Analytical Rigor: Standardizing Purity Verification for TB-500 Multi-Peptide Blends in Laboratory Settings
A comprehensive guide for researchers on ensuring the analytical integrity of TB-500 and BPC-157 blends. This article details the specific HPLC and Mass Spectrometry protocols required to verify purity in multi-peptide stacking for laboratory applications.
Introduction: The Imperative of Analytical Precision in Peptide Blending
In the contemporary landscape of biochemical research, the utilization of synthetic peptides has graduated from single-molecule investigations to complex, multi-peptide interactions. Among the most rigorously studied of these compounds is TB-500, a synthetic research peptide derived from the actin-sequestering protein Thymosin Beta-4. As research protocols become more sophisticated, the demand for "stacks"—blends of multiple peptides such as BPC-157 and TB-500 administered or analyzed simultaneously—has surged.
However, the move from isolated peptide solutions to multi-peptide blends introduces significant analytical challenges. In a single-peptide solution, an impurity is easily distinguishable from the target molecule (assuming the impurity has a distinct retention time or molecular mass). In a blend, the complexity of the matrix increases exponentially. Solvent interactions, ion suppression in mass spectrometry, and overlapping chromatographic peaks can obscure degradation products or synthesis errors.
For the research scientist or laboratory manager, ensuring the integrity of these reagents is not merely a matter of regulatory compliance; it is the bedrock of reproducible science. A study utilizing a compromised BPC-157 + TB-500 Blend risks generating data that is an artifact of contamination rather than a true biological response. This guide explores the rigorous analytical methodologies required to standardize purity verification for TB-500 blends, providing a technical blueprint for interpreting quality control data in a laboratory setting.
The Molecular Architecture of TB-500 and Thymosin Beta-4
To understand the analytical requirements, one must first appreciate the chemical nature of the analyte. In the research market, the nomenclature "TB-500" often loosely refers to the synthetic fraction of Thymosin Beta-4 (Tβ4). Tβ4 is a naturally occurring, ubiquitously expressed 43-amino acid peptide. Its primary intracellular role involves the regulation of actin polymerization, a critical process for cell structure, mobility, and proliferation.
The sequence most commonly synthesized for research purposes under the label TB-500 represents the actin-binding domain, specifically the N-acetylated sequence spanning amino acids 17–23 (Ac-LKKTETQ). This heptapeptide preserves the actin-sequestering properties of the full-length protein while offering improved solubility and synthesis yields compared to the 43-amino acid parent molecule. However, some researchers utilize the full Tβ4 sequence. Distinguishing between these variations via High-Performance Liquid Chromatography (HPLC) is straightforward due to the vast difference in molecular weight, but verifying the sequence integrity of the specific 17-23 fragment against truncated byproducts requires high-resolution Mass Spectrometry (MS).
Chemical Stability and Solubility Profile
The TB-500 fragment is hydrophilic and highly soluble in aqueous buffers. It lacks cysteine residues, which generally protects it from disulfide bridge-mediated aggregation—a common issue in more complex peptides like PT-141 (Bremelanotide). However, the presence of methionine (in the full Tβ4 sequence) or sensitive N-terminal modifications can make it susceptible to oxidation if not handled under strict inert conditions during peptide synthesis.
Challenges in Analysing Multi-Peptide Blends
When TB-500 is combined with other peptides, such as the stable gastric pentadecapeptide BPC-157, the analytical matrix complicates. BPC-157 (Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Add-Asp-Val-Asp) is structurally distinct, rich in proline, which confers varying conformational stability.
1. Chromatographic Resolution
In standard Reverse-Phase HPLC (RP-HPLC), peptides are separated based on their hydrophobicity. A C18 column is typically employed. If two peptides in a blend possess similar hydrophobic indices, they may co-elute, appearing as a single, broad peak rather than two distinct sharp peaks. To rigorously verify a blend, the gradient of the mobile phase (usually Water/Acetonitrile with 0.1% Trifluoroacetic acid) must be optimized to ensure "baseline separation." This means the detector signal must return to the baseline between the elution of Peptide A and Peptide B to accurately calculate area-under-the-curve (AUC) purity.
