Beyond Inherent GLP-1 Signaling: The Nuanced Role of GIP Agonism in Tirzepatide-Mediated Insulin Sensitivity Research
Discover how Tirzepatide goes beyond appetite suppression. We explore the nuanced science of GIP agonism and how it specifically targets insulin sensitivity and metabolic dysfunction.
The Next Evolution of Metabolic Optimization
If you have been following the explosion of research into metabolic health and weight management, you have almost certainly heard of GLP-1 agonists like Semaglutide. These compounds have revolutionized how we approach appetite suppression and blood sugar control. However, the scientific community has already moved toward something even more sophisticated: the "dual agonist."
Enter Tirzepatide. While it shares some DNA with its predecessors, it introduces a critical new player to the game: Glucose-dependent Insulinotropic Polypeptide, or GIP.
For a long time, researchers thought GIP wasn't useful for weight loss because it didn't seem to suppress appetite on its own. But recent deep dives into metabolic syndrome models have revealed that GIP is actually a powerhouse when it comes to insulin sensitivity and fat metabolism. It works differently than GLP-1, and when combined, the results are compounding.
This article explores specifically how the GIP component of Tirzepatide changes the equation, helping your body utilize insulin more effectively and handle calories more efficiently than GLP-1 alone.
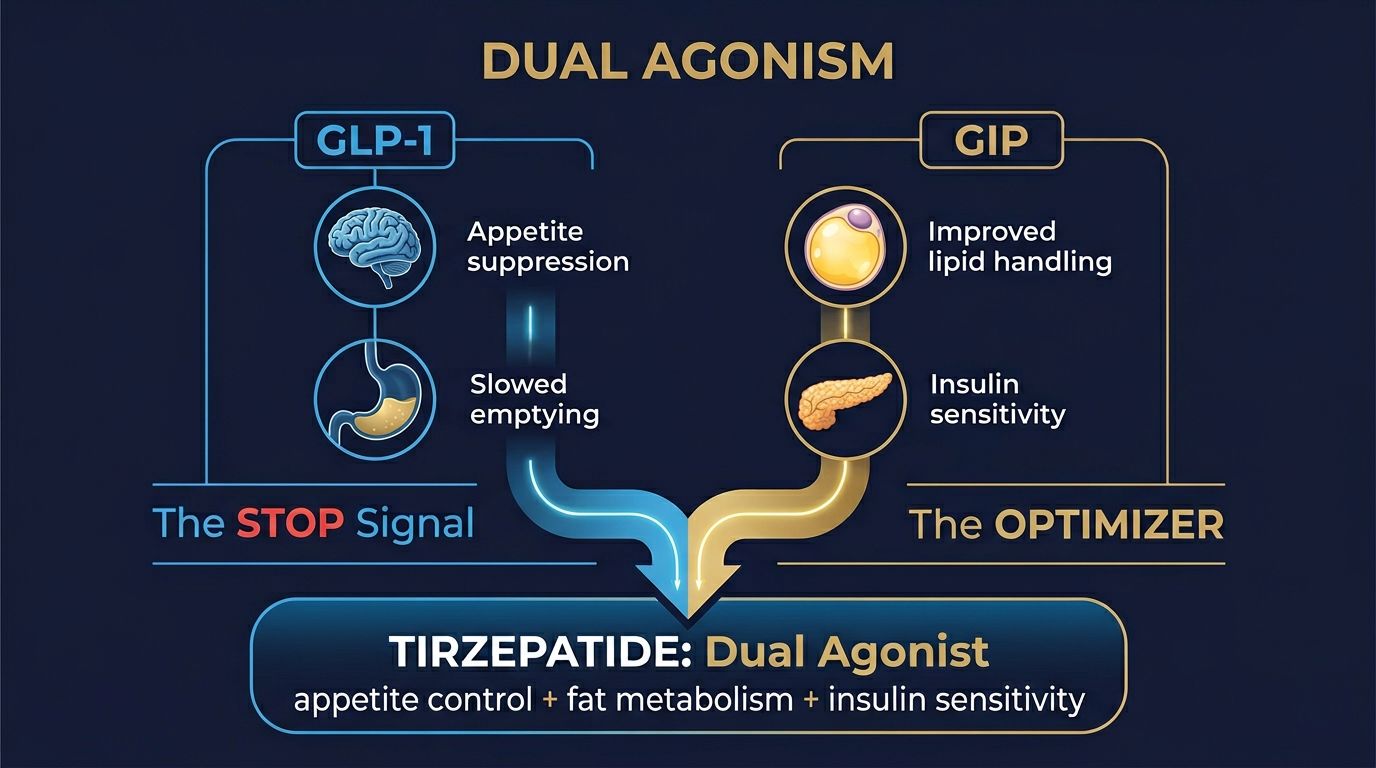
The Basics: GLP-1 vs. GIP
To understand why the addition of GIP is such a big deal, we first need to clarify the difference between these two "incretin" hormones. Your body naturally produces both in the gut after you eat.
