes.
Introduction: The Metabolic Flux Dilemma
In the landscape of metabolic research, the regulation of adipose tissue extends far beyond simple caloric storage. White adipose tissue (WAT) acts as a dynamic endocrine organ, influencing systemic energy homeostasis, insulin sensitivity, and inflammatory cascades. A central challenge in modern metabolic research is addressing the "metabolic brake" phenomenon—whereby cellular mechanisms inherently resist the oxidation of stored lipids despite energetic deficits. This resistance is often driven by the depletion of essential coenzymes and the upregulation of savage pathways that divert substrates away from energy production.
Two distinct agents have emerged as critical tools for modulating this energetic landscape: Nicotinamide Adenine Dinucleotide (NAD+) and the small molecule inhibitor 5-amino-1mq. While investigated individually for their roles in cellular longevity and obesity resistance respectively, their combined application represents a theoretically synergistic approach to accelerating adipocyte metabolic flux. By inhibiting the "leak" in the NAD+ salvage pathway via 5-amino-1mq and simultaneously bolstering the coenzyme pool with exogenous NAD+, researchers can potentially unlock a magnified rate of mitochondrial lipid oxidation.
This article provides a comprehensive analysis of the biochemical mechanisms underpinning this combination, exploring the Nicotinamide N-methyltransferase (NNMT) pathway, the role of sirtuins in beta-oxidation, and the experimental implications of this dual-modality approach. As a leading supplier of research compounds, Alpha Carbon Labs is committed to exploring the high-level science behind these molecules, ensuring that experimental designs are informed by rigorous biochemical logic and the highest standards of peptide synthesis and chemical manufacturing.
The Nicotinamide N-methyltransferase (NNMT) Pathway: The Metabolic Brake
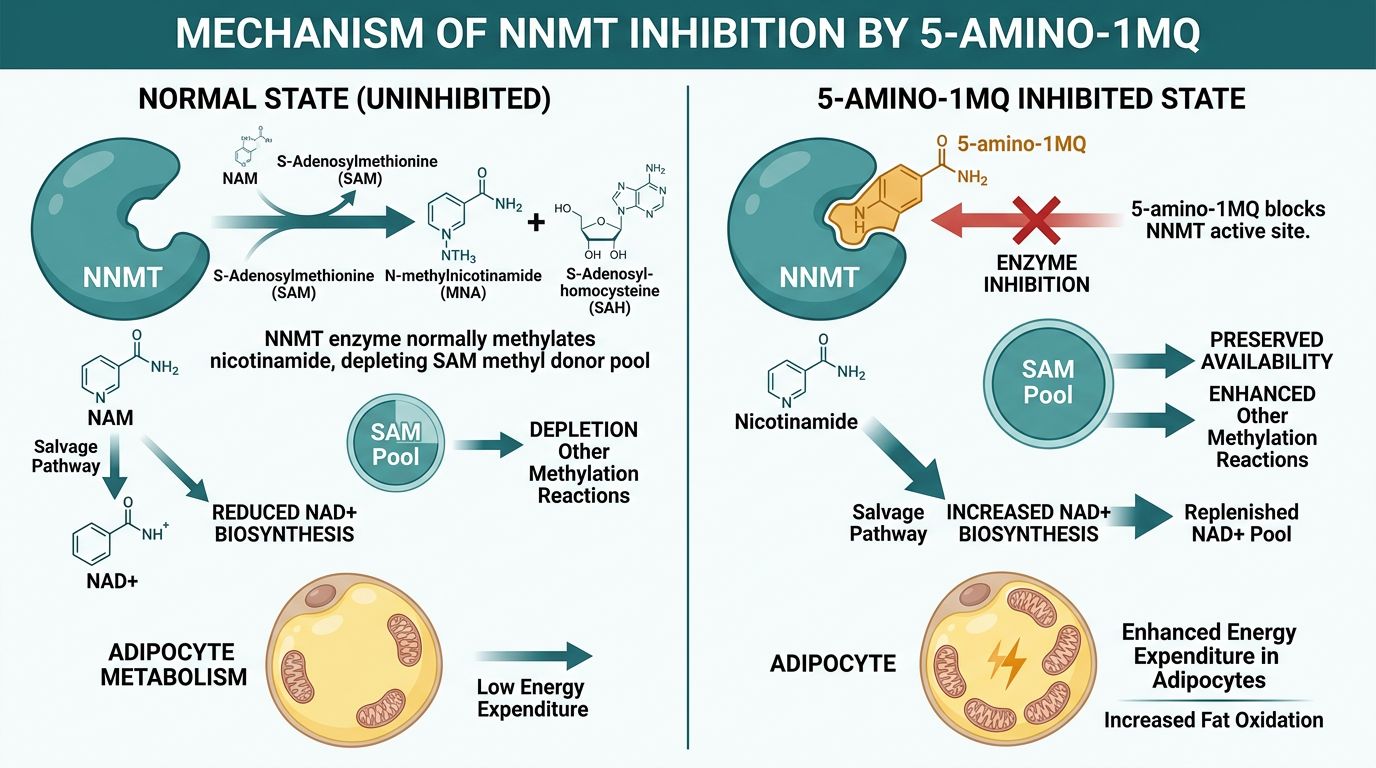
To understand the synergy between 5-amino-1mq and NAD+, one must first characterize the metabolic bottleneck they address: the enzyme Nicotinamide N-methyltransferase (NNMT). NNMT is a cytosolic enzyme that catalyzes the methylation of nicotinamide (NAM) using S-adenosylmethionine (SAM) as a methyl donor. This reaction produces 1-methylnicotinamide (1-MNA) and S-adenosylhomocysteine (SAH).