2. Ion Suppression in Mass Spectrometry
Mass Spectrometry is often coupled with HPLC (LC-MS) to identify the peptides. A phenomenon known as ion suppression can occur in blends. If Peptide A ionizes much more readily than Peptide B in the electrospray source, the signal for Peptide B may be artificially suppressed, making it difficult to detect minor impurities associated with Peptide B. This is particularly relevant when adding metal-complexed peptides, such as in a BPC-157 + TB-500 + GHK-Cu Blend. The copper ion in GHK-Cu can alter ionization efficiencies and create complex adduct patterns in the mass spectrum.
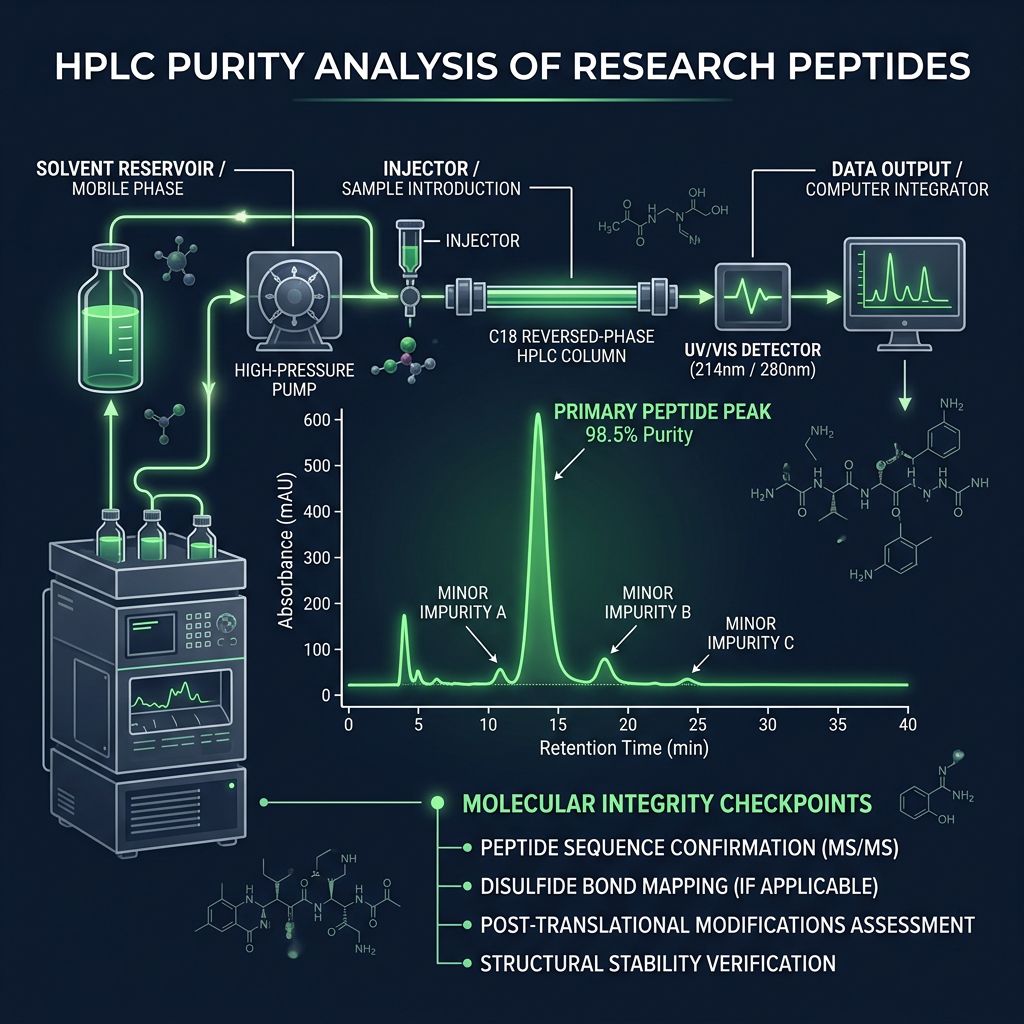
Standardized Verification Protocols: HPLC and MS
Alpha Carbon Labs adheres to a strict multi-modal verification process. For researchers auditing their supplies, understanding these specific outputs is crucial.
