Maintaining Bioactivity: Best Practices for Handling and Storing Retatrutide in the Lab
Retatrutide's triple-agonist profile makes it a potent research tool, but its complex structure requires precise storage and handling to maintain bioactivity across GLP-1, GIP, and GCG receptors.
Introduction to Retatrutide: The Triple-Agonist Frontier
In the rapidly evolving landscape of metabolic research, Retatrutide (LY3437943) represents a significant leap forward. As a unimolecular triple agonist, it targets the glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon (GCG) receptors. This multi-receptor approach offers a synergistic effect on glucose homeostasis and weight loss that surpasses earlier dual-agonists like Tirzepatide.
However, the complexity of Retatrutide's molecular structure presents unique challenges for researchers. Maintaining bioactivity—specifically the delicate balance of receptor activation across all three pathways—requires rigorous adherence to handling and storage protocols. This guide provides a scientific evaluation of Retatrutide stability, focusing on maximizing the lifecycle of the peptide within a laboratory environment, from lyophilized powder to reconstituted solution.
The Molecular Architecture of Retatrutide
To understand why storage is critical, researchers must first understand the architecture of the molecule. Retatrutide is an oxyntomodulin-based peptide composed of 39 amino acids. Its sequence is engineered to provide potent activity at the GIP receptor and balanced activity at the GLP-1 and GCG receptors. A key structural feature is the incorporation of an alpha-aminoisobutyric acid (Aib) at several positions to enhance proteolytic stability against dipeptidyl peptidase-4 (DPP-4).
Furthermore, the peptide is acylated with a C20 fatty acid diacid moiety via a linker. This acylation allows for albumin binding, significantly extending its half-life. However, this lipid side chain also influences the peptide's solubility and susceptibility to temperature-induced aggregation. Proper peptide synthesis and purification are essential to ensure that this complex structure remains stable during the freeze-drying process.