In the context of healthy physiology, NNMT serves to regulate excess nicotinamide levels and manage methyl group balance. However, in states of obesity and metabolic dysfunction, NNMT expression is often drastically upregulated in adipose tissue and the liver. This upregulation has two catastrophic consequences for cellular metabolism:
- Depletion of the NAD+ Salvage Pool: Nicotinamide (NAM) is the primary precursor for the regeneration of NAD+ via the salvage pathway. By methylating NAM into 1-MNA, NNMT effectively removes it from the pool of available precursors. 1-MNA cannot be converted back into NAD+; it is excreted in urine. This creates a "sink" that drains cellular NAD+ levels.
- Alteration of the Epigenetic Landscape: By consuming SAM (the universal methyl donor), high NNMT activity reduces the SAM:SAH ratio. This inhibits other methyltransferases responsible for DNA and histone methylation, leading to epigenetic shifts that favor adipogenesis (fat creation) and fibrosis while suppressing genes related to energy expenditure.
Research has identified NNMT as a critical link between adipose tissue accumulation and systemic metabolic decline. Mice that overexpress NNMT mimic the phenotype of diet-induced obesity even without excessive caloric intake, while NNMT knockout models are protected against high-fat diet-induced weight gain. This identifies NNMT as a high-value target for therapeutic intervention.
5-amino-1mq: Precision Inhibition of NNMT
5-amino-1mq (5-amino-1-methylquinolinium) represents a breakthrough in metabolic pharmacology. It is a membrane-permeable small molecule designed specifically to inhibit the catalytic activity of NNMT. Structurally, it mimics the quinolinium core of the enzyme's substrates, allowing it to bind to the active site and prevent the methylation of nicotinamide.
The primary mechanism of action for 5-amino-1mq involves the competitive inhibition of NNMT. By blocking this enzyme, 5-amino-1mq achieves several downstream effects documented in preclinical literature:
- Restoration of Intracellular NAD+: By preventing the conversion of NAM to 1-MNA, the salvage pathway is forced to recycle NAM back into NAD+. This leads to a spontaneous rise in intracellular NAD+ concentrations within the adipocyte.
- Enhanced Mitochondrial Function: Higher NAD+ levels fuel the electron transport chain and activate sirtuins (specifically SIRT1), which deacetylate and activate PGC-1alpha, the master regulator of mitochondrial biogenesis.
- Reduction of Adipocyte Size: In murine models, administration of 5-amino-1mq resulted in a significant reduction in adipocyte volume and a 30% reduction in white adipose tissue mass without changes in food intake.
For researchers, ensuring the structural fidelity of this molecule is paramount. Alpha Carbon Labs provides access to COA documents verifying the purity of 5-amino-1mq, ensuring that inhibition studies are not compromised by synthesis byproducts.
NAD+: The Essential Coenzyme of Lipid Oxidation
While 5-amino-1mq stops the "leak," NAD+ (Nicotinamide Adenine Dinucleotide) represents the "fuel." NAD+ is the central coenzyme of metabolism, required for the enzymatic reactions of glycolysis, the Krebs cycle (TCA cycle), and fatty acid beta-oxidation. It accepts electrons to become NADH, which then donates them to the electron transport chain to generate ATP.
However, NAD+ is not merely an electron carrier; it is also a consumed substrate for signaling enzymes, most notably the Sirtuins (SIRT1-7) and PARPs (Poly-ADP-ribose polymerases). As organisms age or undergo metabolic stress, systemic NAD+ levels decline precipitously. This decline is exacerbated in adipose tissue by the aforementioned upregulation of NNMT.
