Analytical Validation: Ensuring Chemical Identity and Purity When Sourcing Semaglutide for Sale
A technical guide for laboratories on the analytical validation of Semaglutide, focusing on HPLC, Mass Spectrometry, and avoiding counterfeit peptide risks in research settings.
The Landscape of Synthetic Semaglutide Research
In the burgeoning field of metabolic research, Semaglutide has emerged as perhaps the most significant peptide molecule of the last decade. As a long-acting glucagon-like peptide-1 (GLP-1) receptor agonist, its role in modulating glucose-dependent insulin secretion and suppressing glucagon release has revolutionized the study of Type 2 Diabetes (T2D) and obesity. However, the surge in global demand has led to a fragmented marketplace. For the biochemical researcher, the challenge is no longer just finding Semaglutide for sale, but ensuring that the material acquired meets the rigorous standards of chemical identity, purity, and conformational integrity required for reproducible data.
At Alpha Carbon Labs, we recognize that research outcomes are only as reliable as the raw materials used. This guide provides an in-depth exploration of the analytical validation processes required to distinguish high-purity research-grade Semaglutide from subpar or counterfeit alternatives often found in the "grey market."
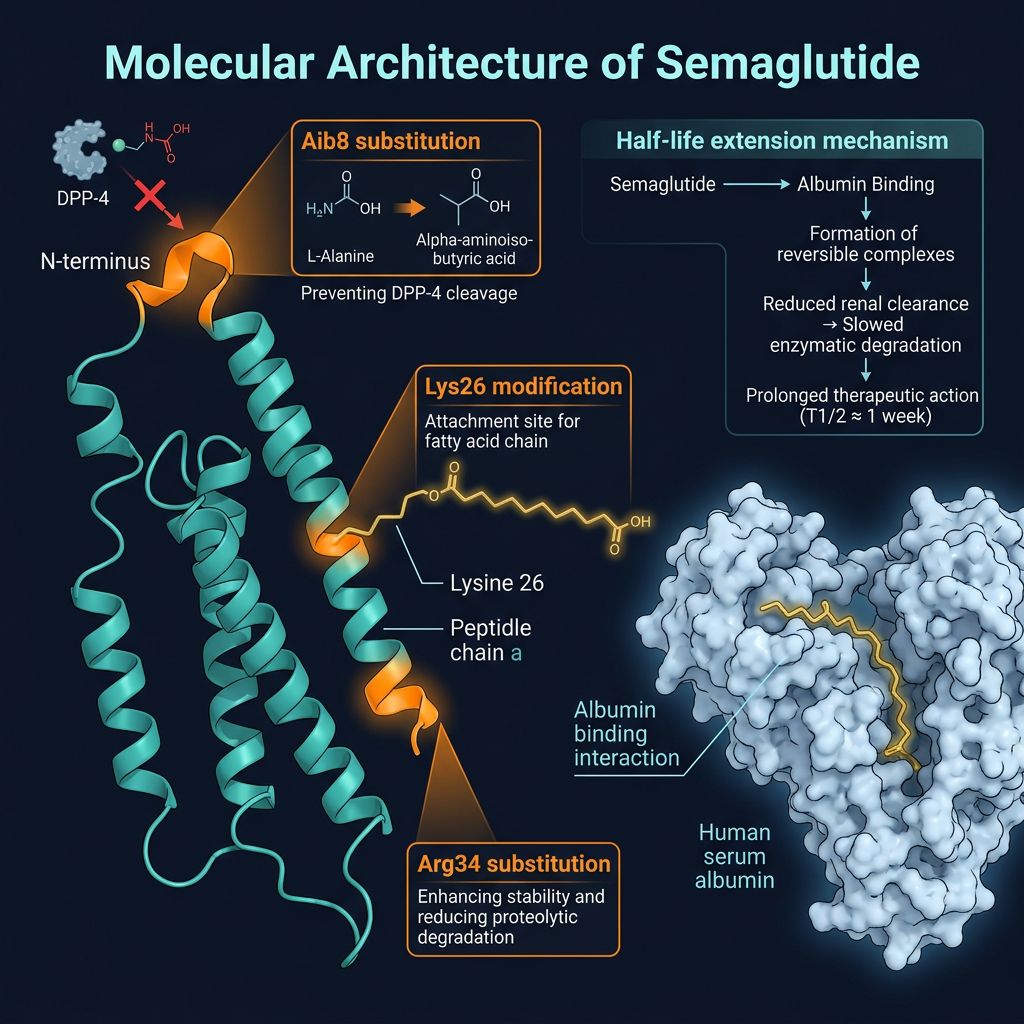
Molecular Architecture of Semaglutide
Understanding the validation process requires a deep dive into the molecule itself. Semaglutide is a 31-amino acid peptide that shares 94% structural homology with human GLP-1 (7-37). Its extended half-life (approximately 165 hours) is the result of three specific chemical modifications:
- Amino Acid Substitution: Alanine at position 8 is replaced by alpha-aminoisobutyric acid (Aib), which protects the peptide from degradation by the enzyme dipeptidyl peptidase-4 (DPP-4).