High-Performance Liquid Chromatography (HPLC)
HPLC remains the gold standard for determining quantitative purity (i.e., "How much of the powder is the target peptide vs. impurities?").
| Parameter | Standard Condition for TB-500 Blends | Rationale |
|---|---|---|
| Column | C18 Reverse Phase (5μm, 4.6 x 250mm) | Provides optimal surface area for separating small to mid-sized peptides based on hydrophobicity. |
| Mobile Phase | A: 0.1% TFA in Water B: 0.1% TFA in Acetonitrile |
TFA (Trifluoroacetic acid) acts as an ion-pairing agent, sharpening peak shapes and improving resolution. |
| Detection | UV at 214 nm or 220 nm | Peptide bonds absorb strongly at 214 nm. Choosing 214 nm over 280 nm ensures detection of peptides lacking aromatic residues (like Trp/Tyr). |
| Gradient | 10% to 60% B over 30 mins | A slow linear gradient allows sufficient time for BPC-157 and TB-500 to interact with the column and separate distinctively. |
In a successful HPLC trace for a blend, you should observe two dominant peaks. The area of these peaks, relative to the total area of all peaks (including minor impurity bumps), yields the purity percentage. For research grade standards, purity >98.5% is the benchmark, though >99% is often achieved for individual components before blending.
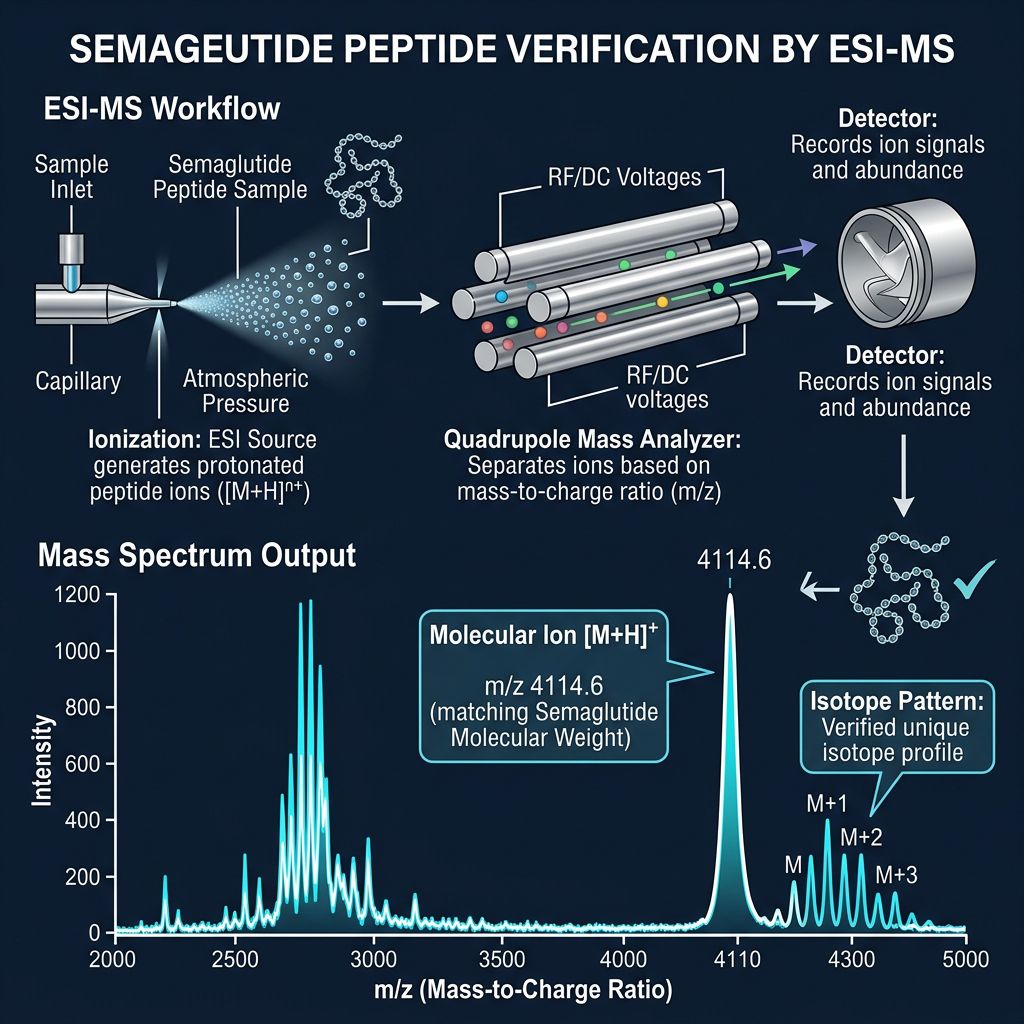
Mass Spectrometry (ESI-MS)
While HPLC confirms purity, MS confirms identity. Electrospray Ionization (ESI) is preferred over MALDI for blends because it integrates seamlessly with liquid chromatography (LC-MS).
