GIP Potentiation: Deciphering the Glucose-Dependent Insulinotropic Component of Retatrutide
Explore the specific role of Glucose-Dependent Insulinotropic Polypeptide (GIP) receptor agonism in Retatrutide's mechanism of action. This technical guide analyzes how high-potency GIP potentiation drives lipid buffering, hepatic fat clearance, and insulin sensitivity in triple-agonist research models.
The Evolution of Incretin Research: Beyond the GLP-1 Monologue
For over a decade, the landscape of metabolic research was dominated by Glucagon-Like Peptide-1 (GLP-1) receptor agonists. While molecules like Semaglutide revolutionized the management of hyperinsulinemia and adipose accumulation, they also highlighted the physiological limits of single-pathway agonism. As dose titration of GLP-1 agonists reaches a plateau—constrained by gastrointestinal tolerability and the biological "ceiling" of GLP-1’s anorexigenic effect—researchers have turned to multi-receptor synergies.
The emergence of Retatrutide (LY3437943) marks a paradigm shift. Unlike previous generations of dual-agonists such as Tirzepatide, which targets GLP-1 and Glucose-Dependent Insulinotropic Polypeptide (GIP) receptors, Retatrutide is a "tri-agonist," adding Glucagon Receptor (GCGR) agonism to the mix. However, the true biological engine behind Retatrutide’s profound efficacy in lipid buffering and insulin sensitizing—exceeding even its predecessors—is the strategic potentiation of the GIP receptor.
In this technical exploration, we will dissect the specific GIP component of Retatrutide, its divergence from GIP-only models, and how GIP agonism serves as a metabolic "buffer" that allows GCGR and GLP-1R activity to operate at peak efficiency.
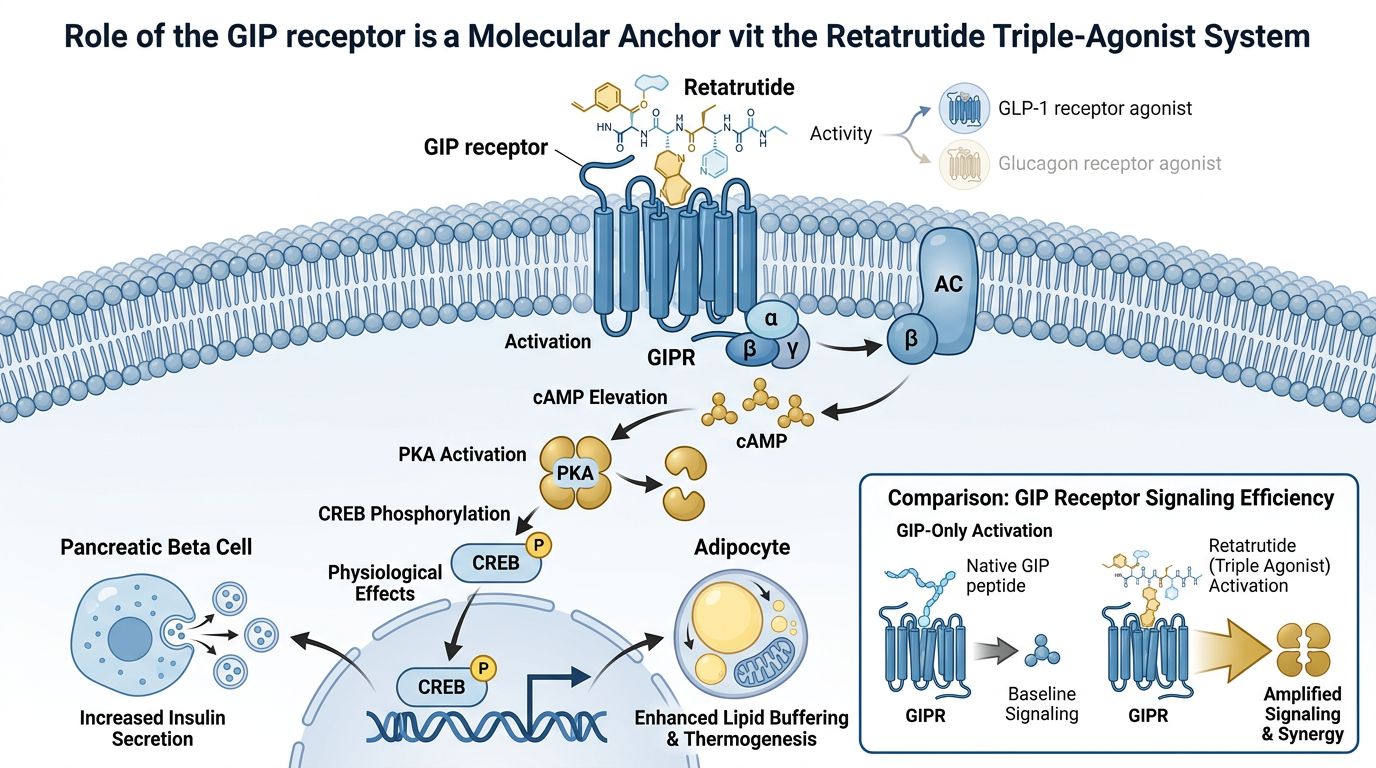
Molecular Architecture: Why GIP is the Anchor