When NAD+ levels drop, the activity of SIRT1 plummets. SIRT1 is crucial for lipid metabolism because it deacetylates Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha (PGC-1alpha). Active PGC-1alpha turns on the genes required for fatty acid transport (CPT1) and oxidation. Therefore, low NAD+ equals low SIRT1 activity, which equals stalled fat burning.
Direct vs. Precursor Application
Research has historically focused on precursors like NMN or NR. However, direct application of oxidized NAD+ is gaining traction in research settings due to findings regarding extracellular transport mechanisms (e.g., Connexin 43 channels) and its immediate availability for redox reactions upon cellular entry. Combining exogenous NAD+ with NNMT inhibition theoretically bypasses the rate-limiting steps of precursor conversion while simultaneously preventing the rapid degradation of the newly introduced coenzyme.
The Synergistic Mechanism: Plug the Hole, Fill the Bucket
The core hypothesis supporting the combined use of NAD+ and 5-amino-1mq lies in the concept of metabolic synergy. Used in isolation, each compound faces a limiting factor:
- NAD+ alone: If NNMT activity is high (as in obese adipocytes), a significant portion of the supplemented NAD+ (or its salvageable breakdown products) will be methylated and excreted as 1-MNA. The "bucket" has a hole.
- 5-amino-1mq alone: While it prevents the loss of NAM, it relies on the cell's existing salvage machinery to rebuild NAD+ stocks. If the basal pool is already severely depleted due to chronic metabolic stress, the recovery of optimal NAD+ levels may be slow.
The Combined Effect: By administering 5-amino-1mq, the researcher inhibits the NNMT "sink." Concurrently administering NAD+ saturates the salvage pathway. Because the exit route (methylation) is blocked, the exogenous NAD+ is funneled almost exclusively toward energetic and signaling pathways.
Impact on Mitochondrial Beta-Oxidation
The convergence of these pathways results in a profound upregulation of mitochondrial fatty acid beta-oxidation. The mechanism proceeds as follows:
- 5-amino-1mq raises endogenous NAM; Exogenous NAD+ increases total pool size.
- Intracellular NAD+/NADH ratios shift heavily toward the oxidized state (NAD+).
- High NAD+ concentrations activate SIRT1.
- SIRT1 activates PGC-1alpha via deacetylation.
- PGC-1alpha upregulates Carnitine Palmitoyltransferase I (CPT1), the rate-limiting enzyme that shuttles long-chain fatty acids into the mitochondria.
- Simultaneously, ample NAD+ is available to serve as the electron acceptor for the dehydrogenases involved in the beta-oxidation spiral.
This creates a "furnace" effect where the machinery for burning fat (mitochondria/enzymes) is upregulated, and the fuel for that machinery (NAD+) is abundant.
Comparative Analysis: NNMT Inhibition vs. Traditional Targets
To contextualize 5-amino-1mq within the broader spectrum of metabolic research agents, it is useful to compare its mechanism against other popular peptides available at Alpha Carbon Labs, such as Semaglutide and AOD9604. While all affect body composition, their routes of administration and mechanisms differ fundamentally.
| Compound | Primary Mechanism | Primary Target | Role of NAD+ Synergy |
|---|---|---|---|
| 5-amino-1mq | Enzyme Inhibition (NNMT) | Adipocytes, Liver | High: Directly rescues NAD+ salvage pathway; enhances basal metabolic rate. |
| Semaglutide | GLP-1 Receptor Agonism | Pancreas, Brain (Hypothalamus) | Moderate: Improves insulin sensitivity, but primarily drives weight loss via satiety and delayed gastric emptying. |
| AOD9604 | Lipolysis Stimulation (HGH Fragment) | Adipocytes (Beta-3 receptors) | Low/Neutral: mechanically liberates fatty acids but does not inherently upregulate the oxidative machinery to burn them. |
| MOTS-c | Mitochondrial Derived Peptide | Folate Cycle / AICAR pathway | High: Also targets metabolic flexibility, potentially distinct but complementary to NNMT inhibition. |
This comparison highlights the unique position of 5-amino-1mq. Unlike GLP-1 agonists which largely act centrally to reduce intake, 5-amino-1mq acts peripherally to increase energy expenditure (flux). This makes it an ideal candidate for "fat browning" research or correcting metabolic deceleration.
Research Findings and Preclinical Data
The foundational study by Neelakantan et al. (2018) provided the proof of concept for 5-amino-1mq. In diet-induced obese mice, treatment with the inhibitor resulted in a 30% loss of body weight over 10 days, primarily from fat mass, without altering food intake. Key biomarkers observed included:
- Plasma Lipid Profiles: Significant reductions in total cholesterol and triglycerides.