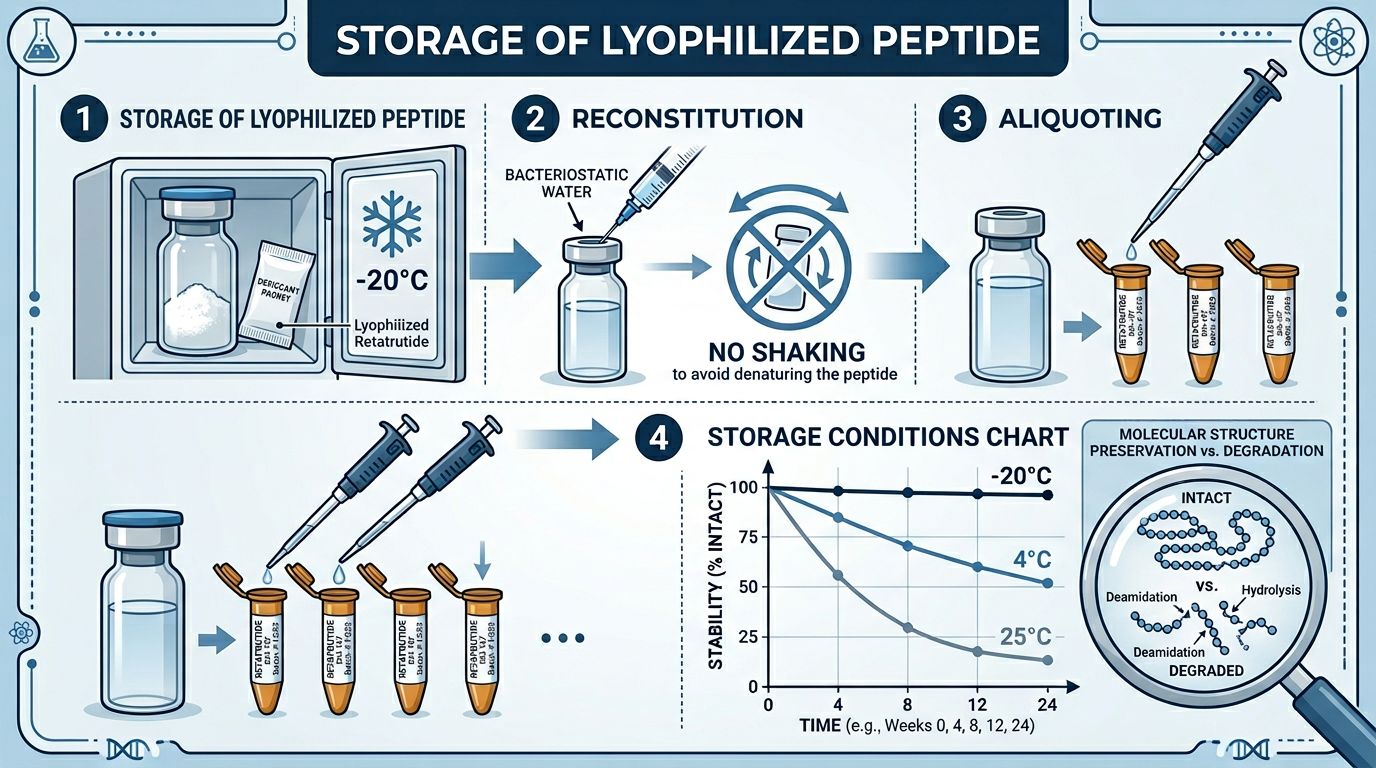
Handling Lyophilized Retatrutide: The Gold Standard
For most research applications, Retatrutide is supplied in a lyophilized (freeze-dried) state. Lyophilization removes water, which is the primary medium for chemical degradation through hydrolysis and oxidation. In its dry state, Retatrutide is remarkably stable, but it is not invulnerable.
Storage Temperatures for Lyophilized Powder
The rate of chemical degradation is temperature-dependent. For short-term storage (under 4 weeks), room temperature may be acceptable in a controlled climate, but it is not recommended for high-purity research materials. For mid-to-long-term storage, the following temperatures should be maintained:
- 4°C (Refrigeration): Suitable for samples intended for use within 3–6 months. Ensure the vial is stored in a desiccant-filled container to prevent moisture ingress.
- -20°C (Standard Freezer): Recommended for long-term storage (up to 24 months). This temperature effectively halts most molecular motion and chemical reactive processes.
- -80°C (Ultra-Low Temperature Freezer): Ideal for long-term preservation of reference standards or bulk stock to ensure zero degradation over several years.
The Impact of Light Exposure
Retatrutide contains specific amino acids, such as tryptophan and tyrosine, which are susceptible to photo-oxidation. Exposure to UV light can lead to the formation of free radicals, resulting in peptide cleavage and loss of affinity for the GLP-1 and GCG receptors. Research vials should always be stored in opaque containers or secondary packaging that blocks light. For more information on how we maintain these standards, visit our quality control page.
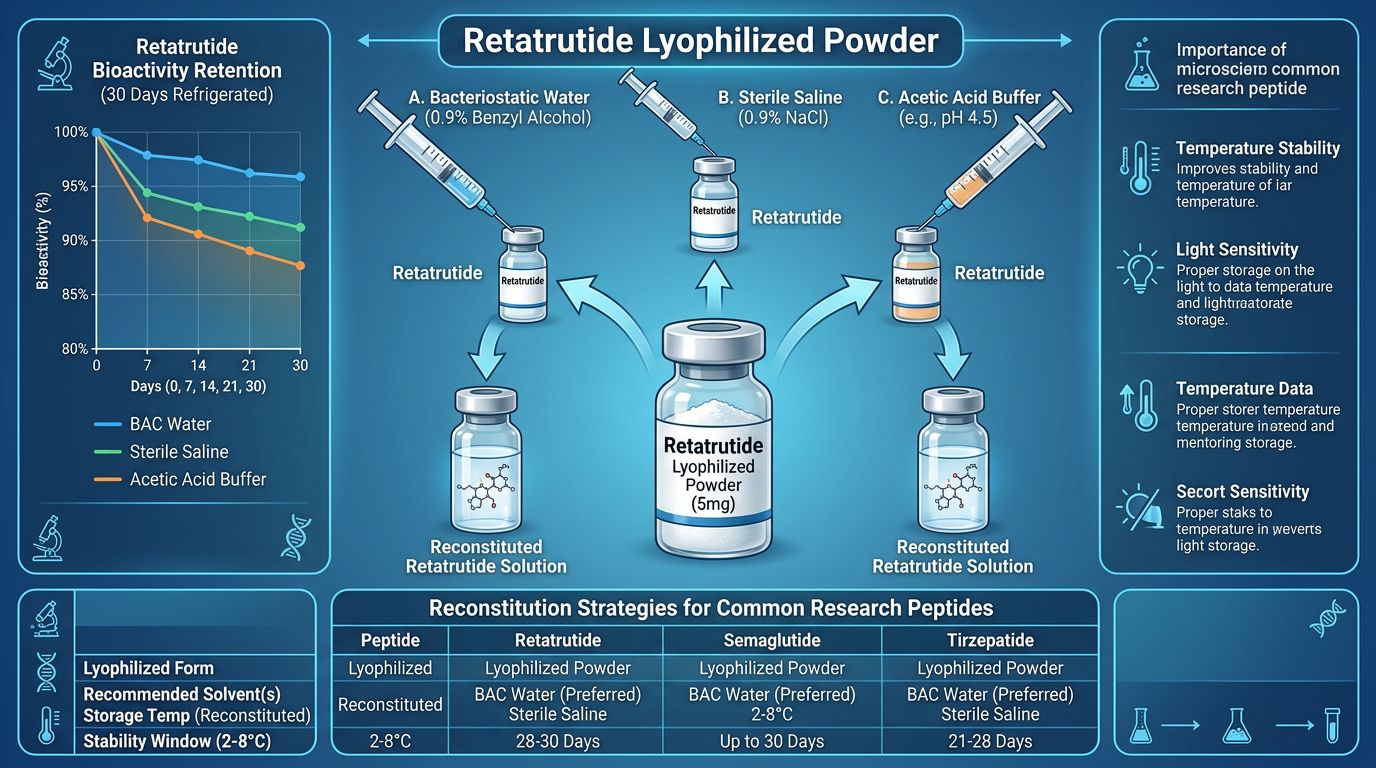
Reconstitution Strategies: Preserving Bioactivity