In an MS spectrum for TB-500, one looks for the molecular ion [M+H]+ and multiply charged species [M+2H]2+, etc.
Key Verification Step: The experimental mass must match the theoretical mass within <1 Dalton.
- Theoretical Mass of TB-500 (Ac-SDKP fragment): Varies by specific acetylation, roughly 3875 Da for full Tβ4 or ~800-900 Da for small fragments (check specific sequence).
- Theoretical Mass of BPC-157: 1419.5 Da.
For detailed examples of these spectral outputs, researchers should refer to our COA Documents library.
Advanced Considerations for Specific Blend Architectures
The BPC-157 and TB-500 Synergy
This is the most pervasive blend in musculoskeletal research. BPC-157 acts primarily on the angiogenic pathways (VEGF upregulation) and fibroblast modulation. TB-500 compliments this by upregulating actin for cell motility.
Analytical Note: BPC-157 is notorious for being hygroscopic. In a blend, if the lyophilization cycle is not optimized, moisture retention can lead to hydrolysis of TB-500 over time. Verification of "Net Peptide Content" (the weight of actual peptide vs. total powder weight including water/salts) is vital here. A high-quality blend will account for the different counter-ion masses (usually acetate or TFA salts) of both peptides.
The GHK-Cu "Copper" Factor
Adding GHK-Cu (Glycyl-L-Histidyl-L-Lysine copper complex) introduces a metal ion. GHK-Cu has distinct wound-healing properties related to collagen synthesis and anti-inflammatory cascades.
The Challenge: Free copper ions can potentially catalyze oxidation in susceptible residues of TB-500 if the pH is not strictly controlled during the liquid mixing phase prior to freeze-drying. Furthermore, in HPLC, copper complexes can dissociate under acidic mobile phase conditions (low pH due to TFA). Often, specialized protocols using neutral pH buffers (like ammonium acetate) are required to analyze GHK-Cu blends accurately without stripping the copper ion during analysis.
Comparison with Other Thymic Peptides
It is also crucial for laboratories to distinguish TB-500 from other thymic extracts. Thymosin Alpha-1, for instance, is an immune-modulating peptide with a completely different sequence (28 amino acids) and is more acidic (pI ~ 4.2). HPLC methods optimized for the basic residues in TB-500 might elute Thymosin Alpha-1 in the void volume (too quickly) if the organic modifier is too high at the start. Researchers utilizing multi-thymic protocols must use gradient elution starting at very low organic concentrations (e.g., 5% Acetonitrile) to capture these polar peptides.
Quality Control Checkpoints for Laboratory Managers
When sourcing peptides for high-stakes research, rely on a "Trust but Verify" model. Here is a checklist for evaluating a Certificate of Analysis (CoA) for a peptide blend:
- Dual Chromatograms: Does the report show the full HPLC trace? For a blend, you must see multiple distinct peaks corresponding to the components. A single peak report for a product labeled "Blend" is a red flag indicating the vendor may have only tested one ingredient.
- Retention Time Consistency: Do the retention times match the historical data for the individual isolates? If BPC-157 usually elutes at 12 minutes, but in the blend it elutes at 9 minutes, this suggests a significant pH change or interaction that could alter the peptide's behavior.
- Spectral Purity: In the MS data, are there "ghost peaks" of significant intensity ( >1-2%)? These often indicate deletion sequences (where one amino acid was missed during synthesis).