- GLP-1 (Glucagon-Like Peptide-1): This is the "stop" signal. It slows down gastric emptying (how fast food leaves your stomach), signals the brain that you are full, and tells the pancreas to release insulin when blood sugar is high. This is the primary mechanism behind Semaglutide.
- GIP (Glucose-dependent Insulinotropic Polypeptide): This is the "storage and utilization" manager. Historically, it was known for stimulating insulin release. However, modern research shows it also acts directly on fat cells (adipocytes) to encourage safe lipid storage and improve how the whole body responds to insulin.
The magic of Tirzepatide isn't just that it does both; it's that GIP seems to fix the metabolic "broken gears" that GLP-1 alone cannot fully reach.
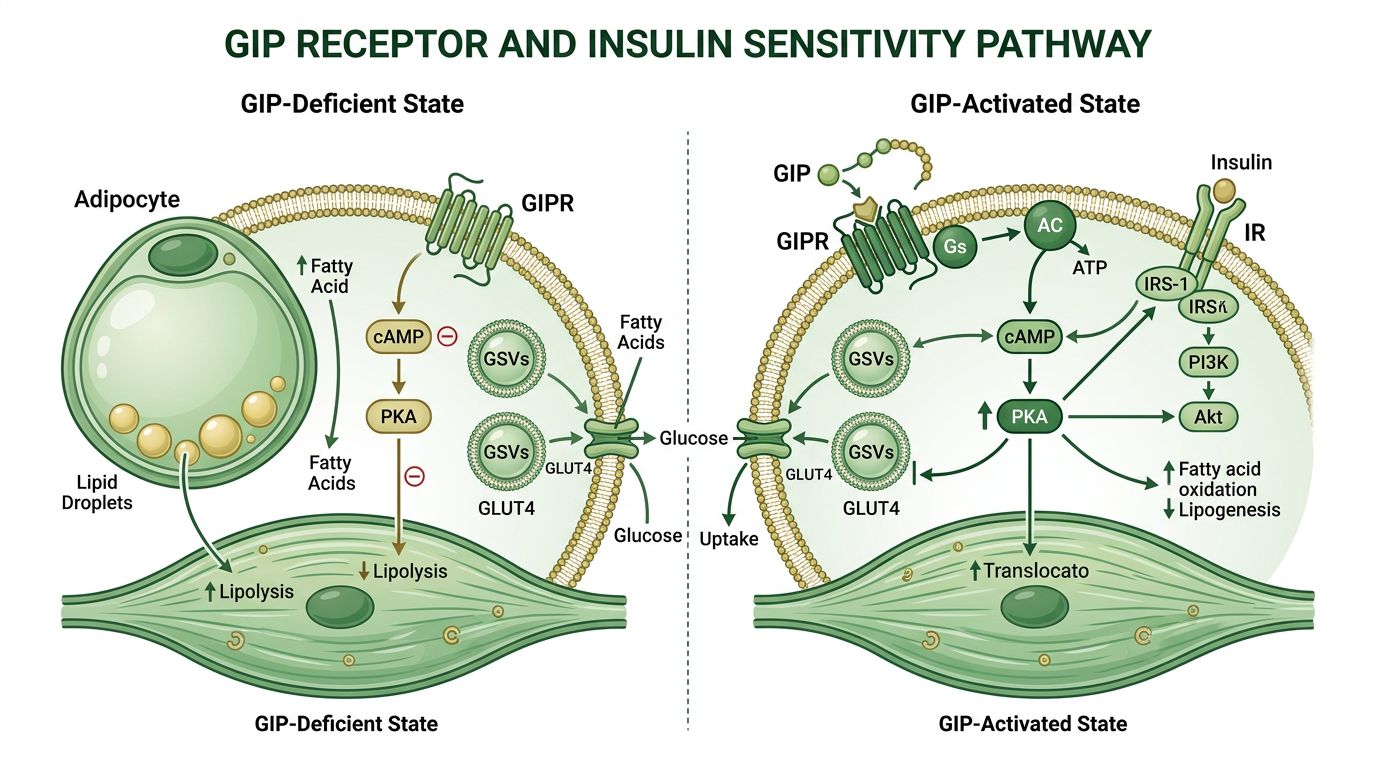
Deep Dive: How GIP Improves Insulin Sensitivity
Insulin sensitivity is a measure of how efficiently your cells respond to the hormone insulin. When you are insulin sensitive, your body needs only a small amount of insulin to manage blood sugar levels. When you are insulin resistant (a hallmark of metabolic syndrome), your pancreas works overtime pumping out massive amounts of insulin just to keep up.