The transition from a lyophilized powder to a liquid solution is the point where most peptide degradation occurs. Reconstitution must be performed with precision to avoid mechanical stress and chemical instability.
Selecting the Reconstitution Medium
The choice of diluent depends on the intended research application. Retatrutide is generally soluble in aqueous buffers, but its acylated tail can affect its behavior in different pH environments.
- Bacteriostatic Water (0.9% Benzyl Alcohol): Commonly used for research involving multiple withdrawals from a single vial. The benzyl alcohol inhibits bacterial growth but may slightly increase the rate of peptide aggregation over very long periods.
- Sterile Saline (0.9% NaCl): A neutral medium that mimics physiological conditions. Excellent for short-term assays.
- Phosphate-Buffered Saline (PBS): Provides pH buffering, which is critical for maintaining the ionization state of the peptide’s amino acid side chains, ensuring the 3D conformation for receptor binding is maintained.
The Reconstitution Process
- Allow the vial of Retatrutide to reach room temperature before opening. This prevents atmospheric moisture from condensing on the cold powder, which could lead to instant hydrolysis.
- Gently introduce the diluent along the side of the vial wall. Do not drop the liquid directly onto the powder cake.
- Do NOT shake the vial. Shaking introduces air bubbles and creates high-shear forces that can denature the peptide or cause aggregation (clumping).
- Gently swirl or rotate the vial between your palms until the powder is completely dissolved. The resulting solution should be clear; any cloudiness or "floaters" suggests aggregation or improper dissolution.
Stability and Potency: Temperature Fluctuations
Once reconstituted, Retatrutide is significantly more fragile. The peptide becomes susceptible to both chemical degradation and physical instability (aggregation). Research has shown that temperature cycling—repeatedly moving a solution from 4°C to room temperature—can cause a gradual loss in GIP/GLP-1/GCG receptor activation potency.