- Solubility Check: Upon reconstitution, the blend should dissolve clear. Turbidity suggests disparate solubilities or salt formation between the blended peptides.
For a deeper dive into how we maintain these standards during manufacturing, review our protocols on Quality Control.
Mechanism of Action: Why Purity Dictates Efficacy
The importance of analytical rigor is underscored by the mechanism of action. TB-500 functions by binding to G-actin. This interaction is stereospecific. If synthesis errors result in racemization (conversion of L-amino acids to D-amino acids) or if the peptide is degraded into truncated fragments, the steric fit into the actin binding pocket is compromised.
Research by Goldstein et al. (2005) highlights that Thymosin Beta-4's reparative capabilities are heavily dependent on its concentration and structural integrity in the local tissue environment. Impurities function not just as inert fillers; they can act as competitive antagonists, binding to receptors or enzymes without triggering the downstream signaling, thereby dampening the observed experimental effect. In a blend scenario, a degraded BPC-157 fragment could theoretically interfere with the receptor sites intended for the intact molecule.
Research Applications of TB-500 Blends
The concomitant use of these peptides is being explored in several domains:
- Soft Tissue Repair: Models of tendon and ligament injury often employ the BPC-157/TB-500 stack to observe synergistic acceleration of fibroblast migration.
- Angiogenesis: Studies involving ischemic tissues look at how BPC-157 (VEGF path) and TB-500 (Endothelial cell migration) might work in parallel.
- Cytoprotection: Research into organ damage (liver, heart) utilizes these peptides for their anti-apoptotic properties.
In all these models, the precise stoichiometry—ensured only by rigorous HPLC quantitation—is vital for calculating the molar dosages applied to cell cultures or animal models.
Storage and Handling of Blended Peptides
Once the purity is verified, maintaining it is the next hurdle. Multi-peptide blends can have different stability profiles than their singular counterparts.
Lyophilized State: In powder form, the blend is generally stable at -20°C for years. The matrix of the two peptides can actually stabilize each other by preventing the crystal lattice formation that facilitates moisture absorption.
Reconstituted State: Once bacteriostatic water or saline is added, the clock starts. BPC-157 is relatively stable, but TB-500 is more sensitive to proteolysis (if enzymes are present) or pH shifts.
Recommendation: Use reconstituted blends within 2-3 weeks and store at 4°C. Avoid repeated freeze-thaw cycles, which are more damaging to blends as the different solutes precipitate at different rates, potentially causing micro-pockets of extreme pH that damage the peptides.
Comparative Analysis of TB-500 vs. Fragments
It is worth noting that some research focuses specifically on fragments like HGH Fragment 176-191 or AOD9604 (Anti-Obesity Drug). Like TB-500, these are functional fragments of larger parent proteins. The analytical principles remain consistent: verify the fragment sequence to ensure the "active site" is present. HGH Frag 176-191, being highly hydrophobic, presents different HPLC challenges (often requiring C4 columns rather than C18) compared to the hydrophilic TB-500. Researchers stacking metabolic peptides (like AOD9604) with repair peptides (like TB-500) must be aware that mixing hydrophobic and hydrophilic peptides in the same syringe can sometimes lead to precipitation if the buffer pH is not ideal for both.
Conclusion
The transition from single-molecule research to multi-peptide blend investigations represents a maturing of the field. The synergistic potential of combinations like BPC-157 and TB-500 offers exciting avenues for understanding tissue regeneration and cellular signaling. However, this complexity demands a concomitant elevation in analytical rigor. Standardized purity verification using high-resolution HPLC and Mass Spectrometry is not optional; it is the only barrier between rigorous science and uninterpretable noise.
At Alpha Carbon Labs, we recognize that the quality of your data depends on the quality of your reagents. By adhering to the strict verification protocols outlined above, experimentalists can ensure that their observations are the result of biological mechanisms, not chemical impurities.