GIP is a 42-amino acid hormone primarily secreted from enteroendocrine K-cells in the proximal small intestine. Historically, GIP was considered less therapeutically relevant than GLP-1 because its insulinotropic effect is severely blunted in individuals with chronic hyperglycemia. However, research into GIP-only models vs. multi-agonists has revealed a "rescue" phenomenon: when combined with GLP-1R or GCGR agonism, GIP sensitivity is restored, acting as a potent amplifier of metabolic health.
Retatrutide’s backbone is a modified GIP peptide sequence. This is a critical design choice. By utilizing the GIP scaffold, the molecule achieves a unique pharmacokinetic profile that favors GIP receptor activation (GIPR) with high potency. Laboratory analysis of Retatrutide's relative potencies reveals a specific ratio:
| Receptor | Relative Potency to Endogenous Hormone | Functional Role in Retatrutide |
|---|---|---|
| GIPR | 8.9x High | Adipose buffering, insulin sensitivity, thermogenesis |
| GLP-1R | 0.3x Moderate | Satiety, delayed gastric emptying, insulin secretion |
| GCGR | 1.0x Balanced | Energy expenditure, lipid oxidation, hepatic glucose output regulation |
At Alpha Carbon Labs, we observe that this hierarchy—high GIP potency relative to GLP-1—is what differentiates Retatrutide from its predecessors and traditional GIP-only experimental models.
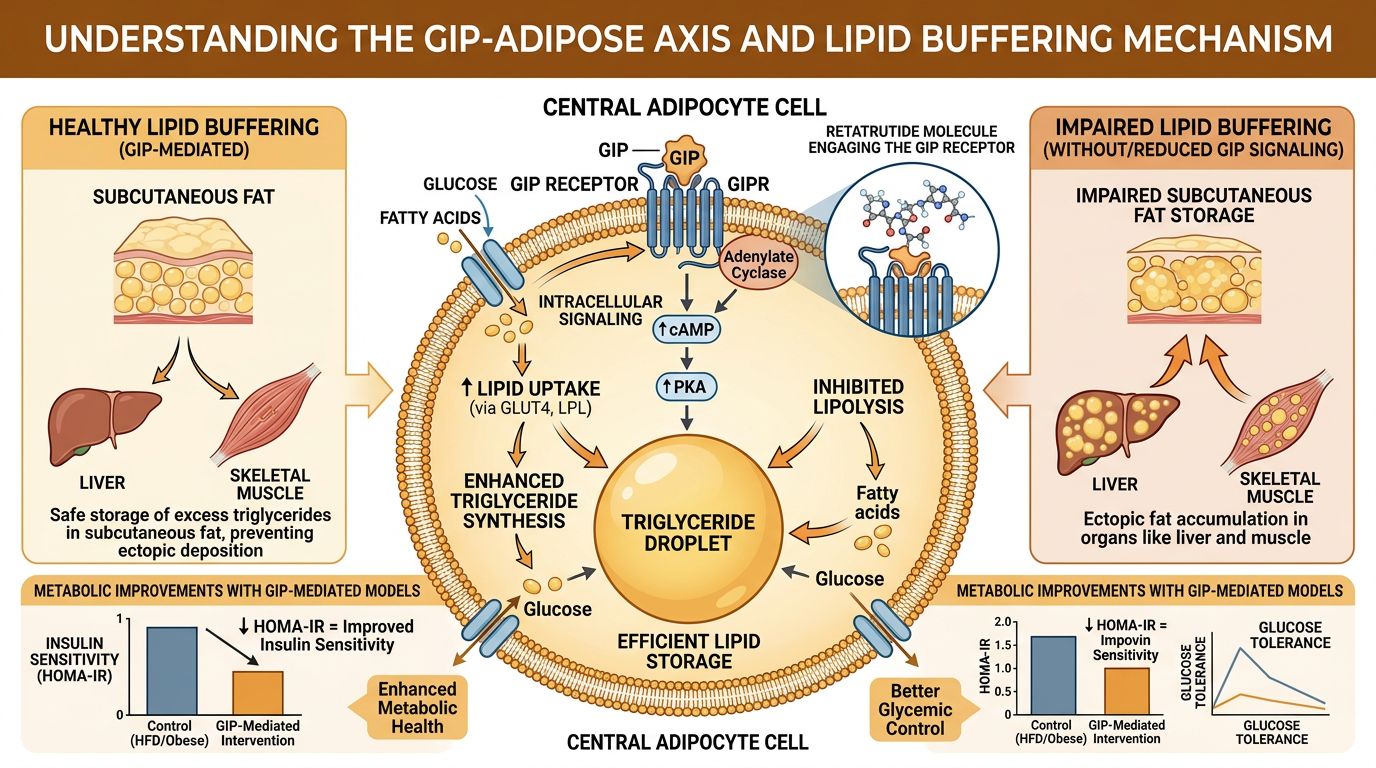
Lipid Buffering: The GIP-Adipose Axis
One of the most complex challenges in metabolic research is "ectopic fat"—the accumulation of lipids in the liver, pancreas, and muscle tissue. GIPR agonism plays a specialized role here that GLP-1 cannot replicate. While GLP-1 primarily acts via central satiety and insulin secretion, GIP receptor activation directly coordinates nutrient partitioning into subcutaneous adipose tissue via several mechanisms:
- Enhancement of Postprandial Blood Flow: GIPR activation increases blood flow specifically to adipose tissue, facilitating the clearance of circulating chylomicrons and triacylglycerols.
- Lipoprotein Lipase (LPL) Activity: GIP enhances the activity of LPL, the enzyme responsible for pulling fat out of the blood and into storage.