- Acylation: A C18 fatty diacid chain is attached to Lysine at position 26 via a hydrophilic spacer (gamma-Glu and two OEG units). This allows for high-affinity albumin binding, reducing renal clearance.
- Substitution at Position 34: Lysine is replaced by Arginine to ensure the attachment of the fatty acid occurs specifically at position 26.
These complex modifications—particularly the acylation step—present significant challenges during peptide synthesis. Minor errors in the synthesis of the side chain can result in "isobaric impurities" that are difficult to detect without advanced analytical instrumentation.
Establishing Chemical Identity: Beyond the Label
The first step in validating Semaglutide for sale is confirming its primary sequence and the presence of the lipid side chain. Simple visual inspection or 1D-NMR is often insufficient for macro-molecular peptides of this complexity.
High-Resolution Mass Spectrometry (HRMS)
HRMS is the "gold standard" for determining the molecular weight of a peptide. For Semaglutide, the theoretical monoisotopic mass is approximately 4111.12 Da. When auditing a COA (Certificate of Analysis), researchers should look for the "Found Mass" vs. "Calculated Mass."
Crucially, HRMS can detect "deletion sequences"—peptides missing a single amino acid—which might only differ by a few hundred Daltons. A high-resolution instrument (such as an Orbitrap or Q-TOF) provides the precision necessary to verify that the acylation group is correctly attached, a common point of failure in low-cost synthesis protocols.
Amino Acid Analysis (AAA)
While MS confirms mass, AAA confirms the ratio of amino acids present. This ensures that the peptide backbone is constructed according to the specific Semaglutide template. If the ratio of GLU to LYS deviates from the expected 3:1 (accounting for the spacer), it suggests a synthesis error or the presence of a different GLP-1 analogue like Liraglutide.
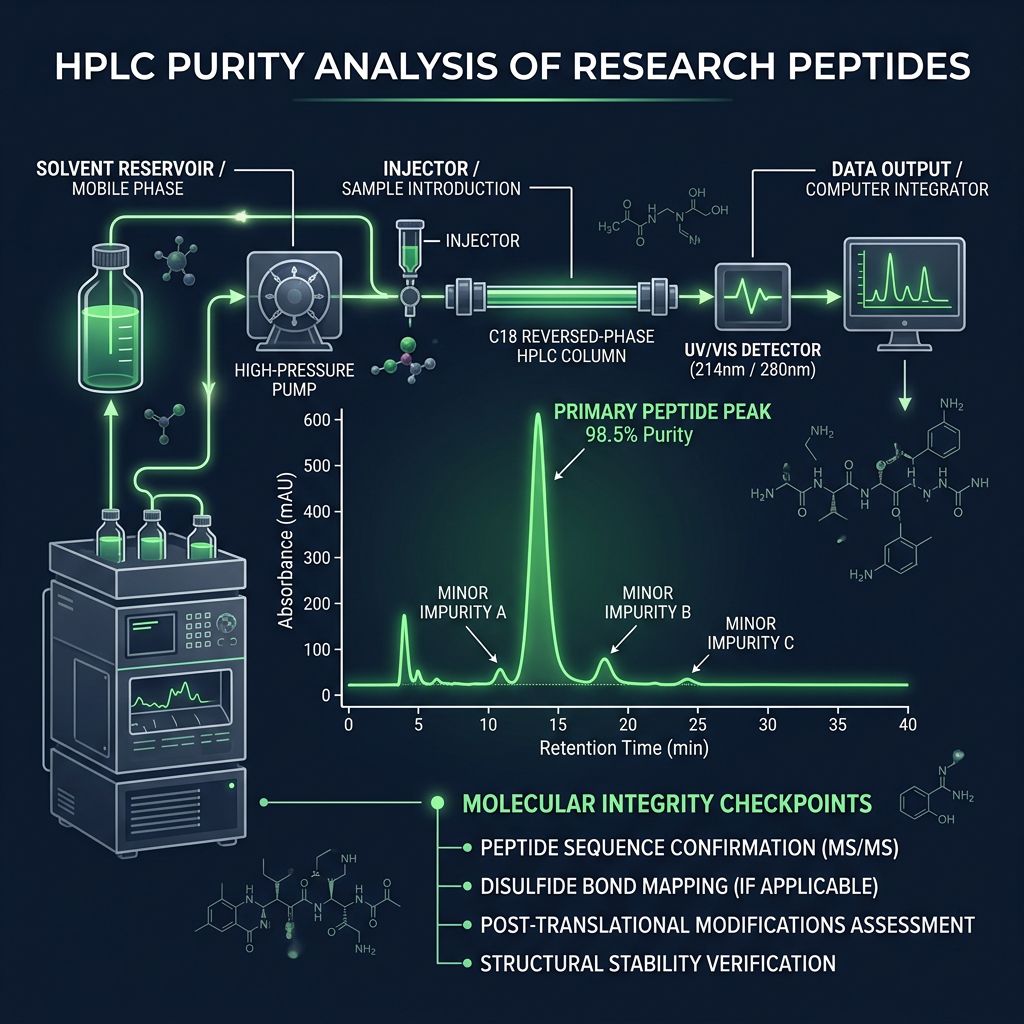
Quantifying Purity: High-Performance Liquid Chromatography (HPLC)