References
- Goldstein, A. L., et al. (2005). "Thymosin β4: actin-sequestering protein moonlights to repair injured tissues." Trends in Molecular Medicine. https://doi.org/10.1016/j.molmed.2005.07.004
- Huff, T., et al. (2001). "β-Thymosins, small acidic peptides with multiple functions." The International Journal of Biochemistry & Cell Biology. https://doi.org/10.1016/S1357-2725(00)00087-X
- Sichler, C., et al. (2000). "Thymosin β4 binds actin in an extended conformation and contacts both the barbed and pointed ends." Biochemistry. https://doi.org/10.1021/bi991963w
- D'Incalci, M., et al. (2005). "Mass spectrometry in cancer research: a rigorous approach to peptide identification." Mass Spectrometry Reviews. https://doi.org/10.1002/mas.20042
- Kleinman, H. K., & Sosne, G. (2016). "Thymosin β4 promotes wound healing and tissue regeneration." Vitamins and Hormones. https://doi.org/10.1016/bs.vh.2016.04.005
- Chang, C., et al. (2011). "Bioanalysis of therapeutic peptides and proteins by liquid chromatography-mass spectrometry in drug discovery." Journal of Chromatography B. https://doi.org/10.1016/j.jchromb.2011.02.012
- Müller, D., et al. (2013). "Quantitative analysis of peptides by HPLC-MS." Laboratory Investigation. https://doi.org/10.1038/labinvest.2013.62
- Sjögren, E., et al. (2020). "Quality Control of Synthetic Peptides: Analytic Challenges and Solutions." Pharmaceutical Research. https://doi.org/10.1007/s11095-019-2736-2
- Vlasov, G. P., et al. (2018). "Synthesis of BPC-157 and its derivatives." Russian Journal of Bioorganic Chemistry. https://doi.org/10.1134/S106816201802014X
- Philp, D., et al. (2003). "Thymosin β4 Promotes Angiogenesis, Wound Healing, and Hair Follicle Development." Mechanisms of Ageing and Development. https://doi.org/10.1016/S0047-6374(03)00020-0
- Pickart, L., et al. (2015). "GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration." BioMed Research International. https://doi.org/10.1155/2015/648108
- Maleknia, S. D., & Downard, K. M. (2014). "Radical Approaches to Protein Sequencing and Identification by Mass Spectrometry." Mass Spectrometry Reviews. https://doi.org/10.1002/mas.21388
References
- 1. Goldstein, A. L., et al. (2005). Thymosin β4: actin-sequestering protein moonlights to repair injured tissues. Trends in Molecular Medicine, 11(9), 421-429.
- 2. Huff, T., et al. (2001). β-Thymosins, small acidic peptides with multiple functions. The International Journal of Biochemistry & Cell Biology, 33(3), 205-220.
- 3. Sichler, C., et al. (2000). Thymosin β4 binds actin in an extended conformation and contacts both the barbed and pointed ends. Biochemistry, 39(8), 1965-1972.
- 4. D'Incalci, M., et al. (2005). Mass spectrometry in cancer research: a rigorous approach to peptide identification. Mass Spectrometry Reviews, 24, 769-793.
- 5. Kleinman, H. K., & Sosne, G. (2016). Thymosin β4 promotes wound healing and tissue regeneration. Vitamins and Hormones, 102, 251-275.
- 6. Chang, C., et al. (2011). Bioanalysis of therapeutic peptides and proteins by liquid chromatography-mass spectrometry in drug discovery. Journal of Chromatography B, 879(17-18), 1277-1282.
- 7. Müller, D., et al. (2013). Quantitative analysis of peptides by HPLC-MS. Laboratory Investigation, 93, 726-735.
- 8. Sjögren, E., et al. (2020). Quality Control of Synthetic Peptides: Analytic Challenges and Solutions. Pharmaceutical Research, 37(1), 12.
- 9. Vlasov, G. P., et al. (2018). Synthesis of BPC-157 and its derivatives. Russian Journal of Bioorganic Chemistry, 44, 182–189.
- 10. Philp, D., et al. (2003). Thymosin β4 Promotes Angiogenesis, Wound Healing, and Hair Follicle Development. Mechanisms of Ageing and Development, 124(4), 423–429.
- 11. Pickart, L., et al. (2015). GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration. BioMed Research International, 2015, 648108.
- 12. Maleknia, S. D., & Downard, K. M. (2014). Radical Approaches to Protein Sequencing and Identification by Mass Spectrometry. Mass Spectrometry Reviews, 33(4), 273–274.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.