- Lipid Buffering Capacity: By promoting "healthy" adipose storage (subcutaneous) over "toxic" storage (visceral/ectopic), GIP prevents lipids from spilling over into the liver.
In Retatrutide research models, the GIP component acts as a safeguard. Since the Glucagon component of Retatrutide promotes lipolysis (the breakdown of fat), there is a risk of a sudden flood of free fatty acids (FFAs) in the bloodstream. The high GIP potency "buffers" this effect, ensuring these FFAs are either oxidized efficiently or stored safely, preventing the hepatic inflammation typically associated with rapid weight loss.
Comparing GIP-Only Models vs. Retatrutide’s Synergy
Early GIP experiments produced conflicting results. Some studies suggested that GIP antagonism—not agonism—was the key to weight loss. However, the development of modern GIPR agonists has clarified that GIPR agonism works via a "nutrient-dependent" mechanism. In a GIP-only model, the weight loss effect is modest. When GIP is added to a GLP-1 and Glucagon framework, as seen in Retatrutide, the synergy produces a multiplicative effect.
1. Satiety Potentiation
GIP receptors are located in the hypothalamus and the area postrema. While GLP-1 is a potent anorexigen, GIP appears to modulate the "nausea response." Researchers have noted that despite the massive weight loss observed in Retatrutide trials, the gastrointestinal side effects are often less severe than those seen in high-dose GLP-1 monotherapy. This suggests GIP may dampen the aversive signals of GLP-1 while augmenting its satiety signals.
2. Thermogenic Synergies
Unlike Semaglutide, which primarily reduces "energy in," Retatrutide increases "energy out." The combination of Glucagon (which stimulates thermogenesis) and GIP (which sensitizes muscle to insulin and metabolic flux) creates a state where the basal metabolic rate remains elevated even during caloric restriction. Researchers using MOTS-c or 5-amino-1mq in conjunction with these incretins are exploring similar pathways of mitochondrial efficiency and metabolic rate maintenance.