- Adipokine Profile: Inhibition of NNMT reversed the inflammatory profile of WAT, reducing cytokine expression.
- Muscle Anabolism: Surprisingly, the preservation of the SAM pool led to enhanced muscle satellite cell differentiation. This suggests 5-amino-1mq may prevent the muscle wasting (sarcopenia) often associated with rapid weight loss.
When considering NAD+ supplementation, studies by Trammell et al. and others have demonstrated that systemic elevation of NAD+ improves mitochondrial function in aged muscle and liver tissue. The theoretical intersection—using 5-amino-1mq to lower the threshold for NAD+ efficacy—is currently a frontier in metabolic research, suggesting that lower doses of NAD+ might achieve higher efficacy when the NNMT sink is plugged.
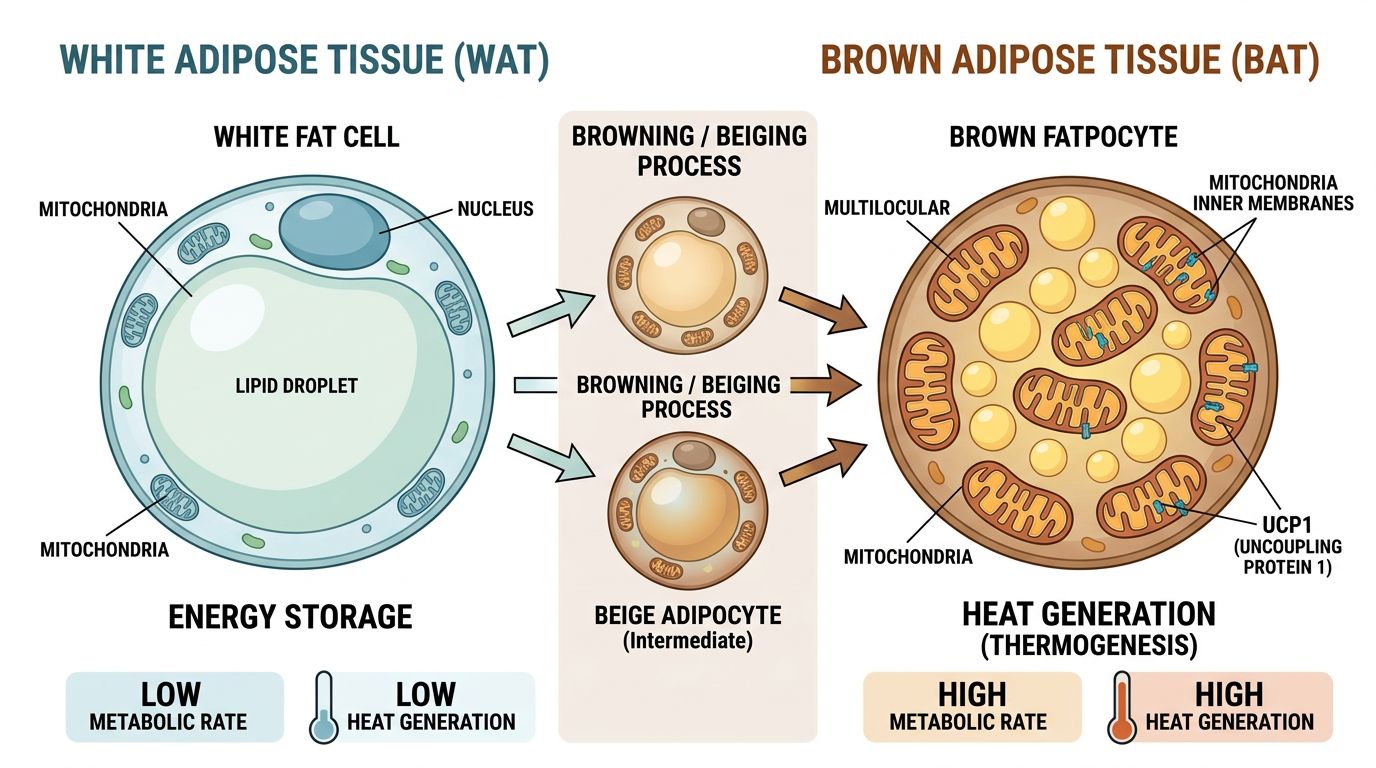
Implications for Adipocyte "Browning"
One of the most intriguing aspects of this metabolic synergy is the potential for phenotypic switching in adipose tissue. White fat stores energy; Brown fat burns it for heat (thermogenesis). "Beige" fat is white fat that has acquired brownish characteristics, rich in mitochondria and UCP1 (Uncoupling Protein 1).