When searching for Semaglutide for sale, the term "99% Purity" is frequently cited. However, the validity of this claim depends entirely on the chromatographic method used. HPLC is the standard technique for separating the target peptide from related substances, such as:
- Diastereomers: Peptides where one amino acid has undergone racemization (from L-form to D-form).
- Truncated Sequences: Incomplete chains resulting from inefficient coupling steps.
- Oxidation Products: Specifically at the Methionine residues (though Semaglutide lacks Met, other GLP-1s contain it, making this a useful check for cross-contamination).
- Dimerized Peptides: Two Semaglutide molecules bonding together, which can significantly alter receptor binding kinetics.
The Importance of Gradient Elution
A "flat" or poorly optimized HPLC gradient can mask impurities by causing them to co-elute with the main peak. At Alpha Carbon Labs, our quality control protocols utilize stringent gradient profiles to ensure that closely related impurities are resolved and accounted for in the final purity percentage.
| Impurity Type | Source | Detection Method | Impact on Research |
|---|---|---|---|
| Trifluoroacetic Acid (TFA) | Cleavage/Purification residue | 19F-NMR / Ion Chromatography | Cellular toxicity; altered pH in assays |
| Deamidation Peaks | Storage/Handling degradation | RP-HPLC / LC-MS | Reduced biological activity |
| Residual Solvents | Acetonitrile/DMF residues | GC-HS (Gas Chromatography) | In-vitro cell line interference |
| Counterfeit Peptides | Intentional substitution | MS/MS Sequencing | Invalidation of all data; safety risks |
The Risk of Counterfeit and "Grey Market" Peptides
With the rise of "Ozempic-style" weight loss trends, the market has seen an influx of counterfeit products. These are often produced in facilities that do not adhere to Good Manufacturing Practices (GMP) and may contain:
- None of the active ingredient (Semaglutide).
- Incorrect dosages (often underdosed by 30-50%).
- Alternative, cheaper peptides like Tirzepatide or Retatrutide mislabeled as Semaglutide.
- Bacterial endotoxins from substandard purification.