Here is where the GIP component of Tirzepatide shines giving it a distinct advantage over single-receptor agonists.
1. The "Buffering" Effect on Fat Cells
One of the most fascinating nuances of GIP research is its effect on white adipose tissue (body fat). In metabolic dysfunction, fat cells are often stressed and inflamed. They "leak" fatty acids into the bloodstream, which then clog up muscle and liver cells, causing insulin resistance.
GIP agonism helps these fat cells function correctly again. It improves the ability of fat tissue to act as a safe buffer for excess energy. By stopping the "leak" of lipids into the bloodstream, GIP indirectly clears the way for your muscles and liver to listen to insulin signals again. This is a mechanism largely independent of the appetite suppression you get from GLP-1.
2. Direct Action on the Pancreas
While GLP-1 is great at stimulating insulin, GIP is actually the dominant incretin hormone in healthy individuals, accounting for the majority of the insulin response after a meal. By stimulating GIP receptors, Tirzepatide produces a robust, natural insulin response that closely mimics healthy physiology.
Research indicates that GIP also supports the health of beta-cells (the cells that make insulin). Instead of just whipping a tired horse, GIP helps support the horse itself, potentially preserving pancreatic function over time.
3. Reducing Nausea to Allow Higher Dosing
This is a practical benefit that influences results. GLP-1 receptors in the brain are responsible for the feeling of fullness, but also for nausea. There is a "ceiling" to how much GLP-1 you can tolerate.
GIP, interestingly, seems to have an anti-emetic (anti-nausea) effect in some models. It acts on different receptors in the brain unrelated to nausea. This allows the combined therapy to push metabolic levers harder without triggering the side effects that would occur if you simply doubled the dose of a GLP-1 agonist.
Metabolic Synergy: Why 1 + 1 = 3
The most compelling aspect of Tirzepatide is the synergy. It is not just doing two things at once; the two pathways amplify each other. This is crucial for anyone studying peptides for optimizing body composition.
| Feature | GLP-1 Only (e.g., Semaglutide) | GLP-1 + GIP (Tirzepatide) |
|---|---|---|
| Primary Mechanism | Appetite suppression & delayed gastric emptying | Appetite suppression + Enhanced lipid handling & insulin sensitization |
| Insulin Sensitivity | Improved primarily via weight loss | Improved via weight loss AND direct cellular signaling |
| Fat Metabolism | Indirect (via calorie deficit) | Direct action on adipocyte function |
| Typical Trial Results | High efficacy (~15% weight reduction) | Very high efficacy (~20-25% weight reduction) |
For those researching metabolic efficiency, the addition of GIP essentially "unlocks" fat stores that are resistant to mobilization solely through calorie restriction. It creates a metabolic environment where the body is more willing to burn fat because it is no longer fighting against high circulating insulin levels.
Beyond Weight: The Anti-Inflammatory Connection
Chronic inflammation is the silent killer behind metabolic syndrome. It makes insulin resistance worse, creates brain fog, and hinders recovery.
Recent studies suggest that GIP receptors are found on immune cells as well. Activation of these receptors may promote a systemic anti-inflammatory effect. When you combine this with the weight loss effects, you get a profound reduction in systemic inflammation markers like C-reactive protein (CRP).