High NNMT levels are a hallmark of profound "whiteness" in adipose tissue. Inhibition of NNMT has been shown to induce the expression of thermogenic genes (UCP1, PRDM16) within white fat depots. This browning effect is energy-expensive and relies heavily on oxidative capacity. The addition of NAD+ ensures that these newly recruited mitochondria have the redox potential required to sustain high rates of uncoupled respiration.
Safety Considerations and Quality Control
While the synergy of 5-amino-1mq and NAD+ is promising, rigorous safety evaluation is required in any experimental protocol. The primary consideration involves methylation balance. Use of 5-amino-1mq increases the pool of SAM. While generally beneficial for preventing steatosis and fibrosis, theoretically excessive methylation could have off-target effects. However, current animal data suggests the maximum tolerated dose is quite high without overt toxicity.
Furthermore, the source of research materials dictates the validity of the data. Impurities in 5-amino-1mq synthesis, such as unreacted quinolines or heavy metal catalysts, can induce cytotoxicity that mimics metabolic stress. Alpha Carbon Labs adheres to strict quality control protocols, utilizing HPLC and Mass Spectrometry to verify that all metabolic agents meet >99% purity standards suitable for sensitive cell culture and in vivo analysis.
Practical Application in Research Design
Solubility and Stability
Researchers handling these compounds must note their physicochemical properties. NAD+ is hygroscopic and sensitive to moisture; it must be stored desiccated at -20°C. 5-amino-1mq is generally supplied as a stable powder but requires specific solvents for reconstitution depending on the study type (in vitro vs. in vivo). Common vehicles include DMSO or sterile water, though 5-amino-1mq’s membrane permeability makes it more forgiving than many peptide sequences.
Dosing Protocols in Murine Models
Based on published literature, effective conversion dosing for 5-amino-1mq typically ranges from 10mg/kg to 20mg/kg in murine models via subcutaneous or intraperitoneal injection. NAD+ protocols vary widely, but "loading" phases are common. A synergistic protocol might involve establishing NNMT inhibition for 3-5 days prior to introducing diverse NAD+ boluses to observe the differential impact on metabolic flux compared to monotherapy.
Conclusion
The combination of NAD+ and 5-amino-1mq represents a sophisticated, mechanism-based approach to metabolic research. By targeting the enzymatic root of cellular energy dissipation (NNMT) and simultaneously replenishing the essential fuel for oxidation (NAD+), researchers can probe the upper limits of adipocyte metabolic flux.
This dual-pathway modulation moves beyond simple appetite suppression, addressing the fundamental bioenergetics of the fat cell. It offers a potential roadmap for reversing diet-induced obesity, improving insulin sensitivity, and combating the metabolic decline associated with aging. As research continues to unravel the complexities of the NAD+ salvage pathway, the utility of high-purity NNMT inhibitors will undoubtedly remain central to the conversation.
At Alpha Carbon Labs, we remain dedicated to supporting this vanguard of scientific inquiry with the highest quality research molecules available.
References
- 1. Neelakantan, H., et al. (2018). Selective and membrane-permeable small molecule inhibitors of nicotinamide N-methyltransferase reverse diet-induced obesity in mice. Biochemical Pharmacology, 147, 141-152.
- 2. Kraus, D., et al. (2014). Nicotinamide N-methyltransferase knockdown protects against diet-induced obesity. Nature, 508(7495), 258-262.
- 3. Yoshino, J., Baur, J. A., & Imai, S. I. (2018). NAD+ intermediates: the biology and therapeutic potential of NMN and NR. Cell Metabolism, 27(3), 513-528.
- 4. Cantó, C., & Auwerx, J. (2009). PGC-1alpha, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Current Opinion in Lipidology, 20(2), 98-105.
- 5. Schmeisser, K., et al. (2013). Role of sirtuins in lifespan regulation is linked to methylation of nicotinamide. Nature Chemical Biology, 9(11), 693-700.
- 6. Lomes, I., & Mayo, J. (2022). The role of Nicotinamide N-methyltransferase in metabolic disease and fibrosis. Frontiers in Endocrinology, 13.
- 7. Trammell, S. A., et al. (2016). Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nature Communications, 7, 12948.
- 8. Sperling, D., et al. (2023). 5-amino-1-methylquinolinium is a potent inhibitor of human nicotinamide N-methyltransferase. Journal of Enzyme Inhibition and Medicinal Chemistry, 38(1).
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.