Researchers must be wary of "rebrands" where medicinal vials are emptied and refilled. True research-grade Semaglutide should arrive as a lyophilized (freeze-dried) powder in a sterile vacuum-sealed vial, intended for reconstitution with bacteriostatic water.
Stability and Storage Considerations
The stability of Semaglutide is heavily dependent on its lyophilized state and the removal of residual moisture. Even 99% pure Semaglutide will degrade rapidly if exposed to ambient temperatures for extended periods or if the pH of the salt form is not properly balanced during the final purification stages.
- Long-term Storage: Lyophilized powder should be stored at -20°C or -80°C.
- Short-term Storage: At 4°C, though moisture ingress must be prevented.
- After Reconstitution: The peptide is significantly more vulnerable to enzymatic degradation and temperature fluctuations.
For researchers studying metabolic pathways alongside other peptides, such as MOTS-c for mitochondrial function or 5-amino-1mq for NNMT inhibition, ensuring the stability of the Semaglutide component is vital for maintaining the integrity of co-administration studies.
Peptide Synergy in Research
In advanced research settings, Semaglutide is rarely studied in isolation. Investigating its interactions with GHRHs (Growth Hormone Releasing Hormones) like CJC-1295 or Ipamorelin allows scientists to explore the relationship between insulin sensitivity and growth hormone secretion. Furthermore, its combined use with tissue repair peptides like BPC-157 is a growing area of interest regarding systemic inflammation modulation.
However, these multi-peptide studies amplify the need for analytical validation. If the Semaglutide source is impure, it introduces uncontrolled variables into the study, making it impossible to determine which compound is responsible for the observed biological effect.
Checklist for Sourcing Semaglutide for Research
When evaluating a supplier for Semaglutide, researchers should insist on the following documentation:
- Batch-Specific HPLC: Shows the purity percentage and the resolution of the main peak.
- Mass Spectrometry (MS): Confirms the identity and molecular weight.
- Visual Inspection of Lyophilized Cake: A "proper" lyophilization should result in a solid, uniform cake, not a collapsing or "sticky" residue, which indicates high residual moisture or sugar fillers.
- Transparency regarding Salts: Is the peptide provided as a TFA salt, Acetate salt, or HCl salt? This affects solubility and cellular compatibility.
Conclusion: The Alpha Carbon Labs Commitment
The pursuit of scientific discovery requires materials that are beyond reproach. When you find Semaglutide for sale at Alpha Carbon Labs, you are not just purchasing a chemical; you are acquiring a validated research tool. Our commitment to rigorous quality control and transparent COA reporting ensures that your focus remains on the data, not the quality of your reagents.
Whether you are investigating the cardiovascular benefits of GLP-1s or exploring the synergistic effects of Cagrilintide and Semaglutide, starting with a validated, high-purity molecule is the only way to ensure the reproducibility and credibility of your research.
References
- 1. Mahapatra MK, Karuppasamy M, Sahoo BM. Semaglutide, a glucagon-like peptide-1 receptor agonist with cardiovascular benefits for management of type 2 diabetes. Rev Endocr Metab Disord. 2022.
- 2. Albericio F, Kruger HG. The Most Amazing De Novo Peptides of 2021. International Journal of Molecular Sciences. 2022.
- 3. Swarbrick J. Encyclopedia of Pharmaceutical Technology. CRC Press. (Peptide Synthesis and Validation).
- 4. World Health Organization (WHO). Alert No. 6/2023: Falsified semaglutide products. 2023.
- 5. Prasad-Reddy L, Isaacs D. A Review of Semaglutide: A Once-Weekly Glucagon-Like Peptide-1 Receptor Agonist. Annals of Pharmacotherapy. 2017.
- 6. Lau J, Bloch P, Schäffer L, et al. Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. Journal of Medicinal Chemistry. 2015.
- 7. D'Hondt M, Bracke N, Taevernier L, et al. Quality control of peptides: Analytical challenges and strategies. Journal of Pharmaceutical Analysis. 2014.
- 8. Evrard AF, et al. Analytical validation of therapeutic peptides by UHPLC-MS/MS. Journal of Chromatography B. 2021.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.