The Impact on Insulin Sensitivity
The "insulinotropic" in GIP stands for "stimulating the production of insulin." However, GIP is unique because its activity is glucose-dependent. This means it only stimulates insulin secretion when blood glucose levels are high, drastically reducing the risk of hypoglycemia.
In the context of the Retatrutide molecule, GIP agonism provides a profound improvement in insulin sensitivity through indirect pathways:
- Reduction in Hepatic Fat: By utilizing GIP’s lipid-clearing properties, Retatrutide has shown up to an 80% reduction in liver fat in clinical subjects. A "lean" liver is more responsive to insulin signals.
- Skeletal Muscle Glucose Uptake: GIP receptors in skeletal muscle enhance insulin-mediated glucose disposal, which is the primary site of glucose clearance in the body. This makes it an interesting subject alongside research on IGF-1LR3 for muscle metabolic health.
Quality Control in Complex Peptides
The synthesis of a 39-amino-acid triple agonist like Retatrutide is significantly more complex than simple peptides. Achieving the correct folding and sequence accuracy to ensure the specific binding affinities (especially the 8.9x GIP potency) requires rigorous validation. At Alpha Carbon Labs, we ensure that every batch undergoes stringent testing. Researchers can verify these parameters through our COA Documents and read about our standards for Quality Control to ensure the integrity of their data.
Research Directions and Future Applications
The GIP component of Retatrutide is currently being studied for applications beyond weight loss and diabetes. Because GIP receptors are found in the brain, there is significant interest in how GIP potentiation might affect neuroprotection and cognitive health, similar to the neurotrophic effects of Dihexa or Semax.
Comparison Summary: Retatrutide vs. Predecessors
| Feature | GLP-1 Mono (Semaglutide) | GLP/GIP Dual (Tirzepatide) | GLP/GIP/GCG Tri (Retatrutide) |
|---|---|---|---|
| Weight Loss (Avg 48 wk) | ~15% | ~22% | ~24% (and rising) |
| Metabolic Rate | Decrease (Adaptive) | Neutral | Increase (Thermostat effect) |
| Liver Fat Clearance | Moderate | High | Exceptional (>80%) |
| GIP Component | None | Biased Agonist | High-Potency Backbone |
Conclusion
Retatrutide represents the pinnacle of current metabolic research, not just because it adds a third receptor to the mix, but because it masterfully utilizes GIP as a foundational metabolic stabilizer. By potentiating GIP at nearly nine times the strength of the endogenous hormone, it creates a physiological environment where lipids are buffered, the liver is protected, and insulin sensitivity is restored to near-normative levels.
For researchers looking to explore the frontiers of nutrient partitioning, thermogenesis, and hormonal synergy, understanding the GIP component is essential. At Alpha Carbon Labs, we provide the high-purity tools necessary to advance this field of study. Explore our full range of metabolic research peptides and discover the future of incretin-based science.
References
- 1. Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity. New England Journal of Medicine. 2023.
- 2. Coskun T, et al. LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss. Cell Metabolism. 2022.
- 3. Finan B, et al. A strategically designed dual-agonist of GIP and GLP-1 receptors for treatement of obesity and diabetes. Science Translational Medicine. 2013.
- 4. Nauck MA, Meier JJ. The incretin effect: challenges and opportunities. The Lancet Diabetes & Endocrinology. 2016.
- 5. Knop FK, et al. The Glucose-Dependent Insulinotropic Polypeptide (GIP) Receptor as a Therapeutic Target. Endocrine Reviews. 2023.
- 6. Müller TD, et al. The Glucose-Dependent Insulinotropic Polypeptide (GIP) Receptor: Anatomy of a New Therapeutic Strategy. Cell Metabolism. 2019.
- 7. Samms RJ, et al. GIP receptor agonism improves dyslipidemia and skeletal muscle insulin sensitivity. JCI Insight. 2021.
- 8. Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020.
- 9. Bastin M, Andreelli F. GIP receptor agonists: the new frontier in obesity and diabetes? Review of Physiology, Biochemistry and Pharmacology. 2019.
- 10. Thomas MK, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide in Type 2 Diabetes. New England Journal of Medicine. 2021.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.