This is where other peptides might also play a supportive role. For example, researchers looking into recovery and inflammation often explore compounds like BPC-157 or TB-500 alongside metabolic agents to support overall tissue health, though their mechanisms are entirely different from incretins.
Comparing the Landscape: Tirzepatide vs. Other Agonists
The peptide industry moves fast. While Tirzepatide is the current star of dual agonism, researchers are already looking at "triple agonists" and other variations.
- Semaglutide: The gold standard for single receptor agonism. Excellent for appetite control but lacks the specific insulin-sensitizing "fat cell" magic of GIP.
- Retatrutide: A newer "triple agonist" effectively targeting GLP-1, GIP, and Glucagon receptors. The addition of Glucagon increases energy expenditure (calorie burning), potentially raising the bar even higher than Tirzepatide.
- Mazdutide: A dual agonist targeting GLP-1 and Glucagon (skipping GIP). This approach focuses heavily on burning energy but may have a different side effect profile regarding heart rate.
- Survodutide: Another glucagon/GLP-1 dual agonist showing promise in liver health research.
However, for purely targeting insulin resistance and classic "metabolic syndrome" (belly fat, high sugar), the GIP/GLP-1 combination of Tirzepatide remains the most clinically validated leap forward from standard therapies.
What This Means for Metabolic Research
If you are exploring peptides for wellness or optimization, understanding GIP changes your perspective. It shifts the focus from "starving the body" (suppressing appetite) to "optimizing the body" (fixing how the body processes fuel).
When the body becomes insulin sensitive via GIP agonism, you may notice:
- More Stable Energy: Less crashing after meals because blood sugar is regulated efficiently.
- Better Nutrient Partitioning: Calories are more likely to be directed toward muscle repair (especially if paired with resistance training) rather than fat storage.
- Stubborn Fat Loss: Mobilization of fat from visceral areas (around the organs) that are typically protected by insulin resistance.
Quality Matters in Peptide Research
Because Tirzepatide acts on such precise biological levers, the purity of the research compound is paramount. Impurities or incorrect peptide sequencing can result in reduced efficacy or unwanted immune reactions.
At Alpha Carbon Labs, we emphasize rigorous testing standards. All our compounds undergo High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry analysis to ensure they meet the highest purity standards for research applications. Dealing with complex molecules like dual agonists requires a commitment to synthesis excellence that not all suppliers provide.
Conclusion: The GIP Advantage
The narrative that "calories in, calories out" is the only thing that matters is slowly dying. Hormones dictate *how* those calories are used. The nuanced role of GIP in Tirzepatide research proves that we can do more than just suppress appetite—we can fundamentally alter the metabolic environment of the body.
By improving insulin sensitivity directly at the cellular level and modulating how fat tissue buffers lipids, the GIP component of Tirzepatide offers a sophisticated tool for reversing metabolic dysfunction. Whether you are researching for anti-aging, body composition, or general wellness, understanding the power of the dual agonist approach is essential for modern metabolic optimization.
Disclaimer: The rigorous research into GIP and GLP-1 agonists highlights their potential, but these compounds are sold for research and laboratory use only. They are not intended for use as food, additives, or drugs in humans.
References
- 1. Finan, B., et al. (2013). Unimolecular dual incretins maximize metabolic benefits in rodents, monkeys, and humans. Science Translational Medicine.
- 2. Coskun, T., et al. (2018). LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Molecular Metabolism.
- 3. Frias, J. P., et al. (2021). Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine.
- 4. Heise, T., et al. (2022). GIP receptor agonism in the treatment of type 2 diabetes and obesity: Potential mechanisms and clinical implications. Diabetes, Obesity and Metabolism.
- 5. Samms, R. J., et al. (2020). GIPR agonism mediates weight-independent improvements in systemic insulin sensitivity and lipid metabolism. Cell Metabolism.
- 6. Nauck, M. A., & D'Alessio, D. A. (2021). Tirzepatide, a dual GIP/GLP-1 receptor co-agonist for the treatment of type 2 diabetes with unmatched effectiveness regarding glycaemic control and body weight reduction. Diabetes/Metabolism Research and Reviews.
- 7. Müller, T. D., et al. (2019). The biological architecture of the GIP receptor. Molecular Metabolism.
- 8. Baggio, L. L., & Drucker, D. J. (2021). Glucagon-like peptide-1 receptor co-agonists for treating metabolic disease. Molecular Metabolism.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.