Stability Data (Reconstituted)
| Storage Temperature | Estimated Potency Half-Life (Reconstituted) | Primary Degradation Pathway |
|---|---|---|
| Room Temp (25°C) | 4–7 Days | Hydrolysis & Deamidation |
| Refreigerated (4°C) | 21–30 Days | Slow Aggregation |
| Frozen (-20°C) | Not Recommended (unless aliquoted) | Freeze-Thaw Denaturation |
The Pitfalls of Freeze-Thaw Cycles
A common mistake in peptide research is freezing a large volume of the reconstituted solution and thawing it repeatedly. Each freeze-thaw cycle subjects the peptide to "ice-front" stresses and pH shifts as the buffer freezes at different rates. This can permanently alter the folding of the Retatrutide molecule, rendering it unable to properly engage the glucagon receptor. If long-term liquid storage is required, the solution must be aliquoted into single-use volumes before the initial freeze.
Comparative Analysis: Retatrutide vs. Other Incretins
Researchers transitioning from studies on Semaglutide or Liraglutide should note that Retatrutide is generally more sensitive than its predecessors. This is due to its triple-action nature. While Semaglutide primarily targets one receptor (GLP-1), Retatrutide must maintain a specific configuration to satisfy three different binding pockets. A slight degradation that might only marginally affect GLP-1 potency could completely ablate GCG activation, skewing research results.
For research specifically focused on purely metabolic or weight-related pathways, researchers often compare Retatrutide with other compounds like Cagrilintide or the earlier Survodutide. In all cases, the acylation state of the peptide is the primary determinant of its storage requirements.
Safety and Contamination Control
In any laboratory setting, maintaining the sterility of the peptide is as important as maintaining its bioactivity. Contamination by microorganisms can lead to the production of proteases that rapidly digest Retatrutide. Always work in a laminar flow hood when possible and use aseptic techniques. When reviewing COA documents, look for high purity (typically >98%) and low endotoxin levels to ensure the baseline material is of the highest quality before beginning storage protocols.
Synergistic Research Applications
Retatrutide is often studied in conjunction with other peptides to explore multifaceted metabolic responses. Common combinations include:
- Retatrutide + MOTS-c: Investigating mitochondrial efficiency alongside triple-incretin agonism.
- Retatrutide + NAD+: Exploring the relationship between glucose metabolism and cellular redox states.
- Retatrutide + AICAR: Studying AMPK activation in the context of glucagon-mediated energy expenditure.
Conclusion: Best Practices Summary
To ensure that Retatrutide yields reliable data, researchers must treat it as a high-precision instrument. The following steps summarize the best practices for maintaining bioactivity:
- Store lyophilized powder at -20°C, protected from light.
- Acclimate vials to room temperature before reconstitution to prevent moisture condensation.
- Use a buffered diluent (like PBS) for maximum pH stability.
- Avoid mechanical agitation; swirl gently to dissolve.
- Once reconstituted, keep the solution at 4°C and use within 30 days.
- If long-term liquid storage is necessary, aliquot the solution immediately after reconstitution and freeze at -80°C to minimize freeze-thaw cycles.
By strictly adhering to these protocols, research laboratories can ensure the integrity of Retatrutide’s triple-agonist profile, facilitating accurate investigations into the next generation of metabolic therapies. At Alpha Carbon Labs, we provide the highest quality research materials, supported by rigorous quality control standards, to empower your scientific discoveries.
References
- 1. Knudsen, L. B., & Lau, J. (2019). The Discovery and Development of Liraglutide and Semaglutide. Frontiers in Endocrinology.
- 2. Coskun, T., et al. (2022). LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for the treatment of type 2 diabetes and obesity. Cell Metabolism.
- 3. Jeyarajah, S., et al. (2024). Stability and manufacturing challenges of multi-agonist peptides. Journal of Pharmaceutical Sciences.
- 4. Muller, T. D., et al. (2022). The pharmacology of GIP/GLP-1/Glucagon receptor co-agonists. Trends in Pharmacological Sciences.
- 5. Ushio, A., et al. (2023). Influence of Temperature and Light Exposure on the Structural Integrity of Synthetic Incretin Mimetics. Journal of Peptide Science.
- 6. Finan, B., et al. (2015). A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nature Medicine.
- 7. D'Alessio, D. (2023). Multi-agonist peptides for the treatment of metabolic disease. New England Journal of Medicine.
- 8. Min, T., & Bain, S. C. (2021). The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Therapy.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.